Tumor Modeling & Pharmacodynamics Services
Introduction
As cancer remains an increasingly severe global public health challenge, its staggering global incidence and mortality rates underscore the urgent need for more effective treatments. Creative Biolabs leverages its extensive experience to develop in vivo and in vitro tumor models. These animal models serve as an indispensable cornerstone in preclinical pharmacology and efficacy studies. These models are core tools that bridge the gap between basic science and clinical translation, recapitulate key features of various human tumors, and are used for validating tumor genesis mechanisms, drug discovery, screening drug candidates, evaluating therapeutic efficacy, and personalized therapy development.
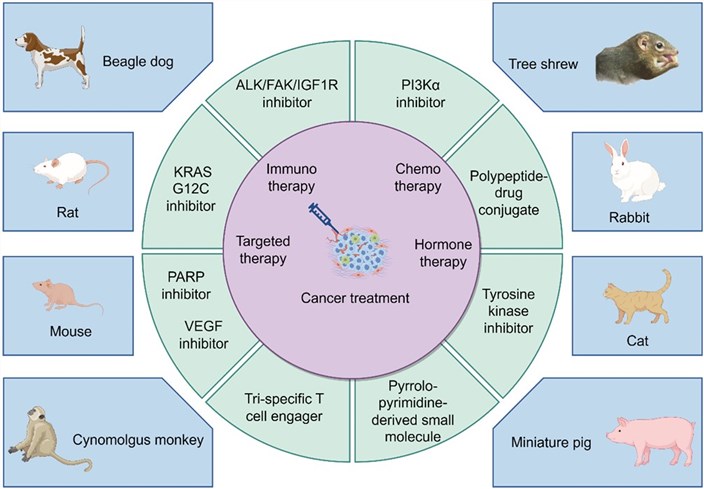 Fig.1 A schematic diagram illustrating the interaction between cancer treatment and preclinical animal models.1,4
Fig.1 A schematic diagram illustrating the interaction between cancer treatment and preclinical animal models.1,4
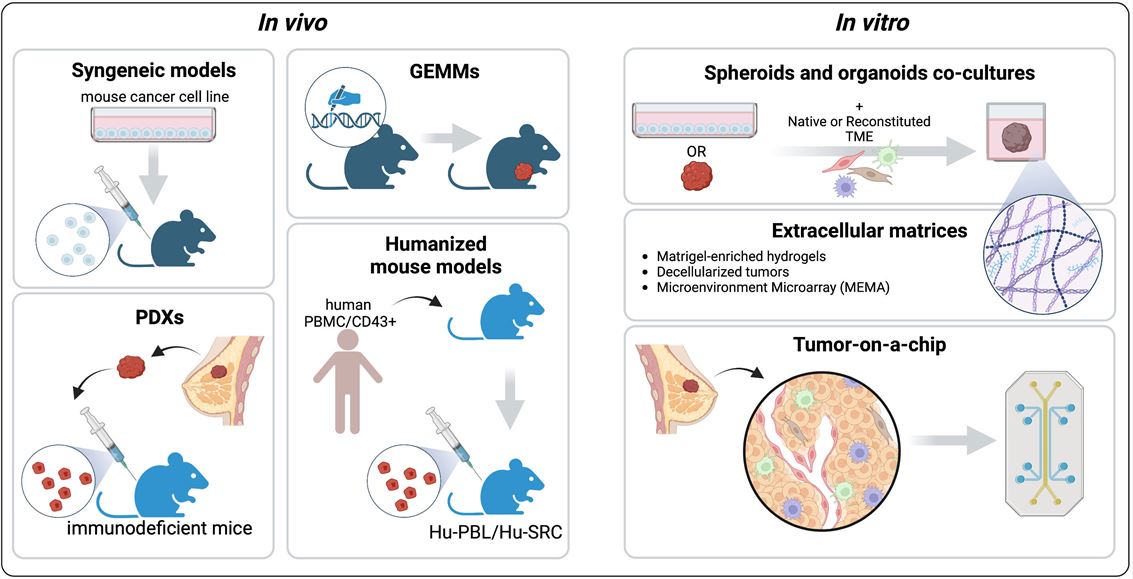 Fig.2 Preclinical models to recapitulate the tumor microenvironment (TME).2,4
Fig.2 Preclinical models to recapitulate the tumor microenvironment (TME).2,4
Available Tumor Models
Creative Biolabs offers a diverse range of in vivo and in vitro models to accurately and cost-effectively mimic human tumor characteristics, facilitating robust efficacy evaluation across various therapeutic modalities.
