Glycosidases: Tools for Glycan Analysis and Modification
Creative Biolabs supports glycosidase-centered research through a combination of anti-glycosidase antibody development, substrate profiling, glycosylation analysis, and custom glycoprotein modification services. For teams working on glycan sequencing, exoglycosidase vs endoglycosidase workflows, deglycosylation strategies, or antibody glycoengineering, this service portfolio can help connect enzyme behavior with glycan readouts and downstream research objectives.
Why Glycosidases Matter in Glycan Research
Glycosidases are glycoside hydrolases that cleave glycosidic bonds in oligosaccharides, glycoconjugates, and glycoproteins. In glycoscience, they are not just degradative enzymes. They are analytical tools, structure-confirmation reagents, and, in selected engineered formats, glycan remodeling catalysts. Their value comes from selectivity. A well-characterized glycosidase can reveal whether a terminal residue is present, whether a linkage is accessible, and whether a core glycan can be selectively trimmed without disrupting the rest of the biomolecule.
That selectivity is why glycosidase applications remain central to glycan analysis. Researchers use these enzymes to decode branching, terminal capping, core structures, and site occupancy. In protein and antibody workflows, glycosidases are often combined with LC, HPLC, capillary electrophoresis, MS, glycopeptide mapping, and orthogonal binding assays. The enzyme step does not replace instrumentation. It sharpens interpretation.
Core Classes of Glycosidases Used in Glycan Analysis
In practice, glycosidase workflows are often built around whether the enzyme removes terminal sugars one residue at a time or cleaves within the glycan core. That distinction shapes experimental design, data interpretation, and the level of structural certainty that can be achieved.
| Enzyme Type | Cleavage Pattern | Typical Readout | Research Value |
|---|---|---|---|
| Exoglycosidases | Remove terminal residues from the non-reducing end | Stepwise shifts in chromatographic, electrophoretic, or MS profiles | Useful for glycan sequencing, terminal residue confirmation, and linkage-sensitive interpretation |
| Endoglycosidases | Cleave within the glycan, often near the chitobiose core | Collapsed glycoform distributions or core-remnant generation | Useful for deglycosylation tools, glycoform simplification, and antibody glycoengineering workflows |
| Peptide-N-glycosidases | Release entire N-glycans from glycoproteins | Released glycan pool for fluorescent labeling or MS analysis | Useful for global N-glycan profiling and orthogonal confirmation after intact analysis |
Exoglycosidase vs Endoglycosidase
Exoglycosidases are best when the question is which terminal sugars are present and in what order. Endoglycosidases are best when the goal is to remove or remodel broader glycan segments, especially on antibodies and Fc-fusion proteins. Many robust workflows use both classes sequentially.
How Glycosidases Support Glycan Sequencing
Glycan sequencing does not usually mean direct base-by-base sequencing in the nucleic acid sense. In glycoscience, it refers to structured deduction of glycan composition, branching, and linkage features through orthogonal evidence. Exoglycosidases are particularly important here because they provide controlled, interpretable changes.
A typical analytical logic may involve released glycans, fluorescent labeling, baseline profiling, sequential digestion, and post-digestion comparison. Each enzyme exposure narrows the structural possibilities. When paired with a reference library, retention-time shifts and mass changes become much more informative than a single untreated trace.
- Terminal neuraminidase treatment can reveal whether sialic acid capping contributes to peak complexity.
- Galactosidase digestion can distinguish galactosylated from agalactosylated structures.
- Fucosidase or mannosidase steps can help differentiate core and branch-specific features in appropriate contexts.
For antibody workflows, this is especially useful because Fc glycans frequently exist as related populations rather than a single species. Enzyme-guided interpretation helps determine whether differences arise from galactosylation, fucosylation, sialylation, mannose retention, or a combination of these traits.
Where Deglycosylation Tools Add the Most Value
Deglycosylation tools are often used before high-resolution analysis, not only after it. Removing all or part of the glycan can simplify spectra, expose peptide backbones, improve site mapping confidence, and make isoform differences easier to compare across lots or constructs. In antibodies, controlled deglycosylation can also help researchers evaluate how glycan heterogeneity contributes to observed functional differences.
For Released-Glycan Studies
PNGase-based release enables global glycan pool analysis. This is often followed by labeling, cleanup, and LC-MS or HILIC-based profiling.
