Anti-N-acetyllactosamine Antibody Development Service
N-acetyllactosamine (LacNAc) and its polymerized forms (poly-LacNAc) represent fundamental structural motifs in glycobiology, serving as backbones for blood group antigens and selectin ligands. At Creative Biolabs, we provide comprehensive services for generating high-specificity antibodies against these ubiquitous yet structurally complex glycans. Our expertise extends to specialized capabilities in Non-Human Glycan Antibody Development. Whether your research focuses on stem cell markers, tumor glycosylation patterns, or immune cell trafficking, our platform is designed to deliver robust reagents that advance your scientific discovery.
Background: The Significance of LacNAc and Poly-LacNAc
N-acetyllactosamine (LacNAc) is a disaccharide unit composed of Galβ1-4GlcNAc. It serves as the "Type 2" carbohydrate chain backbone, distinguishing it from the "Type 1" chain (Galβ1-3GlcNAc, or lacto-N-biose). LacNAc units can be repeated by the action of beta-1,3-N-acetylglucosaminyltransferases (such as B3GNT enzymes) to form linear poly-N-acetyllactosamine chains, also known as poly-LacNAc or the "i-antigen". These linear chains can be further modified by beta-1,6-N-acetylglucosaminyltransferases (such as GCNT2) to create branched structures, referred to as the "I-antigen".
The expression of LacNAc and poly-LacNAc is developmentally regulated and critical for numerous biological processes. In undifferentiated stem cells, specific high-molecular-weight poly-LacNAc profiles serve as markers of pluripotency. In oncology, the overexpression or alteration of poly-LacNAc on cell surface glycoproteins (such as EGFR and CD44) is frequently observed in metastatic cancer cells. These structures act as scaffolds for terminal fucosylation and sialylation, generating ligands for selectins (sialyl-Lewis X) and galectins, thereby facilitating tumor cell adhesion, migration, and immune evasion.
Structural Diversity and Classification
Understanding the precise linkage and branching of LacNAc units is crucial for antibody specificity. The table below outlines the primary structural variations and their defining characteristics:
| Antigen Type | Glycan Structure | Key Characteristic | Primary Distribution |
|---|---|---|---|
| Type 2 Chain (LacNAc) | Galβ1-4GlcNAc | Ubiquitous building block; backbone for ABH antigens | Glycoproteins, Glycolipids |
| Type 1 Chain (LNB) | Galβ1-3GlcNAc | Isomeric to LacNAc; distinct conformation | Secretions, intestinal mucins |
| i-Antigen | Linear Poly-LacNAc | Repeating unbranched units (Galβ1-4GlcNAc)n | Fetal erythrocytes, stem cells |
| I-Antigen | Branched Poly-LacNAc | C6-branched GlcNAc structures | Adult erythrocytes, mucosal epithelia |
Functional Implications in Physiology and Pathology
The presentation of LacNAc residues on cell surfaces dictates critical molecular interactions. These glycans are not merely structural scaffolds but active participants in cellular signaling:
- Immune Regulation: LacNAc termini are often capped with sialic acid or fucose to form Sialyl-Lewis X, the key ligand for Selectins (E-, P-, and L-selectin), mediating leukocyte rolling and extravasation during inflammation.
- Cancer Progression: Altered expression of glycosyltransferases (e.g., Mgat5) in tumor cells leads to increased β1,6-branching (I-antigen). These structures bind Galectins, forming a lattice that stabilizes growth factor receptors (like EGFR/VEGFR) on the cell surface, thereby enhancing pro-survival signaling and metastasis.
- Pathogen Interaction: Many viruses and bacteria exploit LacNAc structures as entry receptors. For instance, specific influenza strains and human noroviruses recognize distinct LacNAc-containing histo-blood group antigens.
Challenges in Developing Anti-LacNAc Antibodies
Despite their importance, developing a high-affinity lacnac antibody is challenging due to inherent characteristics of carbohydrate antigens:
Structural Similarity
Differentiating between Type 2 (Galβ1-4GlcNAc) and Type 1 (Galβ1-3GlcNAc) chains requires antibodies with precise linkage specificity to avoid cross-reactivity.
Topology Recognition
Distinguishing linear poly-LacNAc (i-antigen) from branched poly-LacNAc (I-antigen) often depends on the antibody's ability to recognize the three-dimensional clustering of the glycan.
Low Immunogenicity
As "self" antigens widely distributed in the body, LacNAc structures are poorly immunogenic, often necessitating specialized immunization protocols or synthetic antigen designs.
Context Dependence
The presentation of poly-LacNAc on specific protein carriers (e.g., LAMP-1, Integrins) can influence antibody binding, requiring robust validation on native biological samples.
Project Workflow
Our Service Portfolio
Creative Biolabs offers a flexible antibody development service tailored to the your needs. We utilize advanced platforms to ensure the successful generation of high-quality reagents.
