Acetic Acid induced Mouse Vascular Permeability Modeling & Pharmacodynamics Service
Creative Biolabs offers comprehensive services in establishing and evaluating the Acetic Acid-Induced Mouse Vascular Permeability Model, ensuring high-quality, tailored solutions for your research needs.
Introduction
Acute inflammatory disorders are a group of conditions characterized by sudden-onset inflammation, often because of infection, injury, or immune system activation. Inflammation is a natural response of the body to harmful stimuli, but when it becomes excessive or uncontrolled, it can lead to tissue damage and contribute to disease progression. Common examples of acute inflammatory disorders include rheumatoid arthritis, sepsis, acute respiratory distress syndrome (ARDS), acute pancreatitis, and inflammatory bowel disease (IBD). In these conditions, the body’s immune system rapidly mobilizes white blood cells, cytokines, and other inflammatory mediators to the site of injury or infection, which results in the classic signs of inflammation: redness, heat, swelling, and pain. In the case of sepsis, this inflammatory response can become systemic, leading to widespread tissue damage and organ failure.
Acute inflammatory disorders are typically treated with anti-inflammatory drugs such as corticosteroids, NSAIDs, and immunosuppressive agents to control inflammation and prevent tissue damage. The pathophysiology of these conditions often involves complex interactions between immune cells, cytokines, and the vascular endothelium, making them ideal candidates for targeted therapeutic interventions. However, the rapid and dynamic nature of these conditions presents challenges in drug development, particularly in the need for effective therapies that can quickly modulate the inflammatory response. Preclinical models of acute inflammation are crucial for understanding the mechanisms behind these disorders and testing potential therapies to alleviate symptoms and improve patient outcomes.
Acetic Acid-Induced Mouse Vascular Permeability Model and Applications
The Acetic Acid-Induced Mouse Vascular Permeability Model is established by administering acetic acid intraperitoneally to rodents, which induces acute inflammation and increases vascular permeability. The inflammatory response results in the leakage of fluids and immune cells from the vasculature into surrounding tissues. This model is commonly used to evaluate the effect of drugs on vascular permeability and inflammation. Key advantages of this model include its simplicity, reproducibility, and ability to simulate localized inflammation, which is ideal for testing therapies targeting vascular diseases. However, the primary limitation of this model is that it focuses on acute inflammation, which may not fully replicate chronic conditions such as those seen in long-term vascular diseases or autoimmune disorders.
- Simulates: The Acetic Acid-Induced Mouse Vascular Permeability Model simulates acute inflammatory conditions such as edema, vascular permeability, and tissue injury. It is particularly useful for studying localized inflammatory responses and the mechanisms underlying increased vascular leakage.
- Evaluates Drugs: This model is employed to evaluate the efficacy of anti-inflammatory drugs (e.g., NSAIDs), vasoprotective agents, anti-edema treatments, cytokine inhibitors (e.g., TNF inhibitors), and therapies aimed at protecting endothelial cells or reducing oxidative stress and immune responses in inflammatory conditions.
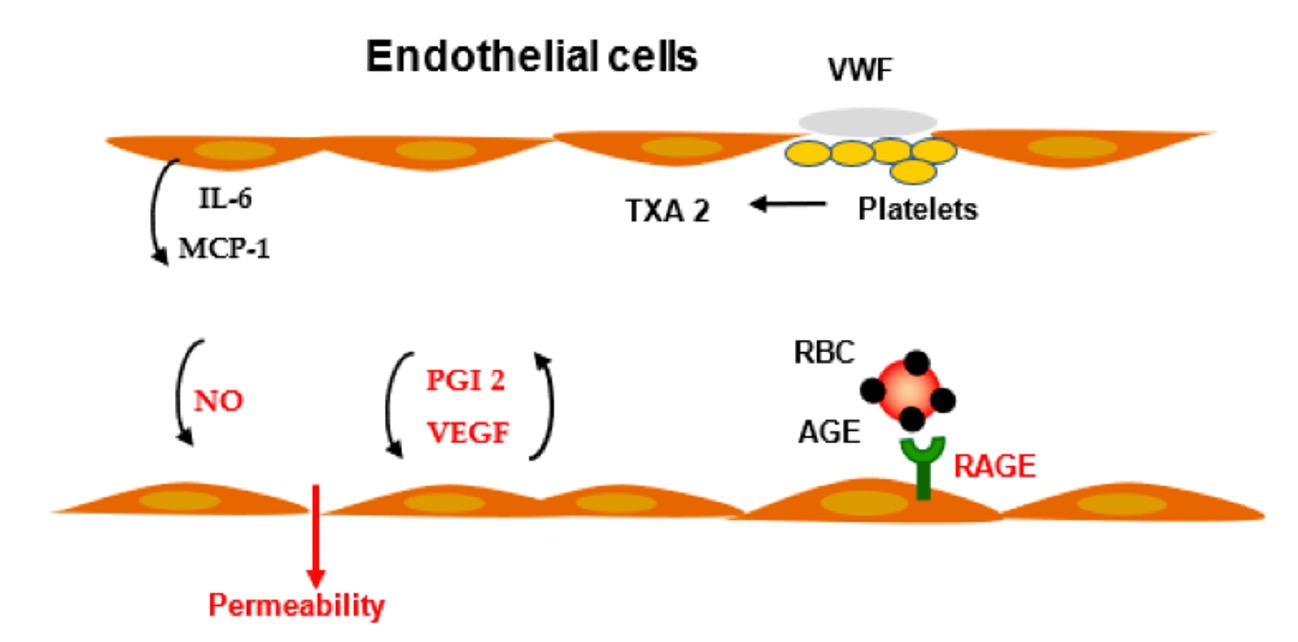 Fig.1 Modulation of vascular permeability and inflammation.1
Fig.1 Modulation of vascular permeability and inflammation.1
Evaluation Platform
- Animals: Mouse, Rat, Hamster, Rabbit, Cat, Dog, NHPs.