| Models | Types | Applications | |
|
Tumor Seeding Type Models | Subcutaneous Tumor Model | Subcutaneous models enable screening, Orthotopic models ensure clinical relevance, Angiogenesis models evaluate anti-angiogenics, and Systemic models are the gold standard for anti-metastasis. |
| Orthotopic Tumor Model | |||
| Angiogenesis Model | |||
| Intraperitoneal/Intraorgan Injection Model | |||
| Systemic (Disseminated) Model | |||
|
Syngeneic Models | Syngeneic Cell Line derived Tumor Model | Using immortalized mouse tumor lines in immune-competent mice, they are the gold standard for studying the complete tumor-immune microenvironment, essential for cost-effective immunotherapy proof-of-concept data. |
| Syngeneic Mice derived Tumor Model | |||
|
Xenograft Models | Cell Line derived Xenograft (CDX) Model | Xenograft Models utilize human tumor cells/tissues in immunodeficient mice, foundational for screening and validating targeted/cytotoxic oncology therapies with high clinical relevance and offering a pathway for personalized medicine. |
| Patient derived Xenograft (PDX) Model | |||
| Immunodeficient-Rat Xenograft Model | |||
|
Humanized Models | Humanized PDX (hu-PDX) Mouse Model | Utilize immunodeficient mice reconstituted with human immune components (e.g., PBMCs or HSCs), often combined with PDX, to study human immune response against human tumorsin vivo. Crucial for novel human biologics and cell therapies. |
| Humanized CDX (hu-CDX) Mouse Model | |||
| Humanized Tumor Target Mouse Model | |||
|
Carcinogen induced Mouse Tumor Models | Carcinogen induced Mouse Tumor Model | Induce tumors via specific chemicals to simulate natural disease progression and molecular pathways, providing valuable insight into etiology, prevention, and inflammation-driven carcinogenesis treatments. |
|
Spontaneous Tumor Animal Models | Common Spontaneous Mouse Tumor Model | Tumors naturally arising in specialized strains that closely mimic human cancer progression and characteristics, facilitating chronic and comprehensive treatment research. |
| Gene-Edited Spontaneous Mouse Tumor Model | |||
|
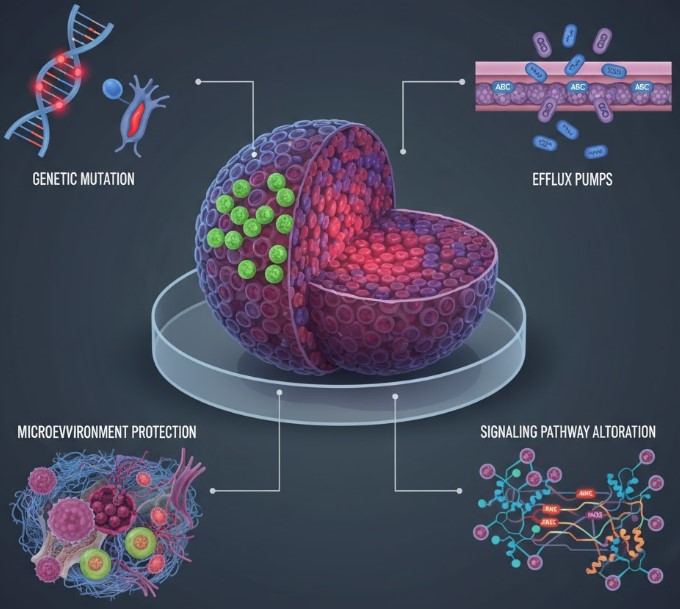
Drug Resistant Models | In Vitro Drug-Resistant Model | Specifically created models (e.g., pre-treated PDX) that accurately capture complex drug resistance mechanisms, offering high commercial relevance for developing next-generation therapeutics to address major clinical challenges. |
| In Vivo Drug-Resistant Model | |||
 Distributed under Open Access license CC BY 4.0, from Wiki, without modification. Distributed under Open Access license CC BY 4.0, from Wiki, without modification.
