Lung Injury/Pneumonia Modeling & Pharmacodynamics Services
Introduction
Lung injury and pneumonia represent critical global health burdens, characterized by high morbidity, mortality, and substantial socioeconomic costs, with pneumonia alone contributing to over 2.5 million deaths annually. The complexity of these diseases, encompassing dysregulated inflammation, alveolar barrier disruption, intricate pathogen-host interactions, and systemic organ crosstalk, necessitates the development of robust preclinical models to facilitate the translation of mechanistic insights into clinical applications. Creative Biolabs has developed validated lung injury/pneumonia models. These models effectively mimic key pathological hallmarks, including neutrophilic infiltration, edema, and hypoxemia. They accelerate the development of targeted interventions to address unmet clinical needs by enabling the dissection of pathogenic mechanisms like cytokine storms and microbial virulence, evaluating novel therapeutics, such as anti-inflammatory agents, vaccines, and optimizing diagnostic biomarkers.
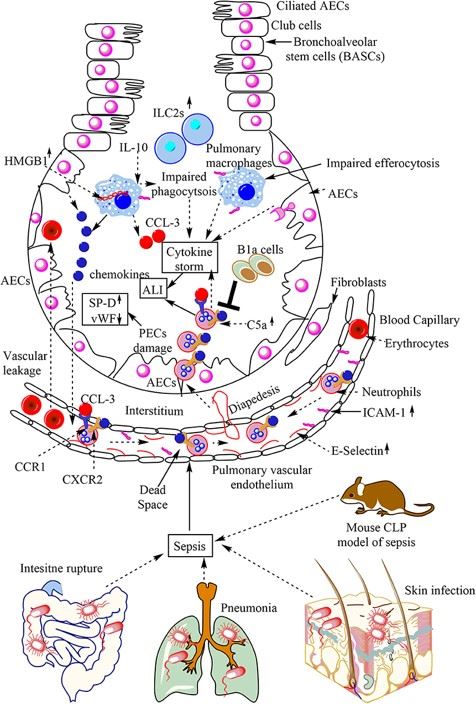 Fig.1 Overview of sepsis-induced ALI. Local infections of the skin (S. aureus), lungs (pneumonia), and intestinal commensal bacteria that leak into the blood may lead to sepsis development.1,3
Fig.1 Overview of sepsis-induced ALI. Local infections of the skin (S. aureus), lungs (pneumonia), and intestinal commensal bacteria that leak into the blood may lead to sepsis development.1,3
Available Lung Injury/Pneumonia Models
Creative Biolabs offers a comprehensive portfolio of preclinical models to simulate human lung injury and pneumonia:
| Lung Injury/Pneumonia Models | Clinical Relevance | Primary Research Applications | Animal Species |
| LPS-Induced Pulmonary Neutrophilia Model | Acute bacterial inflammation, excessive neutrophil recruitment, and early inflammatory signaling (TLR4). | Chemokine Receptor Antagonists (e.g., CXCR2 antagonists), TLR4 signaling inhibitors, and early-stage anti-inflammatories. | Mouse, Rat |
| LPS-Induced Acute Lung Injury (ALI) Models | Sepsis-induced ALI/ARDS, barrier dysfunction, pulmonary edema. | Vasculoprotective Agents (e.g., Angiopoietin mimetics, Statins), Anti-Pulmonary Edema Drugs, and Glucocorticoids. | Mouse, Rat, Rabbit |
| Sephadex G-50-Induced Acute Lung Injury (ALI) Model | Acute injury from particulates/non-infectious emboli, role of macrophages. | Macrophage Function Modulators, Cell Protective Agents (e.g., NAC), and Anti-fibrotic agents (for long-term effects). | Mouse, Rat |
| High Oxygen Environment-Induced Acute/Chronic Lung Injury Model | Hyperoxia-induced injury (BPD in neonates, mechanical ventilation), oxidative stress. | Novel Antioxidants (e.g., SOD mimetics, Vitamin E analogs), and Anti-remodeling Drugs (e.g., Pirfenidone for chronic phase). | Mouse, Rat |
| Papain-Induced ALI Models | Emphysema, chronic inflammation, and elastase activity imbalance. | Matrix Metalloproteinase (MMP) Inhibitors (e.g., Doxycycline), Elastase Inhibitors (e.g., Alpha-1 antitrypsin), and PDE4 Inhibitors. | Mouse, Rat |
| TNF-alpha and IL-17A-Induced ALI Model | Cytokine storm, autoimmune exacerbations, severe viral infection. | Specific Cytokine Inhibitors (e.g., Infliximab, Secukinumab), and targeted Immunomodulators. | Rat |
| Immune Complex-Induced ALI Model | Autoimmune/hypersensitivity pneumonitis, antibody-mediated damage, and complement activation. | Complement System Inhibitors (e.g., Eculizumab), and general Immunosuppressants (e.g., Cyclophosphamide). | Mouse, Rat |
| A. Baumanii-infected Pneumonia Model | Hospital-Acquired/Ventilator-Associated Pneumonia caused by multi-drug resistant bacteria. | Novel Antibiotics (e.g., Colistin, Sulbactam combos, Tigecycline), Vaccine Candidates, and Host Immune Enhancing Therapies. | Mouse, Rat |
| Poly(I:C)-Induced ALI Model | Severe viral pneumonia, injury mimicking double-stranded RNA viruses. | Antiviral Agents (e.g., Ribavirin), Interferon Response Modulators, and TLR3 pathway Inhibitors. | Mouse, Rat |
Evaluation Platform
Our advanced detection platform ensures highly accurate and precise drug development results, thereby accelerating your R&D process:
- Histopathology: Pathological methods, such as H&E staining, are used to assess alveolar architecture, immune infiltration, and collagen deposition.