For Site-Specific Glycoproteomics
Partial or selective digestion can preserve enough structural information to support confident assignment of glycosylation sites and local glycoforms.
For Antibody Characterization
Endoglycosidases can remove Fc glycans down to a core remnant, supporting glycoform simplification, engineered conjugation, or comparative functional testing.
For Enzyme Specificity Studies
Substrate panels and microarrays can show which glycan motifs are efficiently processed and which remain resistant under defined assay conditions.
Glycosidases in Antibody Glycoengineering
Antibody glycoengineering has expanded far beyond cell line optimization. Enzyme-enabled remodeling now supports more controlled post-expression modification of antibody glycans. This is particularly relevant when researchers need a narrower glycoform distribution, a defined conjugation handle, or a more mechanistically interpretable antibody material for downstream assays.
Endoglycosidases such as EndoS2-related systems are important in this field because they can trim native Fc glycans while preserving a useful acceptor site on the antibody. In engineered workflows, the remaining core can then be rebuilt with selected glycan donors or functionalized intermediates. This makes it possible to compare more homogeneous glycoforms more directly than with heterogeneous starting material.
For research applications, this matters in several ways:
- it improves control in structure-function studies focused on Fc receptor binding or complement-related readouts;
- it supports antibody-drug conjugation strategies that use Fc glycans as defined loading sites;
- it enables cleaner comparison between native, trimmed, and remodeled glycoforms in assay development.
These workflows should still be interpreted carefully. Enzyme accessibility depends on glycan context, protein structure, subclass, and reaction conditions. For that reason, glycoengineering projects benefit from combining enzymology, substrate profiling, and analytical confirmation rather than relying on a single reaction endpoint.
Our Solutions for Glycosidase-Centered Glycan Research
Creative Biolabs provides complementary services for teams that need to study glycosidases themselves, profile enzyme-substrate relationships, characterize glycan outcomes, or generate modified glycoproteins for research. These services are especially useful when your project spans more than one layer of the workflow, from enzyme reagent development to glycan readout and remodeled product confirmation.
Anti-Glycosidase (Glycoside Hydrolase) Antibody Development Service
This service fits projects that need high-specificity research antibodies against glycosidases, including enzymes used as pathway markers, remodeling factors, or functional targets. It is particularly useful when you need orthogonal detection of the enzyme itself alongside glycan-level readouts.
Glycosyltransferase & Glycosidase Substrate Microarray
This platform is well suited for mapping enzyme specificity across diverse substrates. It helps define preferred motifs, resistant structures, and assay conditions for glycosidase applications in analytical development and enzyme engineering.
Glycosylation Analysis Service
This service supports released-glycan profiling, site-specific analysis, and orthogonal glycoprotein characterization. It is a strong fit when glycosidase digestion needs to be connected to rigorous LC-MS or multi-method interpretation.
Custom Glycosylation of Proteins
This option is relevant for protein and antibody studies that require designed glycoforms, glycan remodeling support, or project-specific glycosylation strategies for research-stage material preparation and confirmation.
How We Typically Support This Type of Project
| Project Need | Typical Starting Point | Recommended Service | Expected Output |
|---|---|---|---|
| Need to study glycosidase expression or localization | Defined enzyme target or isoform panel | Custom Anti-Glycosidase Antibody Development Service | Research antibodies for WB, IHC, IF, or related assays |
| Need to compare enzyme specificity across glycan motifs | Purified enzyme, mutant, or assay design question | Glycosidase Substrate Microarray Service | Specificity profile, preferred substrates, quantitative interaction data |
| Need to interpret digestion outcomes at the glycan level | Protein, antibody, cell-derived sample, or released glycan pool | Comprehensive Glycosylation Analysis Service | Annotated glycan profile and structure-focused analytical report |
| Need remodeled protein glycoforms for downstream research | Protein or antibody candidate with defined design goal | Custom Glycosylation of Proteins Service | Research-stage modified material and confirmation data |
Ready to Scope Your Glycosidase Project?
If you are planning a glycan sequencing workflow, evaluating exoglycosidase vs endoglycosidase strategies, building deglycosylation tools into analytical development, or testing antibody glycoengineering concepts, share your sample type, target glycan class, and preferred readout with our team. We can help align the enzyme question with a practical assay and data package.