Hybridoma Development
Our classic monoclonal antibody development pipeline uses optimized immunization schedules with adjuvants specifically designed for carbohydrate antigens. This approach is ideal for generating high-affinity IgG or IgM antibodies that can recognize repetitive poly-LacNAc epitopes on cell surfaces.
Phage Display Technology
For targets that are non-immunogenic in standard hosts, our synthetic and immune phage display libraries provide a powerful alternative. This allows for the selection of binders under strictly controlled conditions, enabling the isolation of antibodies that discriminate between subtle structural differences, such as sulfated vs. non-sulfated LacNAc.
Specific Linkage Targeting
We develop antibodies with defined specificity for:
- Terminal LacNAc (Galβ1-4GlcNAc-R)
- Internal Poly-LacNAc repeats ([-3Galβ1-4GlcNAcβ1-]n)
- Branched Poly-LacNAc (I-antigen structures)
- Linear Poly-LacNAc (i-antigen structures)
Validation & Characterization
Every antibody undergoes extensive validation. We employ glycan microarrays to map the binding profile against hundreds of related structures. We also perform flow cytometry on cell lines with known glycosyltransferase expression profiles (e.g., CHO vs CHO-Lec8 mutants) to confirm biological reactivity.
Key Features and Benefits
Strict Specificity
Validated discrimination between Type 1 and Type 2 chains to prevent experimental artifacts.
Versatile Formats
Available as IgG, IgM, scFv, or VHH to suit various assay sensitivities and tissue penetration needs.
Data-Driven
Detailed kinetic analysis and epitope mapping provided with every deliverable.
Cost-Effective
Scalable production options that maintain high batch-to-batch consistency.
How to Start
Initiating a project with Creative Biolabs is straightforward. Simply provide us with details regarding your target specificity (e.g., linear vs. branched LacNAc) and the desired end-use application. Our scientific team will design a customized immunization and screening strategy. We offer flexible milestones, allowing you to review data at the end of the immunization and screening phases before proceeding to production.
Inquire Now
Published Data
Recent clinical investigations utilizing spatially resolved mass spectrometry imaging have unveiled a distinct remodeling of the N-glycome during breast cancer metastasis. Analysis of patient-matched primary and metastatic tissues revealed a progressive accumulation of specific glycan signatures along the metastatic trajectory. Beyond the elevation of high-mannose structures, the study identified a critical enrichment of complex branched and fucosylated N-glycans in distant metastases. Crucially, this structural shift correlates with transcriptomic data showing significant overexpression of N-acetylglucosaminyltransferases, such as MGAT5, and poly-LacNAc synthases like B3GNT3 in metastatic lesions.
These enzymes drive the formation of β1,6-branched structures, which serve as scaffolds for poly-N-acetyllactosamine (poly-LacNAc) extension. The resulting aberrant glycosylation on cell surface proteins, including EpCAM, enhances protein stability and modulates cell adhesion properties essential for metastatic dissemination. This clinical evidence underscores the functional importance of complex LacNAc-bearing glycans in disease progression. By targeting these specific tumor-associated carbohydrate antigens, researchers can potentially disrupt key metastatic mechanisms. Our antibody development platform generates high-affinity reagents against poly-LacNAc and branched glycan epitopes for preclinical research, aiding biomarker validation and target characterization.
