Immune Checkpoint Reporter Assay
Immune Checkpoint: A Valuable Therapeutic Target
The balance of co-stimulatory and inhibitory immunological checkpoints is closely linked to immune response modulation, which is employed by many cancers to escape immune system attack. Currently, drugs and therapies designed to target immune checkpoints, such as anti-PD-1 and anti-PD-L1 antibody medications, are demonstrating remarkable efficacy in fighting cancers. However, they face other hurdles, such as dissatisfied overall response rates for low mutational load and side consequences. To overcome these difficulties and accelerate cancer therapy development, a thorough knowledge of immunological checkpoints is extremely important.
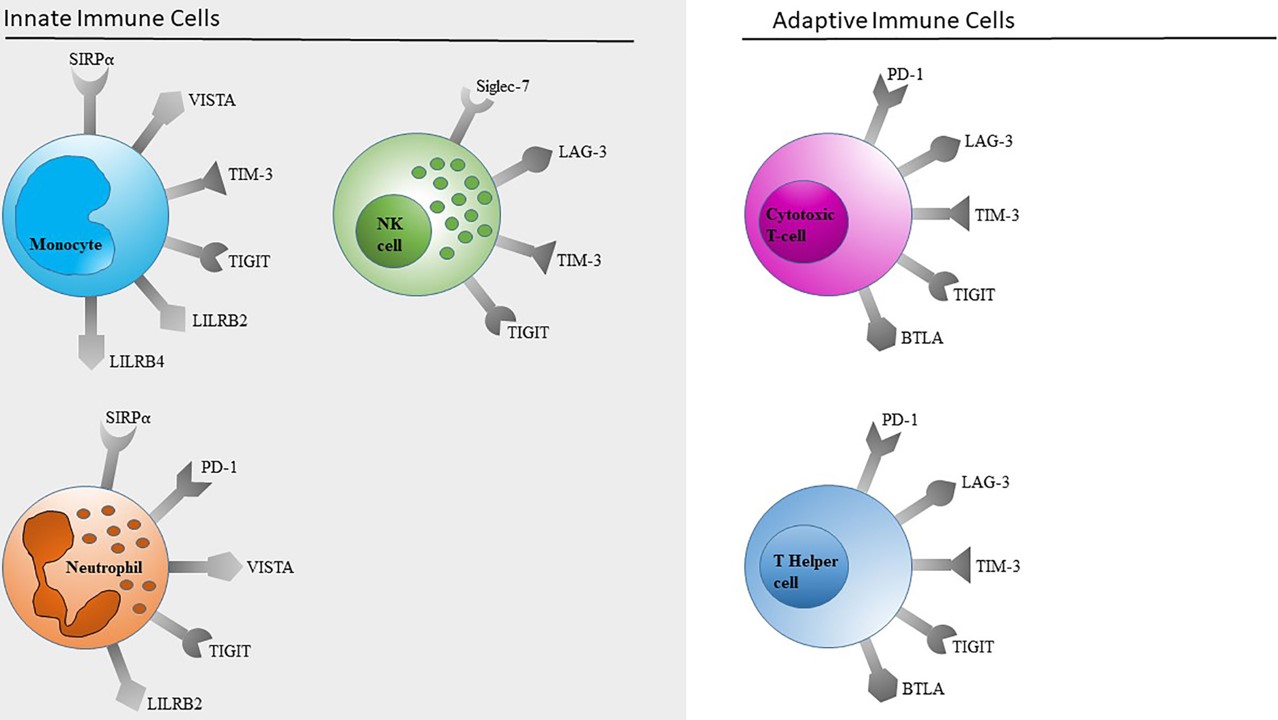 Fig.1 The expression profile of immune checkpoints.1
Fig.1 The expression profile of immune checkpoints.1
Our Immune Checkpoint Reporter Assay
To overcome these difficulties and accelerate cancer therapy development, Creative Biolabs provides a cost-effective immune checkpoint reporter assay service. As a result of their adaptability and efficiency, reporter gene assays are crucial tools in the study of pharmacological compounds. In our immune checkpoint reporter assay, we utilize the reporter gene reaction in response to evaluate antibody drug efficiency. Powered by experts with extensive expertise, Creative Biolabs provides a complete package encompassing everything from design to data qualification, all with the goal of meeting global customers' diverse demands.
