Carrageenan Air Pouch Modeling & Pharmacodynamics Service
Creative Biolabs offers comprehensive services, including the establishment of this model, drug efficacy evaluation, and the customization of protocols to meet the specific needs of your research. We ensure high-quality, reliable results and expert support throughout the process.
Introduction
Acute inflammation is a rapid and localized response of the body's immune system to harmful stimuli, such as infection, injury, or irritants. It is characterized by the classic signs of redness, heat, swelling, pain, and loss of function. This physiological process is essential for protecting the body by eliminating harmful agents and initiating tissue repair. However, if acute inflammation becomes dysregulated or prolonged, it can contribute to the development of chronic inflammatory diseases such as rheumatoid arthritis, asthma, inflammatory bowel disease, and cardiovascular disorders. The inflammatory response involves the activation of immune cells, the release of inflammatory mediators (like cytokines, prostaglandins, and histamines), and changes in blood flow that lead to increased vascular permeability. These changes enable immune cells to reach the site of injury or infection. While acute inflammation is crucial for recovery, it must be tightly regulated to prevent tissue damage and disease progression.
Carrageenan Air Pouch Model and Applications
The Carrageenan Air Pouch Model involves injecting carrageenan into a subcutaneous air pouch created in rodents, typically in the dorsum. This method induces an acute inflammatory response, which can be measured by assessing changes in the inflammatory exudate, including immune cell infiltration and cytokine release. However, the model's limitation lies in its focus on acute inflammation, not chronic inflammatory diseases. The response can vary based on carrageenan dosage and animal strain.
- Simulates: It is commonly used to mimic the inflammatory processes involved in conditions such as rheumatoid arthritis and osteoarthritis, where inflammatory cytokines and mediators play a crucial role in tissue damage and disease progression.
- Evaluates Drugs: It also helps assess the efficacy of analgesics for pain relief and immune-modulating therapies aimed at reducing the severity of inflammation and tissue damage.
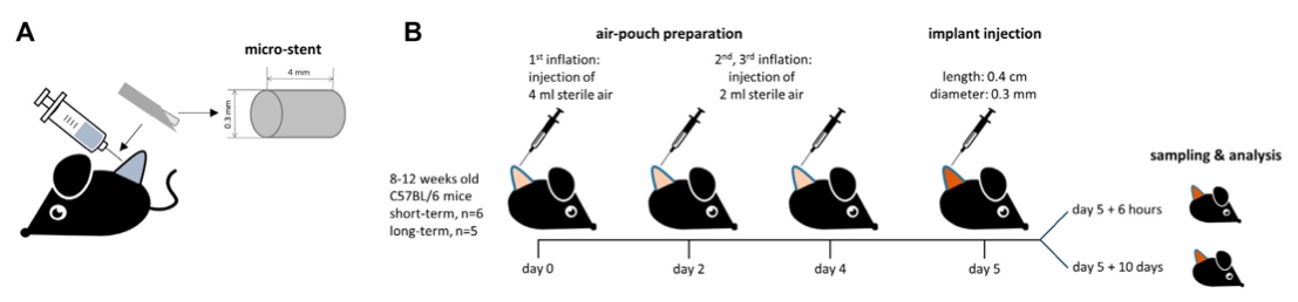 Fig. 1 Implant and air-pouch study design.1,3
Fig. 1 Implant and air-pouch study design.1,3
Evaluation Platform
- Animals: Mouse, Rat, Hamster, Rabbit, Cat, Dog, NHPs.
-
Measurements
We provide comprehensive measurements for assessing drug efficacy in the Carrageenan Air Pouch Model, utilizing state-of-the-art technologies such as:- General observations: Inflammatory exudate volume, body weight, and mortality rate.
- Histological analysis: Tissue inflammation, immune cell infiltration, tissue damage.
- Cytokine profiling: TNF-α, IL-1β, IL-6 levels via ELISA.
- Flow cytometry: Immune cell subtypes in the pouch exudate.
- Gene/protein expression analysis: RT-qPCR and Western blot to assess inflammatory mediators.
Our services are designed to provide you with a detailed understanding of the therapeutic potential of drugs for inflammatory diseases, supported by expert guidance at each project stage.
Our advantages
- Reproducibility: Highly consistent results across different experiments.
- Comprehensive Analysis: Multi-dimensional assessment of both local and systemic responses.
- Expert Support: Dedicated scientific team to assist in model design and data interpretation.
- Customizable Protocols: Tailored solutions for specific research needs, based on literature and prior studies.