-
Measurements
We offer a variety of measurements for evaluating drug efficacy in the Acetic Acid-Induced Mouse Vascular Permeability Model, utilizing advanced technologies such as:- General observations: Changes in body weight, mortality rate, and visual assessment of inflammation.
- Vascular permeability assays: Measurement of fluid leakage into tissues or biomarkers in plasma.
- Histological analysis: Tissue sections stained to observe immune cell infiltration and tissue damage.
- Cytokine profiling (e.g., ELISA): Quantification of inflammatory mediators like TNF-α, IL-6, IL-1β via ELISA.
- Flow cytometry: Immune cell subpopulations involved in the inflammatory response.
- Gene/protein expression analysis: RT-qPCR and Western blotting to assess the expression of key inflammatory markers.
Our advantages
- Reproducibility: Reliable, consistent results for vascular permeability studies.
- Expert Support: Scientific team guidance at every stage, from design to analysis.
- Tailored Solutions: Customized protocols to meet your unique research objectives.
- Comprehensive Services: Full-service support, from model establishment to data interpretation.
- State-of-the-art Technologies: Cutting-edge tools for precise measurements and detailed analyses.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
1. What is the typical timeline for setting up this model?
Setup usually takes 1-2 days, with data collection occurring over a period of 24 to 72 hours, depending on study requirements.
-
2. Can this model be used for chronic vascular permeability studies?
This model is best suited for acute inflammation studies. For chronic conditions, modifications or alternative models are recommended.
-
3. What animals are used for this model?
The model is typically conducted in mice, though rats can also be used depending on research needs.
-
4. Can I evaluate drugs other than anti-inflammatory agents with this model?
Yes, this model can be used to assess a range of drugs targeting vascular integrity, immune modulation, and edema reduction.
-
5. How do the results from this model translate to human conditions?
While the model effectively simulates acute inflammation, additional validation in human-based models is necessary for direct application to clinical settings.
Published Data
In a recent study, the Acetic Acid-Induced Mouse Vascular Permeability Model was established to investigate the mechanisms underlying vascular permeability changes during inflammation. The experiment aimed to evaluate the role of vascular leakage in inflammatory responses and to test potential therapeutic agents targeting vascular integrity. C57BL/6J mice were injected with acetic acid to induce acute inflammatory responses and increase vascular permeability. Key parameters, such as body weight, inflammation markers, and edema formation, were closely monitored throughout the study. The vascular permeability was assessed by measuring the extravasation of Evans Blue dye, which indicates plasma leakage into the surrounding tissues. Histological analysis of tissue samples from the injection site was performed to assess the degree of vascular damage, including endothelial cell disruption and interstitial edema. Inflammatory markers, including TNF-α, IL-1β, and IL-6, were measured to evaluate the involvement of inflammatory cytokines in the regulation of vascular permeability. Additionally, the role of oxidative stress was assessed by measuring malondialdehyde (MDA) and superoxide dismutase (SOD) levels. The results revealed that acetic acid injection significantly increased vascular permeability, leading to tissue edema and the disruption of vascular integrity. Histological analysis confirmed endothelial damage, while elevated levels of inflammatory cytokines and oxidative stress markers indicated the involvement of these pathways in enhancing vascular leakage. This model effectively mimics the inflammatory processes that contribute to vascular permeability changes and provides a useful platform for evaluating therapeutic strategies aimed at reducing vascular leakage in inflammatory diseases.
Reference
- Wautier, Jean-Luc, and Marie-Paule Wautier. "Vascular Permeability in Diseases." International Journal of Molecular Sciences vol. 23,7 3645. 26 Mar. 2022. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/ijms23073645
For Research Use Only.