|
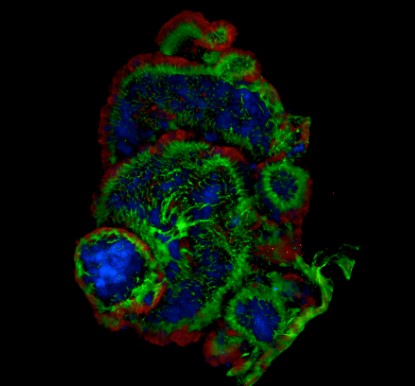
Tumor Organoid | Tumor Organoid Model | Self-organizing 3D tissues replicating tumor complexity and microenvironment, crucial for personalized, high-throughput drug screening, bridging in vitro and in vivo models. |
|
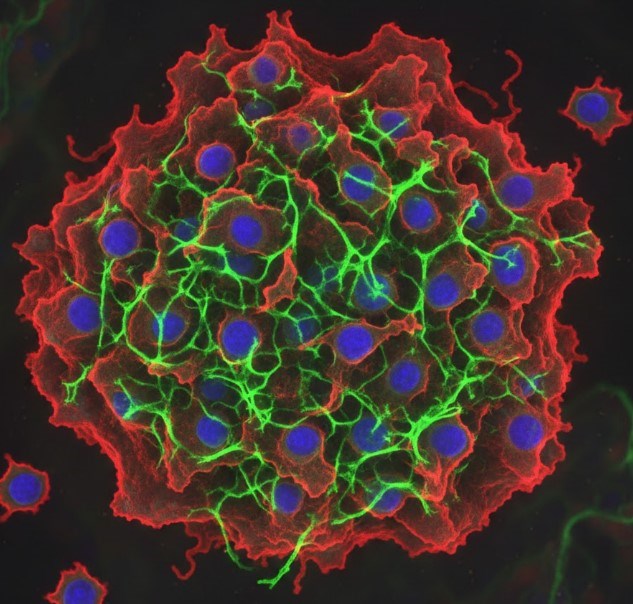
Immuno-Oncology Efficacy Evaluation | XDC based Efficacy Evaluation | Immuno-Oncology (IO) success relies on a comprehensive tumor modeling platform, utilizing syngeneic and humanized models for immunotherapies, PDX/CDX for biologics/small molecules, and supporting advanced therapies (e.g., CAR-T, ADCs), to ensure robust, high-quality preclinical efficacy evaluation across all major therapeutic modalities. |
| CAR-T & CAR-NK Cell based Efficacy Evaluation | |||
| Oncolytic Virus based Efficacy Evaluation | |||
| Cancer Vaccine based Efficacy Evaluation | |||
| CD19+ Humanized CDX Model based CAR-T Evaluation | |||
| CD19+ Humanized PDX Model based CAR-T Evaluation | |||
| Immune Checkpoint Inhibitor based Efficacy Evaluation | |||
|
Other Efficacy Evaluation | Prodrug based Efficacy Evaluation | Our tumor models are crucial for studying unique mechanisms, simulating disease etiology, and developing treatments for chronic progression or drug resistance challenges. |
| Protein Degrader based Efficacy Evaluation | |||
| RNA Therapy based Efficacy Evaluation | |||
| iPSC based Efficacy Evaluation | |||
Evaluation Platform
Leveraging decades of deep expertise in tumor model efficacy services, we stand at the forefront of the industry. We rely on a platform powered by multi-modal, cutting-edge technologies and state-of-the-art precision instruments for comprehensive efficacy assessment. Our commitment is to deliver highly rigorous, reliable, and exceptionally reproducible preclinical data, accelerating your path to drug development.
- Imaging: Uses MRI/PET to visualize tumor growth/metastasis in vivo. Confocal/Fluorescence Microscopy analyzes cellular morphology and protein expression in 3D models.
- Molecular Profiling: RNA Sequencing uncovers tumor heterogeneity and immune interactions. Spatial Metabolomics maps metabolic activity in tumor microenvironments. Phosphoproteomics identifies signaling pathways causing drug resistance.
- Functional Assays: Drug Sensitivity Testing predicts patient responses using PDXs/organoids.
- AI Integration: Machine Learning models analyze circulating free DNA (cfDNA) for accurate lesion classification (91% accuracy). Pathological AI systems classify 57 tumor subtypes across 9 organs, mimicking clinical diagnostics.
Our Advantages
- Accurate Pathological Recapitulation: Our animal models faithfully recapitulate the multi-dimensional complexity of human tumors. By simulating the complex Tumor Microenvironment (TME), including immune cell infiltration, angiogenesis, and stromal cell interactions, we can precisely track the mechanisms of tumor initiation and progression.
- Breadth and Depth of Models: Services range from basic CDX (for rapid, scalable screening) to the most clinically translational PDX and Humanized Mouse Models. We utilize Orthotopic Models to assess primary site invasion and metastasis, and Gene-Edited Models to explore explicit driver gene pathogenesis.
- Exceptional Predictive Accuracy: We adopt a patient-centric strategy, utilizing PDX and Organoid biobanks for drug screening. PDX models retain the tumor's genetic and histological characteristics, achieving a clinical prediction accuracy of 70%–80%.
- Rapid Turnaround: Organoid based functional drug testing can be completed within 2 to 3 weeks, significantly outperforming traditional screening methods and greatly accelerating early drug decision-making.
- High-Fidelity Data: The utilization of Large Animal Models provides high-fidelity translational data for preclinical research.
