Oncolytic Virus based Efficacy Modeling & Pharmacodynamics Service
Introduction
An oncolytic virus (OVs) is a type of virus that has the ability to preferentially infect, replicate in, and kill cancer cells without harming healthy, normal cells. The term "oncolytic" literally means "cancer-breaking." OVs can be naturally occurring or genetically engineered in a laboratory. OVs form a highly effective and specific anti-cancer mechanism through the dual actions of direct killing and immune activation. As a promising cancer treatment, the preclinical evaluation of OVs is highly dependent on appropriate animal models. Given that each animal model has its own unique characteristics, choosing a suitable one is crucial for accurately assessing the safety, biodistribution, immune response, and efficacy of OVs. Creative Biolabs, with years of experience in developing tumor animal models and evaluating drug efficacy, leverages its advanced models to support oncolytic virus development. Furthermore, we specialize in evaluating OV combination therapies, including molecular targeted therapy, chemotherapy, radiotherapy, and CAR-T cell therapy, to maximize treatment output and enhance tumor cell sensitivity to therapeutic factors.
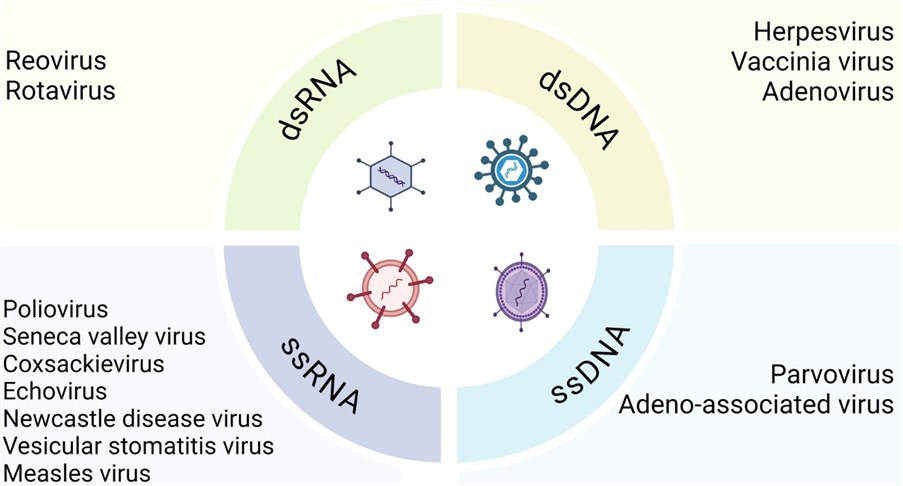 Fig.1 Classification of oncolytic viruses according to the type of viral nucleic acid.1,5
Fig.1 Classification of oncolytic viruses according to the type of viral nucleic acid.1,5
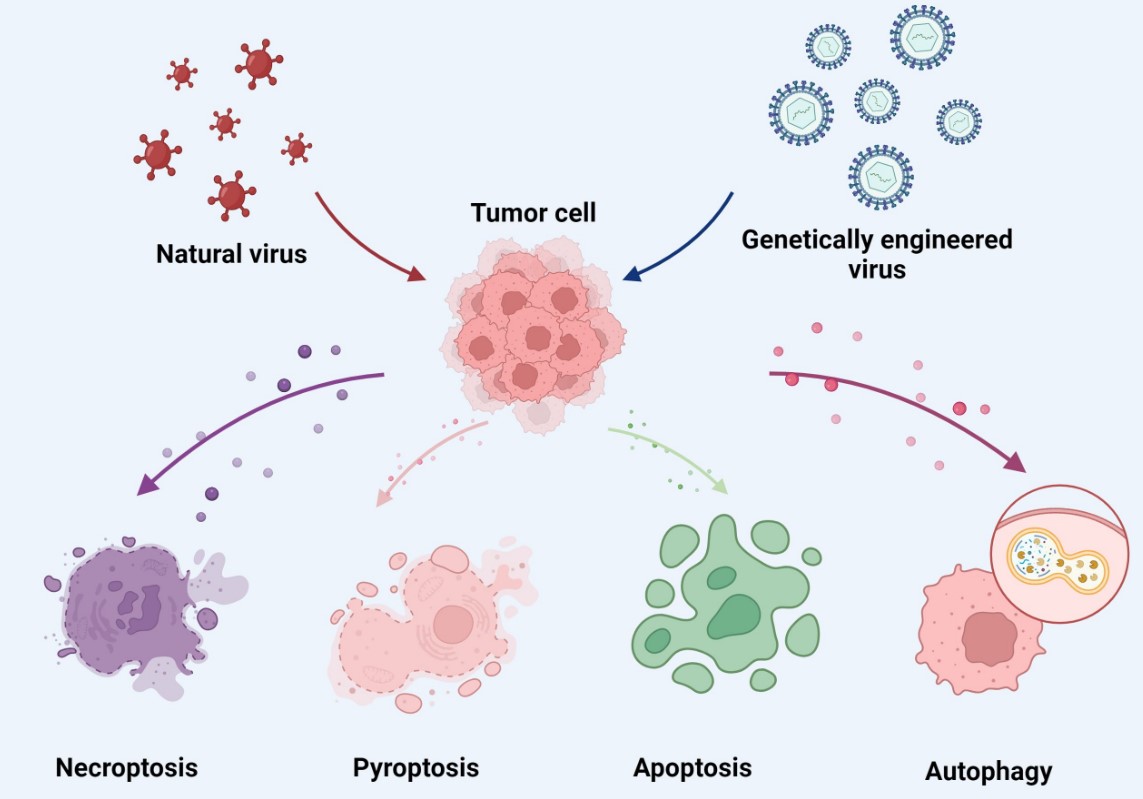 Fig.2 Oncolytic viruses induce necroptosis, pyroptosis, apoptosis, and autophagy in cancer cells.1,5
Fig.2 Oncolytic viruses induce necroptosis, pyroptosis, apoptosis, and autophagy in cancer cells.1,5
Available OVs based Efficacy Services
| Virus Family | Genome Structure | Key Efficacy Metrics | Preferred Animal Models | Typical Application Scenarios |
| Herpes Simplex Virus (HSV) | dsDNA | Tumor Regression Rate, Overall Survival (OS), Viral Replication Titer at the tumor site, and Immunogenicity (Anti-Virus Antibody titer). | Immunodeficient Xenograft (for direct lysis), Immunocompetent Syngeneic (for engineered variants), Humanized models. | Treatment of Melanoma (e.g., T-VEC), Head & Neck Cancer, Glioblastoma (GBM). |
| Adenovirus (AdV) | dsDNA | Tumor Growth Inhibition (TGI), Viral Dissemination (spread within the tumor), Gene Expression of payload (if engineered), Immune Cell Infiltration (e.g., CD8+). | Immunodeficient Xenograft (high efficiency/low immunogenicity models), Immunocompetent Syngeneic. | Intratumoral injection for superficial/resectable tumors, Liver Cancer, applications combined with chemotherapy. |
| Vaccinia Virus (VV) | dsDNA | Tumor Reduction in metastatic models, Systemic Distribution and replication (due to stability), Immune Activation in TME. | Immunocompetent Syngeneic (for systemically administered OVs), Xenograft (for high-burden models). | Systemic Administration (IV), treatment of Metastatic Cancer (e.g., liver, lung), often engineered to carry immune payloads. |
| Reovirus (RV) | dsRNA | Tumor Response Rate, Viral Replication tied to RAS pathway activation, Apoptosis/Necrosis markers, T-cell infiltration in "cold" tumors. | Immunocompetent Syngeneic, Xenograft (especially those with activated RAS/EGFR pathways). | Tumors characterized by activated RAS/EGFR pathways (e.g., Colorectal, Pancreatic, Melanoma), combined with immune checkpoint inhibitors. |
