Pulmonary Nodular Disease Modeling & Pharmacodynamics Services
Introduction
Pulmonary nodular lymphoid hyperplasia is a rare reactive lymphoproliferative disorder that presents as asymptomatic lung masses. It's a debilitating inflammatory granulomatous disease of unknown etiology. Granulomas themselves are multinucleated structures driven by macrophages, believed to be triggered by insoluble environmental substances such as intracellular pathogens, organic antigens, or chemically inert materials, including beryllium or silica. Creative Biolabs utilizes meticulously established and validated pulmonary nodular disease models. These models provide robust research tools, such as for the screening of anti-fibrotic agents and macrophage modulators. We offer high-value solutions to accelerate drug research and development and facilitate essential mechanistic studies in pulmonary nodular disease.
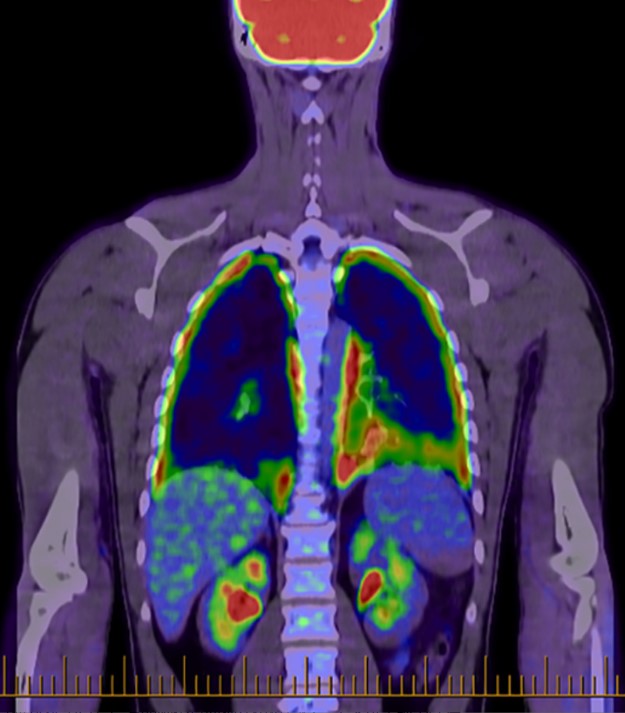 Fig.1 Whole-body positron emission tomography scan with diffuse pleural fluorodeoxyglucose uptake.1,3
Fig.1 Whole-body positron emission tomography scan with diffuse pleural fluorodeoxyglucose uptake.1,3
Available Pulmonary Nodular Disease Models
| Pulmonary Nodular Models | Clinical Relevance | Primary Research Applications | Animal Species |
| SiO2-Induced Pulmonary Nodule Model | Silicosis (occupational disease), Pulmonary Fibrosis, PM2.5-related lung injury. | Screening anti-fibrotic agents (e.g., Pirfenidone, Nintedanib), as well as lung protective agents and macrophage modulators. | Mouse, Rat |
| Carbon nanoparticle-induced sarcoidosis model | Sarcoidosis (granulomatous disease), chronic interstitial lung disease, and lung response to nanomaterials. | Focusing on immunosuppressants/modulators (e.g., Glucocorticoids, Methotrexate) and biopharmaceuticals targeting inflammatory mediators (e.g., the TNF-α inhibitor Infliximab). | Rat |
Evaluation Platform
Backed by extensive R&D expertise, we utilize state-of-the-art platforms to enhance service delivery and accelerate your pharmacodynamic studies.
- Imaging Detection: Using MRI, Micro-CT, and PET-CT, imaging allows for non-invasive assessment and dynamic monitoring of nodule morphology, size, number, and location within the lung tissue.
- Pathology Detection: Via Hematoxylin and eosin, Masson trichrome, Acid-fast, PAS staining, and IHC, pathological analysis is essential to analyze the precise cellular composition, collagen deposition, and specific characteristics of pulmonary nodules (e.g., silicotic or granulomatous).
- Molecular Biology Detection: Techniques like qPCR, WGS, WB, ELISA, and Metabolomics are employed to analyze the molecular mechanisms and pharmacodynamic indicators of nodule formation and progression at the gene, protein, and metabolite levels.
- Cell and Body Fluid Detection: Bronchoalveolar lavage fluid (BALF) analysis and hematological tests are used to evaluate the microenvironment of the nodule (e.g., local immune cells and cytokines) and the systemic immune/inflammatory state.