Request a Glycosidase Research Quote
Published Data
A 2023 open-access study evaluated two endoglycosidase-catalyzed chemoenzymatic glycan remodeling routes for generating glycosite-specific trastuzumab conjugates. In one route, antibodies were remodeled through a one-pot strategy using EndoS2 to trim native Fc glycans and introduce a functionalized disaccharide handle in the same reaction system. In the second route, antibodies were first deglycosylated to a GlcNAc-containing precursor and then rebuilt with a modified sialylated complex-type glycan through a glycosynthase-enabled transglycosylation step.
The study reported that both routes generated highly homogeneous ADCs with minimal aggregation, strong rat-serum stability, and potent activity against HER2-positive BT-474 cells after payload coupling. In a BT-474 xenograft model, these glycosite-specific conjugates showed strong antitumor activity without apparent body-weight loss at the tested doses. The study mainly supports the value of endoglycosidase-enabled Fc glycan remodeling in research-stage antibody modification and conjugation workflows.
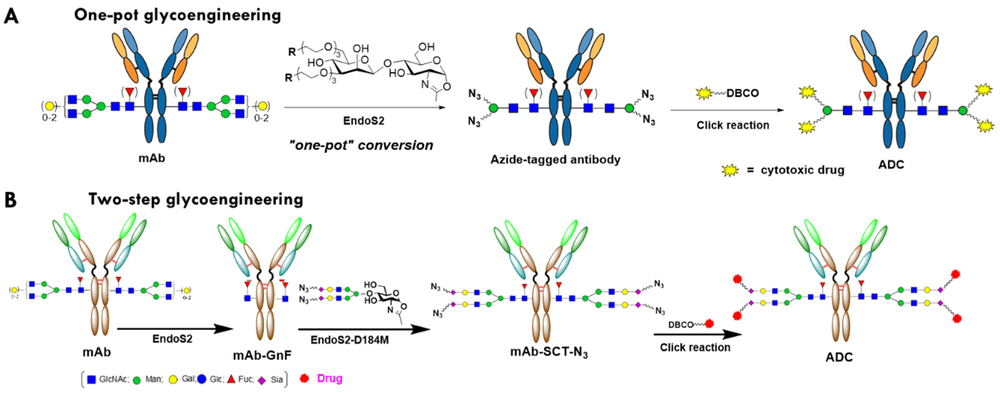 Fig.1 Antibody glycan remodeling workflow for site-specific conjugation.1
Fig.1 Antibody glycan remodeling workflow for site-specific conjugation.1
FAQs
What is the main difference between exoglycosidase and endoglycosidase workflows?
Exoglycosidase workflows are mainly used for residue-by-residue structural confirmation, while endoglycosidase workflows are mainly used to trim broader glycan regions or create defined glycan acceptors for remodeling studies.
Are glycosidases enough on their own for glycan sequencing?
Usually no. Glycosidases work best when paired with LC, MS, electrophoretic profiling, or reference standards. The enzymes create interpretable shifts, and the analytical platform turns those shifts into structural evidence.
Which samples are commonly analyzed in glycosidase-based glycan studies?
Common samples include purified glycoproteins, monoclonal antibodies, Fc-fusion proteins, released glycan pools, enzyme preparations, cell-derived materials, and selected complex matrices after suitable cleanup.
Can glycosidases be used for antibody glycoengineering as well as analysis?
Yes. Certain endoglycosidases and engineered glycosynthase variants are widely used in research workflows to trim Fc glycans and rebuild them with selected structures or functionalized intermediates.
When should I consider a substrate microarray instead of a standard digestion assay?
A substrate microarray is useful when you need broad specificity profiling across many glycan motifs, want to compare enzyme variants, or need higher-throughput evidence for substrate preference before narrowing the assay design.
Can Creative Biolabs help if I need both enzyme profiling and glycan readout?
Yes. We can combine substrate-focused evaluation with downstream glycosylation analysis so enzyme behavior can be interpreted together with glycan structural readouts.
Are these services intended for clinical diagnosis or treatment decisions?
No. The services described on this page are provided for scientific research use only and are not intended for clinical diagnosis or therapeutic decision-making.
Reference:
- Yang, Q., Chen, H., Ou, C., Zheng, Z., Zhang, X., Liu, Y., Zong, G., and Wang, L.-X. Evaluation of Two Chemoenzymatic Glycan Remodeling Approaches to Generate Site-Specific Antibody–Drug Conjugates. Antibodies 12.4 (2023): 71. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/antib12040071