- Time-efficient: Fast model setup and data collection, ideal for early-stage drug screening.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
1. What is the typical duration for setting up the Carrageenan Air Pouch Model?
The setup takes approximately 1-2 days for the procedure, with data collection following over 24-72 hours based on study goals.
-
2. Can this model simulate chronic inflammatory conditions?
This model primarily evaluates acute inflammation. For chronic models, we recommend other established systems or long-term treatment protocols.
-
3. What type of animals are used for this model?
The Carrageenan Air Pouch Model is typically conducted in rodents, with rats and mice being the most common species.
-
4. Can I use this model for drug testing beyond anti-inflammatory agents?
While it's ideal for anti-inflammatory drug testing, modifications can allow for broader research applications, including immunomodulatory and pain-relief treatments.
-
5. Are the results from this model directly translatable to human conditions?
While the model effectively simulates inflammation, direct translation requires additional validation studies in human-based models.
Published Data
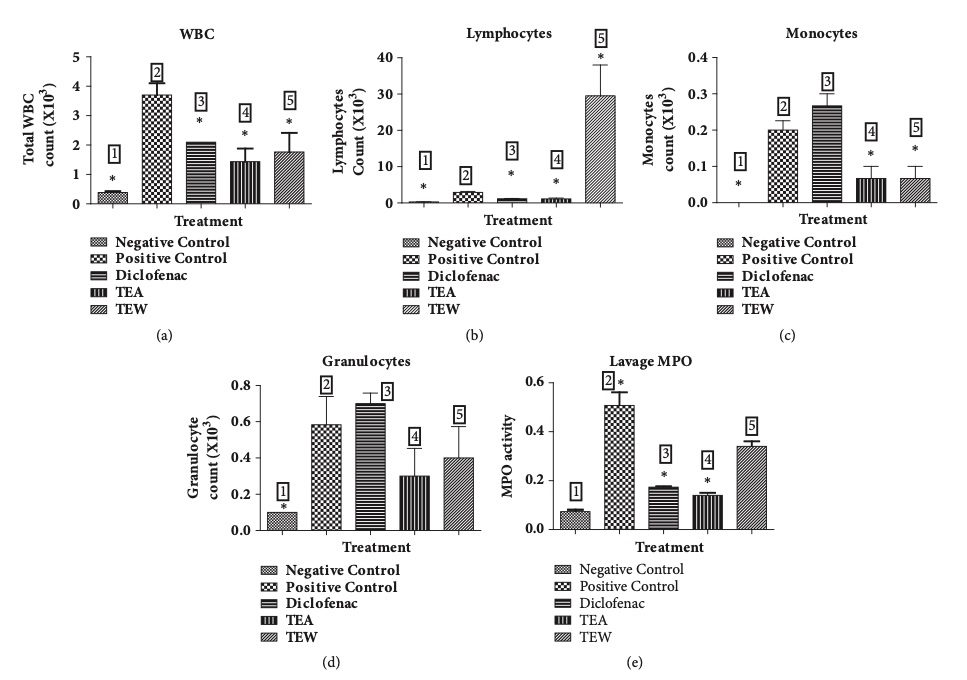 Fig. 2 Carrageenan-induced air pouch inflammation in Wistar rats.2,3
Fig. 2 Carrageenan-induced air pouch inflammation in Wistar rats.2,3
Carrageenan-induced air pouch inflammation was assessed in Wistar rats, with the effects of diclofenac and TTE/TTW treatment evaluated. Diclofenac administration significantly reduced the total white blood cell (WBC) count, lymphocytes, and myeloperoxidase (MPO) activity, but did not significantly affect the granulocyte or monocyte populations. In contrast, treatment with TTE and TTW at an oral dose of 500 mg/kg resulted in a marked decrease in total WBC, lymphocytes, monocytes, granulocytes, and MPO activity in the air pouch lavage. These results were compared to the positive control group, as illustrated in Figure 2.
References
- Woitschach, Franziska et al. "Utilization of a highly adaptable murine air pouch model for minimally invasive testing of the inflammatory potential of biomaterials." Frontiers in bioengineering and biotechnology vol. 12 1367366. 26 Apr. 2024. https://doi.org/10.3389/fbioe.2024.1367366
- Jitta, Srinivasa Reddy et al. "Terminalia tomentosa Bark Ameliorates Inflammation and Arthritis in Carrageenan Induced Inflammatory Model and Freund's Adjuvant-Induced Arthritis Model in Rats." Journal of toxicology vol. 2019, 7898914. 17 Jan. 2019..https://doi.org/10.1155/2019/7898914
- Distributed under Open Access license CC BY 4.0, without modification
For Research Use Only.