- Respiratory Function Detection: Measuring indicators such as lung volume parameters, ventilation function, airway resistance, and pulmonary compliance allows researchers to evaluate the physiological impact of nodules, specifically assessing airway obstruction and reduced lung elasticity.
- Other Auxiliary Detection: This includes specialized platforms for microbial detection and isotope labeling detection, providing supplementary data for investigating infectious etiologies or tracking molecular pathways within the nodules.
Applications
- Mechanistic Analysis: Investigate molecular pathways driving fibrosis and granuloma, and analyze particle cytotoxicity.
- New Drug Development: Primarily screens and validates anti-fibrotic and immunomodulatory agents. Also evaluates protective strategies for preventing nodule formation.
- Disease Simulation and Comparison: Provides platforms to simulate the chronic course of silicosis/sarcoidosis. Used to distinctly compare the pathology of fibrotic vs. granulomatous nodules and conduct environmental risk assessment.
Our advantages
- Professional Model Construction Capabilities: We can establish various pulmonary nodule animal models, covering different species to simulate diverse pathogenic mechanisms of pulmonary nodules.
- Multi-Dimensional Detection Platforms: We assess model success and drug efficacy across multiple levels, including imaging, pathology, molecular analysis, and functional evaluations.
- Customized Services and Technical Innovation Capabilities: We tailor solutions to meet clients' research objectives, encompassing model selection, a combination of detection indicators, and experimental cycle planning. We track cutting-edge technologies in the field to provide clients with more precise and efficient research tools.
- Experienced Team Support: Our interdisciplinary team, composed of experimental animal scientists, pathologists, and molecular biologists, offers professional support throughout the entire process, from experimental design to result interpretation.
- One-Stop Service: In addition to animal models for respiratory diseases, we also provide models for other system diseases, as well as pharmacokinetic and toxicology research.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: What are the advantages and disadvantages of the two models?
A: The Silica-Induced Model is characterized by typical lesions of layered collagen deposition and fibrous nodules, progressing to irreversible and progressive fibrosis. The Carbon Nanoparticle-Induced Model is characterized by the development of immune granulomas, and the inflammatory component of this model is reversible.
-
Q: Besides the models mentioned, can you provide other animal models for pulmonary nodules?
A: Yes, we can. These include gene-edited mouse models (e.g., KRAS/EGFR mutations) combined with chemical induction models, and infectious models, among others.
-
Q: What are the key indicators to confirm successful model establishment?
A: Success needs to be validated through multiple dimensions, including imaging (micro-CT), pathology (H&E staining), and molecular biomarkers (e.g., Ki-67, TTF-1).
-
Q: Can you customize model establishment based on specific molecular pathways?
A: Yes, we can. We can construct single-gene or multi-gene mutation models using CRISPR/Cas9 technology, or create compound models combining chemical induction with gene editing, specifically for targeting particular molecular pathways.
-
Q: Can the data and reports you provide support Investigational New Drug (IND) applications?
A: Yes, they can. Our animal and cell experiments, as well as the evaluation of detection indicators, adhere to SOPs and regulatory standards, meeting the application requirements of various countries.
Published Data
The case study on the role of miR-542-5p in SiO2-Induced pulmonary fibrosis utilized H&E staining to show that treatment reduced pulmonary fibrosis severity, evidenced by decreased nodule size and interstitial thickening. This finding confirms the model's promising application in pulmonary nodular disease research.
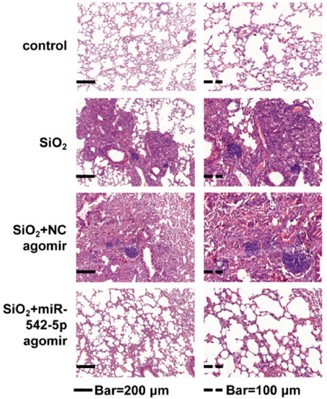 Fig.2 H&E staining of mouse lung tissue.2,3
Fig.2 H&E staining of mouse lung tissue.2,3
References
- Bird, Daniel, and Maninder Singh. "An unusual case of invasive pleuritis and miliary Mycoplasma pneumonia during check-point inhibitor therapy." Respirology Case Reports vol. 9,9 e0813. https://doi.org/10.1002/rcr2.813
- Yuan, Jiali et al. "miR-542-5p Attenuates Fibroblast Activation by Targeting Integrin α6 in Silica-Induced Pulmonary Fibrosis." International Journal of Molecular Sciences vol. 19,12 3717. https://doi.org/10.3390/ijms19123717
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
