Sialidases (Neuraminidases): Functions in Infection and Cancer
Sialidases, also called neuraminidases, are glycosidases that remove terminal sialic acids from glycoconjugates and thereby reshape receptor exposure, molecular recognition, and downstream signaling. For researchers working on host-pathogen interaction, glycan remodeling, or tumor biology, access to high-quality reagents is often the limiting factor. At Creative Biolabs, our Anti-Sialidase/Neuraminidase Antibody Development Service supports studies of sialidase function, neuraminidase mechanism, desialylation, influenza drug target biology, and cancer metastasis with custom antibodies tailored for research use only.
Why Sialidases Matter in Modern Glycobiology
Sialic acids usually occupy the outermost position of glycan chains on glycoproteins and glycolipids. Because they are terminal residues, even limited enzymatic removal can produce major biological consequences. Desialylation can unmask galactose or N-acetylgalactosamine, alter electrostatic charge, change receptor accessibility, and modify the half-life or trafficking of glycoconjugates. For this reason, sialidases influence not only carbohydrate structure but also adhesion, immune recognition, pathogen spread, and tumor-associated signaling.
The term neuraminidase is commonly used in virology, especially for influenza virus enzymes, while sialidase is widely used for mammalian, bacterial, and broader enzymology contexts. In practice, both terms describe enzymes that hydrolyze terminal sialic acid residues, although their substrate preference, localization, and biological role can differ substantially.
| Topic | Core Function | Typical Consequence | Research Relevance |
|---|---|---|---|
| Sialidase function | Removes terminal sialic acid from glycans | Exposure of underlying glycan motifs | Glycan remodeling, receptor mapping |
| Neuraminidase mechanism | Substrate recognition, cleavage, product release | Altered particle release or cell-surface composition | Enzyme assays, inhibitor studies |
| Desialylation | Reduces terminal sialylation density | Changes in adhesion and immune response | Cancer and immunology models |
| Influenza drug target | Viral neuraminidase enables virion release | Drug blockade suppresses spread | Antiviral discovery and resistance research |
| Cancer metastasis | Altered sialylation supports motility and immune escape | More invasive phenotype | Biomarker and target validation |
How Sialidases Remove Terminal Sialic Acid
The catalytic event is simple in concept but broad in consequence. Sialidases recognize terminal sialic acid residues linked to glycan chains, engage the substrate in the catalytic pocket, and hydrolyze the glycosidic bond. Once the terminal sialic acid is removed, the molecular surface changes immediately. This can alter ligand binding, receptor clustering, glycoprotein turnover, and susceptibility to lectin-mediated recognition.
Substrate Access
The enzyme must first access a terminal sialylated epitope on a glycoprotein, glycolipid, or free oligosaccharide.
Catalytic Recognition
Binding geometry determines which linkage and glycan context can be processed efficiently.
Hydrolytic Cleavage
The glycosidic bond is hydrolyzed, releasing the terminal sialic acid and exposing the underlying sugar.
Biological Reset
Desialylation can reset receptor behavior, immune sensing, membrane interactions, and ligand engagement.
In mammalian systems, the four neuraminidases, NEU1, NEU2, NEU3, and NEU4, differ in subcellular localization and substrate preference. This matters experimentally. NEU1 is mainly associated with lysosomes, NEU2 is largely cytosolic, NEU3 is closely linked to the plasma membrane, and NEU4 has been reported in intracellular compartments including mitochondria. Bacterial and viral neuraminidases, by contrast, are often studied for their roles in colonization, nutrient acquisition, mucus penetration, or particle release.
Key Outcomes of Desialylation
- Exposure of galactose-containing structures previously masked by terminal sialic acid
- Changes in receptor residence time and endocytic routing
- Altered interactions with selectins, siglecs, antibodies, and lectins
- Reprogramming of cell adhesion, migration, and immune suppression
Common Experimental Readouts
- Lectin binding shifts after enzymatic treatment
- Loss of sialylated glycan signal in flow cytometry or histology
- Changes in viral spread, cell motility, or immune-cell engagement
- Direct neuraminidase activity by fluorogenic or colorimetric assays
Neuraminidase in Infection and Why It Is an Influenza Drug Target
Influenza virus neuraminidase is one of the clearest examples of how enzymatic desialylation drives infection. Hemagglutinin attaches influenza virions to sialylated receptors on host cells. After replication and budding, the newly formed virions remain vulnerable to re-binding sialylated structures on the producer cell and on neighboring mucus-associated decoy receptors. Viral neuraminidase solves this problem by cleaving terminal sialic acids. The result is more efficient release of progeny virions and improved spread through the local environment.