- Immunology: Flow cytometry is utilized to quantify immune cell subsets and activation states. ELISA and MSD are employed to measure cytokines (e.g., IL-1β, TNF-α), chemokines, and acute-phase proteins. qPCR/RNA-seq are used to profile gene expression and molecular pathways.
- Microbial Load: CFU and PCR are used to evaluate pathogen clearance and drug efficacy.
- Lung Function: FlexiVent is used to measure compliance, resistance, and elastance.
- Omics detection: Metabolomics or genomics are employed to identify dysregulated pathways.
- In Vivo Imaging: MRI and micro-CT are utilized to track real-time disease progression and resolution.
Applications
- Mechanistic Studies: To dissect the complex biology of disease progression, pinpointing core pathogenic drivers, signaling pathways, and cell-cell communication.
- Drug Screening: To rapidly and precisely test novel compounds against specific genetic or molecular defects in the disease pathway.
- Pharmacodynamics (PD) / Efficacy Testing: To validate the therapeutic efficacy of lead drug candidates and study how the drug affects the body (PD).
- Regulatory Validation: To generate high-quality, traceable data that meets regulatory standards for submitting an Investigational New Drug (IND) application.
Our advantages
- Diverse model portfolio: Species-specific models, such as rodents, non-rodents, along with other species, and pathogen/strain-matched systems.
- End-to-end solutions: Offering comprehensive solutions from model establishment to data analysis and manuscript figure preparation, thereby ensuring translational relevance.
- Customization services: Providing customized models tailored to address specific requirements or comorbidities.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: How do you ensure model success rate?
A: We validate models via pilot studies (n=3-5 animals) to optimize induction parameters, such as LPS dose, to ensure success rate exceeds 90% for standard models.
-
Q: What sample types and endpoints are available for analysis?
A: Samples include lung tissue, BALF, serum, and peripheral blood, and endpoints may include survival, histopathology, lung function, and molecular readouts such as cytokines, gene expression, and microbiological (CFU) analyses.
-
Q: Do you offer long term/chronic lung injury models?
A: Yes. We develop chronic models through repeated low-dose LPS exposure, enduring viral infection, or bleomycin, with monitoring extending up to 12 weeks.
-
Q: How do you choose between rodent and large animal models?
A: Rodents like mice or rats are cost-effective for mechanistic studies and high-throughput drug screening. Large animals like pigs or NHPs are preferred for translational studies, such as PK/PD, and device testing, due to their anatomical/physiological similarity to humans.
-
Q: Are immunocompromised models available?
A: Certainly. We provide models with neutrophil depletion, genetically engineered mice, and immunosuppressant pretreatment.
-
Q: Can you support combination therapy testing?
A: Yes. Our platforms enable evaluation of drug-drug or drug-device synergies.
-
Q: How quickly can a custom model be developed?
A: Turnaround time ranges from 4-8 weeks, depending on complexity.
-
Q: Do you offer human tissue-based assays?
A: We utilize primary human alveolar cells and lung organoids for ex vivo validation.
Published Data
The ferret acute lung injury model induced by LPS derived from Pseudomonas aeruginosa displayed alveolar structural disruption and fibrin deposition in lung tissue, confirming that this model effectively replicates the pathological features of human acute lung injury.
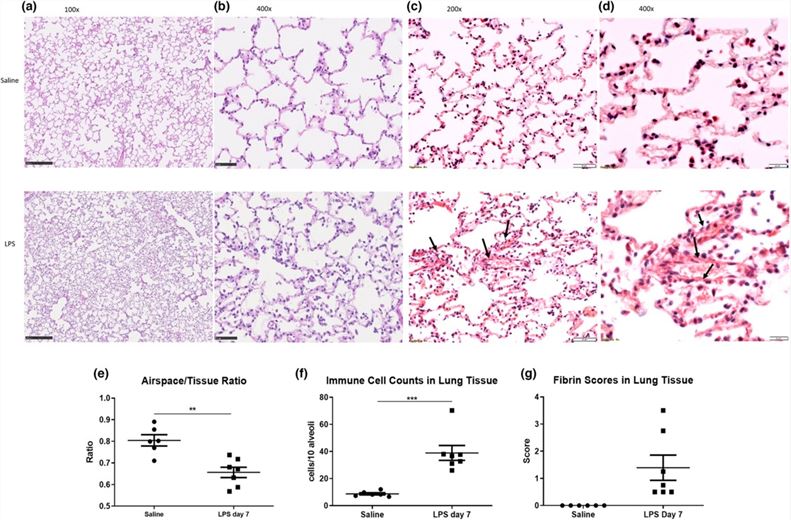 Fig.2 Lung histological changes in ferrets following saline and/or LPS administration.2,3
Fig.2 Lung histological changes in ferrets following saline and/or LPS administration.2,3
References
- Kumar, Vijay. "Pulmonary Innate Immune Response Determines the Outcome of Inflammation During Pneumonia and Sepsis-Associated Acute Lung Injury." Frontiers in immunology vol. 11 1722. https://doi.org/10.3389/fimmu.2020.01722
- Khoury, Oula et al. "Ferret acute lung injury model induced by repeated nebulized lipopolysaccharide administration." Physiological reports vol. 10,20 (2022): e15400. https://doi.org/10.14814/phy2.15400
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