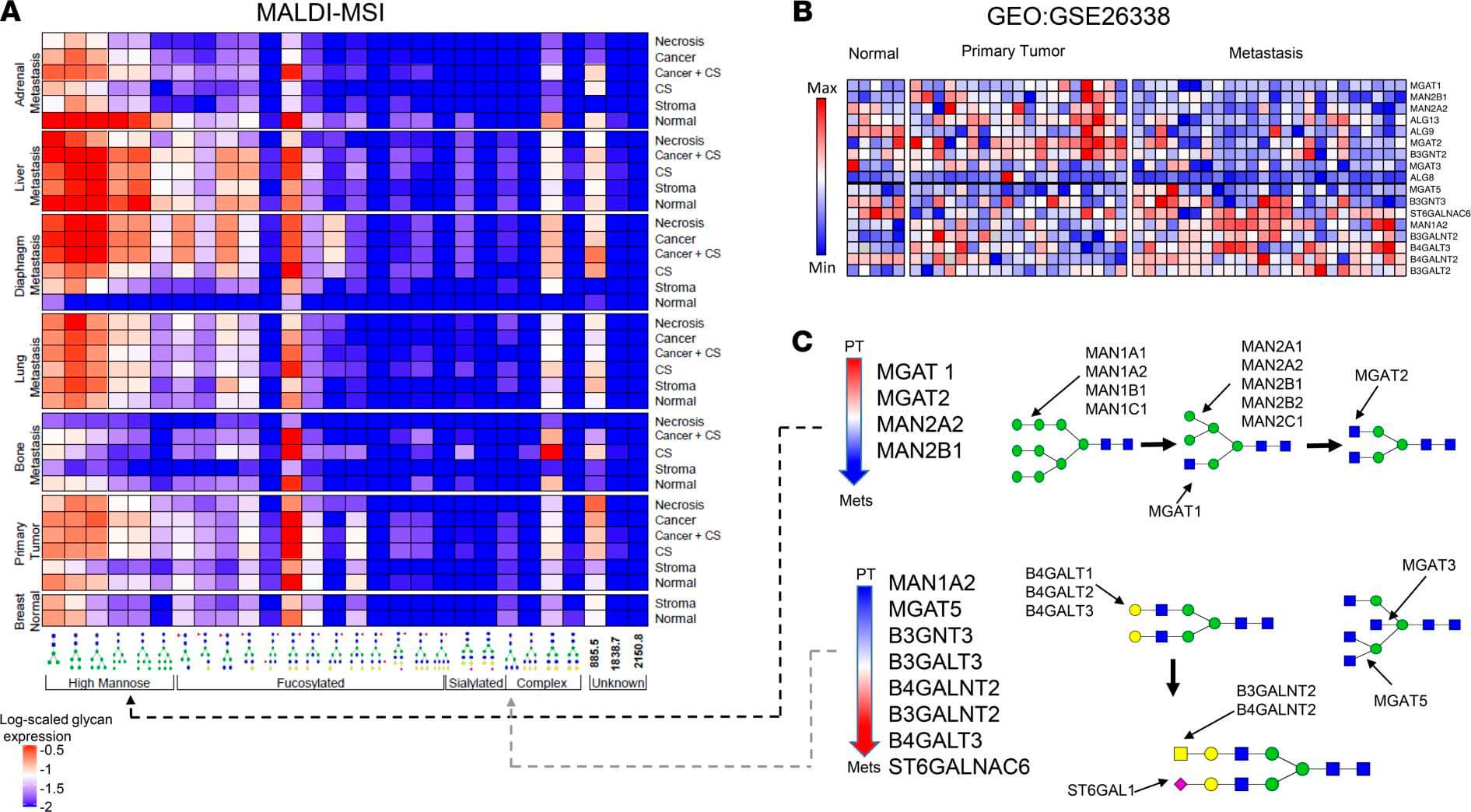 Fig.1 Integrated N-glycomic and transcriptomic analysis reveals upregulation of complex
branched glycan biosynthesis in metastasis.1
Fig.1 Integrated N-glycomic and transcriptomic analysis reveals upregulation of complex
branched glycan biosynthesis in metastasis.1
FAQs
What is the difference between LacNAc and Lacto-N-biose?
LacNAc (N-acetyllactosamine) is a Type 2 chain structure with a Galβ1-4GlcNAc linkage. Lacto-N-biose is a Type 1 chain structure with a Galβ1-3GlcNAc linkage. Antibodies must be carefully screened to distinguish between these two isomers due to their high structural similarity.
Can you generate antibodies specific for Poly-LacNAc?
Yes. We can generate antibodies that recognize the internal repeating units of poly-LacNAc chains (i-antigen) or the branched structures (I-antigen). This is achieved through the use of specific long-chain neoglycolipids or cells overexpression specific glycosyltransferases as immunogens.
What applications are these antibodies suitable for?
Our anti-LacNAc antibodies are commonly used for flow cytometry (to detect cell surface expression), immunohistochemistry (tissue staining), and Western blotting. They are valuable tools for stem cell characterization, tumor phenotyping, and analyzing immune cell subsets.
How do you validate the specificity of the antibodies?
We employ a comprehensive validation strategy that includes glycan microarray profiling against a library of defined glycan structures (including Type 1 and Type 2 chains) and cell-based binding assays using wild-type and glycosyltransferase-deficient cell lines.
What is the typical timeline for a custom antibody project?
A standard monoclonal antibody development project typically takes 4-6 months, depending on the complexity of the antigen and the screening requirements. We provide a detailed timeline upon project initiation.
What Our Customers Say
"The anti-LacNAc antibody we received showed remarkable specificity. We were struggling with cross-reactivity against Type 1 chains, but Creative Biolabs delivered a clone that worked perfectly in our flow cytometry assays."
"Developing antibodies against poly-LacNAc was a major bottleneck for our metastasis project. Their team suggested a phage display approach that yielded high-affinity binders in a surprisingly short time."
"Excellent communication and professional reports. The epitope mapping data provided with the antibody gave us great confidence in our downstream applications. Highly recommended."
"We needed a custom IgM for a specific branched poly-LacNAc structure. Creative Biolabs handled the antigen synthesis and immunization flawlessly. The final product is very stable."
Reference:
- Ščupáková, K. et al. "Clinical importance of high-mannose, fucosylated, and complex N-glycans in breast cancer metastasis." JCI Insight vol. 6,24 (2021): e146945. Distributed under Open Access license CC BY 4.0. https://doi.org/10.1172/jci.insight.146945