This neuraminidase mechanism explains why the enzyme became a classic influenza drug target. When small-molecule inhibitors occupy the active site, they compete with the natural substrate and block catalytic cleavage. Without efficient cleavage, virions remain trapped at the cell surface or in mucus-rich environments, reducing onward transmission in experimental systems. In research settings, neuraminidase inhibitors are therefore useful not only as antivirals but also as mechanistic tools for studying budding, release kinetics, and resistance mutations.
| Step | Role of Viral Glycoprotein | What Neuraminidase Contributes | Why Researchers Care |
|---|---|---|---|
| Attachment | Hemagglutinin binds sialylated receptors | Balances receptor engagement indirectly | Defines entry efficiency |
| Budding | New virions form at the membrane | Cleaves nearby terminal sialic acids | Supports release assays |
| Spread | Virions encounter mucus and cell-surface decoys | Removes decoy receptors that impede movement | Explains inhibitor activity |
| Drug pressure | Active-site occupancy reduces catalysis | Mutations may restore fitness or reduce binding | Resistance profiling |
Why the Active Site Matters
Influenza neuraminidase inhibitors were designed around the conserved catalytic machinery of the enzyme. That conservation is a major advantage for drug design, but it is also why resistance research remains important. Even when the catalytic core is constrained, nearby substitutions can affect pocket shape, local flexibility, hydrogen-bonding networks, or protein stability. For investigators studying neuraminidase mechanism, this balance between structural conservation and adaptive escape remains a central question.
Beyond Influenza
Sialidases in bacteria and other pathogens can support colonization by modifying mucus barriers, exposing cryptic receptors, or liberating sialic acid-containing nutrients. Although these systems differ from influenza neuraminidase, they reinforce the same principle: terminal sialic acid is not a passive decoration. It is a functional interface, and enzymes that trim it can profoundly alter infection biology.
How Sialylation and Desialylation Shape Cancer Progression
In cancer, the most common pattern is not widespread enzymatic stripping but dysregulated sialylation homeostasis. Tumor cells frequently display dense sialylated glycans on their surface, and this altered landscape can arise through increased sialyltransferase activity, reduced neuraminidase activity, or both. The net result is a more protective and signaling-competent glycocalyx.
The relevance to cancer metastasis is substantial. Terminal sialylation can support weak adhesion dynamics, endothelial interactions, immune suppression, and resistance to stress during dissemination. In parallel, experimental desialylation can alter engraftment, growth, and recognition in model systems. For researchers, this makes sialidase-related pathways valuable in both mechanistic studies and reagent development.
Immune Evasion
Sialylated glycans can engage inhibitory siglecs on immune cells and help generate a more suppressive microenvironment.
Migration and Invasion
Altered sialylation can influence integrin signaling, receptor turnover, and cell-cell or cell-matrix interactions linked to motility.
Surface Biomarker Remodeling
Sialyl-Lewis antigens, sialyl-Tn, gangliosides, and related motifs can serve as functional markers rather than passive signatures.
Research Questions Frequently Asked in This Area
- Which neuraminidase family member dominates in a given tumor model?
- Does desialylation change metastatic seeding, adhesion, or immune-cell activation?
- Which glycoproteins or glycolipids lose terminal sialic acid first?
- Can antibody tools distinguish enzyme expression from glycan outcome?
These questions often require highly selective antibodies because enzyme abundance, cellular localization, and functional glycan remodeling do not always move in parallel. A well-designed antibody panel can help separate expression biology from glycan-state biology and give a more interpretable picture of pathway activity.
Our Solution for Sialidase and Neuraminidase Research
Creative Biolabs provides custom antibody development services for researchers investigating mammalian sialidases, viral neuraminidases, and related glycan biology targets. We support discovery and validation workflows built around recombinant proteins, peptides, selected domains, and application-specific screening strategies.
Custom Anti-Sialidase/Neuraminidase Antibody Development
We generate antibodies against viral or mammalian neuraminidase targets with flexible antigen strategy, clone screening, and application-oriented validation.
Glycosylation Analysis
We support desialylation studies with glycan-focused analytical workflows that help connect enzyme biology to measurable glycan changes.
Sialoside Microarray
We provide sialylated substrate and binding-profile support for teams studying glycan recognition, substrate preference, and inhibitor-related binding changes.
Binding and Kinetic Characterization
We can characterize antibody-antigen interactions for affinity ranking and reagent selection in mechanistic studies.
What We Can Tailor to Your Project
- Target species, subtype, or isoenzyme focus
- Preference for monoclonal, recombinant, Fab, or scFv formats
- Validation for ELISA, western blot, IHC, IF, flow cytometry, or other research-use functional assays, where appropriate
- Specificity screening against homologs, related glycosidases, or engineered mutants
Request a Project Plan
If your team is studying influenza neuraminidase as a drug target, comparing mammalian sialidase isoforms, or evaluating glycan-state changes in cancer models, send us your target information, intended applications, and preferred validation strategy. We will build a practical research-use-only workflow around your experimental goal and timeline.