- Dynamic Mechanism Research Support: Tumor animal models allow for precise variable control through human intervention, supporting full-course dynamic tracking and multi-omic analysis of tumor initiation and progression mechanisms.
- Complex Therapy Validation Core: Animal models are the core platform for evaluating therapies dependent on the in vivo immune environment (e.g., CAR-T and cancer vaccines). We provide functional assessment of T cell proliferation, homing (trafficking), and anti-tumor activity.
- Integrated Preclinical Solutions: We offer systematic, one-stop model services, covering Efficacy, Pharmacokinetics (PK)/Pharmacodynamics (PD), and Toxicology evaluations, supporting models for respiratory, metabolic, neurological, and autoimmune diseases.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: How do I choose the right tumor model?
A: Model selection should be based on your research goals: use 3D organoids or GEMMs for mechanistic studies, PDXs or 3D bioprinting models for drug development, and syngeneic mouse models or patient derived immune organoids for immunotherapy.
-
Q: What is the success rate of PDX models?
A: Recent studies achieve 42% success using needle biopsies, with as few as 0.13×10⁶ cells. Key factors include viable cell count and microenvironmental preservation.
-
Q: Can 3D models replace animal studies?
A: Partially. 3D hydrogel models replicate PDX growth without mice, but in vivo models remain critical for metastasis and systemic toxicity testing.
-
Q: How are ethical concerns addressed in PDX/organoid research?
A: Institutions follow IRB-approved protocols for patient sample use. Emerging guidelines emphasize transparency in data sharing and model validation.
-
Q: What are the limitations of 2D cell lines?
A: They lack ECM interactions, hypoxia gradients, and cell polarity, leading to overestimation of drug efficacy by 40–60% compared to 3D models.
-
Q: How do I validate a new tumor model?
A: Validation requires a multi-faceted approach: use genomic/proteomic profiling to compare the model to patient tumors, perform functional assays to test drug response and invasion capacity, and establish clinical correlation by matching model predictions with patient outcomes.
Published Data
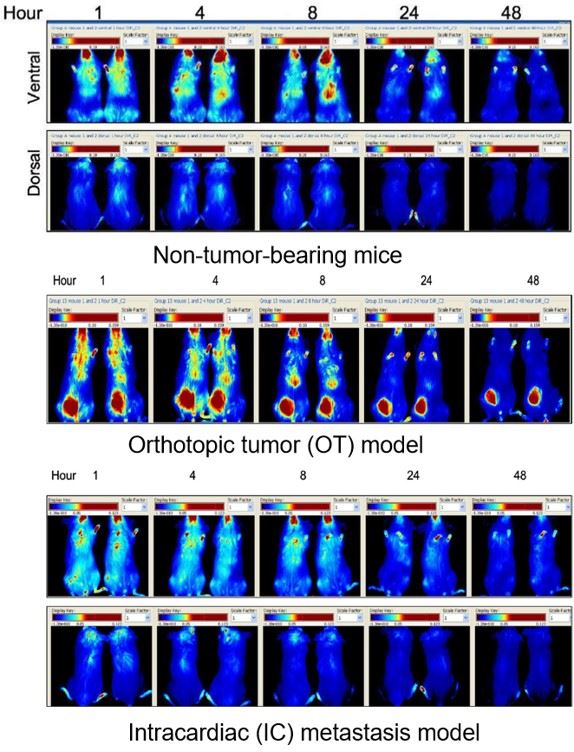 Fig.3 In vivo fluorescence imaging of the distribution of DiR labelled liposomes.3
Fig.3 In vivo fluorescence imaging of the distribution of DiR labelled liposomes.3
The in vivo distribution of liposomes is both organ-specific and time-dependent, and it is not significantly affected by the presence of a tumor. The accumulation of liposomes at the tumor site is notably limited, with the accumulation amount being independent of the model type. Furthermore, accumulation in metastatic lesions is poorer than in the primary orthotopic tumor.
References
- Guo, Haochuan et al. "The Pivotal Role of Preclinical Animal Models in Anti-Cancer Drug Discovery and Personalized Cancer Therapy Strategies." Pharmaceuticals (Basel, Switzerland) vol. 17,8 1048. https://doi.org/10.3390/ph17081048
- Salemme, Vincenzo et al. "The role of tumor microenvironment in drug resistance: emerging technologies to unravel breast cancer heterogeneity." Frontiers in Oncology Vol 13 1170264. https://doi.org/10.3389/fonc.2023.1170264
- Kalra, Jessica et al. "Accumulation of liposomes in metastatic tumor sites is not necessary for anti-cancer drug efficacy." Journal of translational medicine vol. 22,1 21. https://doi.org/10.1186/s12967-024-05428-9. Distributed under Open Access license CC BY 4.0, with modification.
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