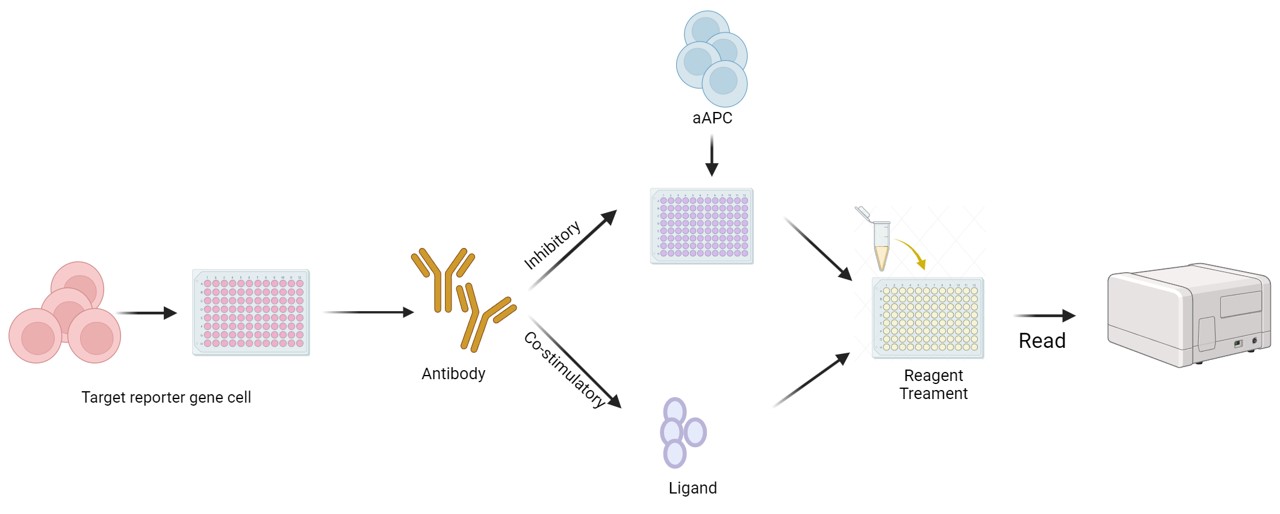 Fig.2 Workflow of our immune checkpoint reporter assay.
Fig.2 Workflow of our immune checkpoint reporter assay.
Our Advantages
- Platform
We have established cell engineering platforms and successfully produced various stable and functional cell lines, which are often picked for their projects by our global customers. Furthermore, our well-established bioassay platform has been applied for tens of thousands of experiments and is now highly mature.
- Research Team
Our research team consists of many experts with experience and expertise. Moreover, our research team keeps up with the pace of development and always equips themselves with the most advanced knowledge. Together with our competent research team, we have more confidence in presenting desirable outcomes for global customers.
- Service
We provide one-stop customized services encompassing everything from design to data qualification to satisfy our global customers' diverse needs.
Assay Type
| Assays | Targets |
| Immune Checkpoint Activation Assay | OX-40; GITR; CD-40; 4-1BB |
| Immune Checkpoint Inhibition Assay | PD-1; TIGIT; PD-L1; LAG3; CTLA-4; TIM-3 |
| Dual Target Bioassay | PD-1/TIGIT; PD-1/4-1BB; PD-1/LAG3; PD-1/CTLA-4 |
| Cytokine and Cellular Signaling-related Bioassay | VEGF-VEGFR2; TIE2-ANG2; IL-1β; IL-33; IL-1α; IL-36 |
| Myeloid cell checkpoint Bioassay | CD47-SIRPα |
Work with Creative Biolabs
Creative Biolabs strives to improve our technologies and services for the purpose of aiding in drug development. In this field, whether you're looking to target co-inhibitory, co-stimulatory, or a mix of receptors on immunological checkpoints, we have the resources to fulfill your demands. For more details about our immune checkpoint assay, please don't hesitate to contact us.
Reference
-
Shibru, Benjamin, et al. "Detection of immune checkpoint Receptors–A current challenge in clinical flow cytometry." Frontiers in Immunology 12 (2021): 694055.
Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only.