Talk to Our Scientific Team
Published Data
A recent open-access review in Cancers summarized aberrant sialylation as a common feature of many tumors and described how hypersialylation can contribute to tumor growth, metastasis, immune evasion, and therapy resistance. The review also highlights Siglec-sialoglycan signaling as an important mechanism by which tumor-associated sialylated glycans can shape an immunosuppressive tumor microenvironment. The figure below illustrates this immune-regulatory axis.
This review does not focus on individual sialidases or provide a direct comparison of specific neuraminidase targets. Instead, it supports the broader point that altered terminal sialylation is functionally relevant in cancer biology and worth characterizing with appropriate detection and analysis tools.
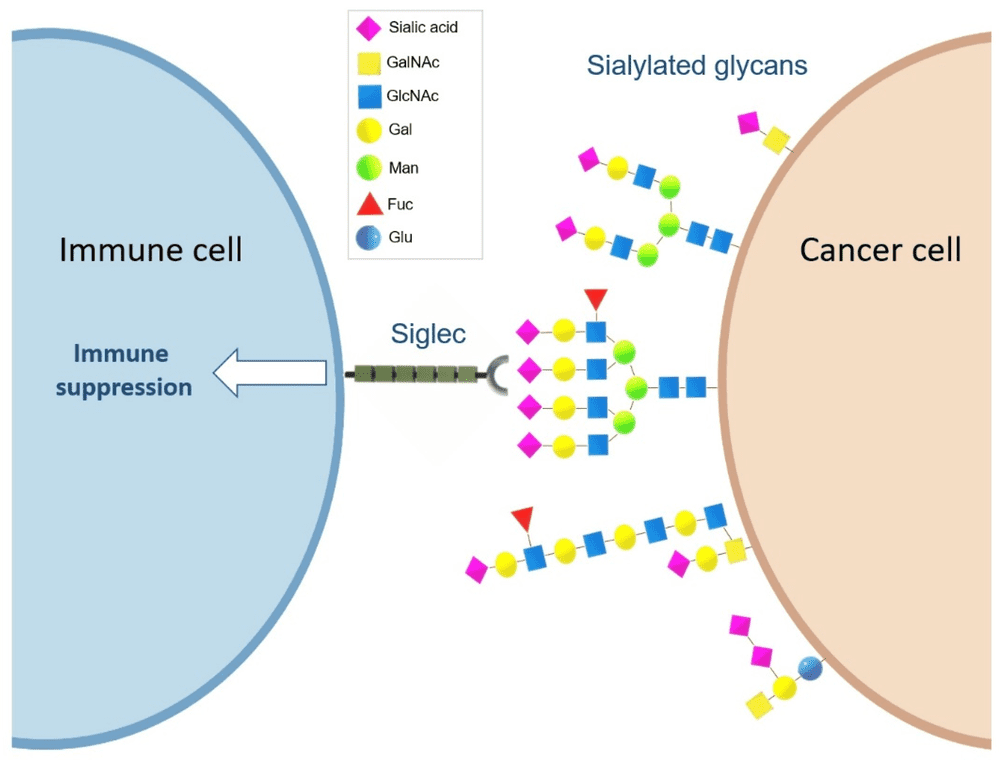 Fig.1 Siglec-sialoglycan interactions can modulate immune cell function and promote an immunosuppressive tumor microenvironment.1
Fig.1 Siglec-sialoglycan interactions can modulate immune cell function and promote an immunosuppressive tumor microenvironment.1
FAQs
What is the difference between sialidase and neuraminidase?
Both terms refer to enzymes that remove terminal sialic acid residues. Neuraminidase is used especially often for influenza and other viral enzymes, while sialidase is more common in mammalian and broader glycobiology contexts.
Why is influenza neuraminidase considered a drug target?
Influenza neuraminidase helps release progeny virions by cleaving terminal sialic acids from host-cell and mucus-associated receptors. Blocking the active site reduces efficient viral release and spread in research models.
Does desialylation always suppress cancer progression?
Not always. The outcome depends on enzyme identity, substrate context, cell type, and model system. Desialylation can change adhesion, immune recognition, and signaling, so the biological effect must be evaluated experimentally.
Can you generate antibodies against specific neuraminidase isoforms or viral subtypes?
Yes. We can design discovery strategies around mammalian isoenzymes, viral neuraminidase domains, subtype-specific proteins, or selected epitopes, with screening tailored to the intended assay format.
What samples are useful for a custom antibody project in this area?
Recombinant proteins, sequence information, peptide regions, mutant constructs, and intended application details are all useful. Even when no material is available, a project can often begin from target sequence and assay goals.
Can these antibodies be validated for multiple applications?
Yes. Validation can be arranged for ELISA, western blot, immunofluorescence, immunohistochemistry, flow cytometry, and related research assays, depending on project design and target behavior.
Are your services intended for clinical use?
No. The services and reagents described on this page are provided for research use only and are not intended for clinical diagnosis or treatment.
Reference:
- Munkley, Jennifer. Aberrant Sialylation in Cancer: Therapeutic Opportunities. Cancers 14.17 (2022): 4248. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/cancers14174248