| Vesicular Stomatitis Virus (VSV) | ssRNA | Rapid Tumor Lysis Kinetics, Systemic Efficacy (due to strong viremia), Cytokine Burst (e.g., IFN type I response), Neuropathogenicity assessment (safety). | Immunocompetent Syngeneic (critical for immune assessment), Xenograft. | Aggressive/Refractory Cancers due to rapid replication, used for systemic delivery due to natural tropism. |
| Newcastle Disease Virus (NDV) | ssRNA | Tumor Regression, Safety Profile (low human toxicity), Immune Stimulation (IFN induction), Anti-metastatic potential. | Immunocompetent Syngeneic models. | Used in Immunotherapy combinations, treatment of Hepatocellular Carcinoma, and melanoma, often as an adjuvant. |
| Coxsackievirus | ssRNA | Specific Receptor Binding (e.g., CAR), Selective Tumor Killing, Viral Spread, Efficacy in Bladder Cancer models. | Xenograft models targeting specific receptors. | Cancers with high expression of specific receptors (e.g., Bladder Cancer, Melanoma). |
Evaluation Platform
Our service provides a comprehensive, end-to-end quantitative evaluation framework, centered around the three pillars of Viral Replication, Oncolytic Lysis, and Immune Activation. By seamlessly integrating in vitro and in vivo data, we offer a full-process quantitative assessment, from initial virus design optimization through to clinical translation, specifically designed to overcome critical pain points such as solid tumor penetration and Tumor Microenvironment (TME) suppression.
I. Multi-Tiered Predictive Modeling Systems:
We utilize a scientifically tiered approach, selecting models specifically for their ability to predict biological outcomes across different phases of development:
A. Foundation and Screening Models
- Tumor Organoid Models: Offer high clinical relevance for in vitro studies by simulating complex 3D structures. They enable the evaluation of direct oncolytic potency, infection efficiency, personalized screening, and assessing synergistic effects with other therapies.
- Xenograft Models (CDX/PDX): Critical for assessing intrinsic oncolytic capability without host immune interference. CDX models facilitate the rapid screening of highly responsive indications, while PDX models simulate clinical tumor heterogeneity, guiding individualized therapeutic prediction and biomarker development.
B. Immune and Systemic Efficacy Models
- Syngeneic Models: The gold standard for studying the virus's dual mechanism involving immune activation. They utilize immunocompetent mice to evaluate the crucial interaction between the OV and the intact host immune system, measuring the ability to induce a systemic anti-tumor adaptive immune response necessary for long-term efficacy and assessing systemic toxicity and anti-viral immunogenicity.
- Immunocompetent Models: Specifically used to evaluate virus induced anti-tumor immune memory, such as the protection rate against a secondary tumor challenge.
C. High-Predictive Translational Models
- Humanized Models: By reconstituting a human immune system in mice, these models bridge the gap to human clinical trials, providing the most predictive data for efficacy, safety, and human immunogenicity by enabling the study of the human immune response and pharmacodynamics (PD) of human-specific OVs.
- Genetically Engineered Mouse Models (GEMMs): Used for mechanistic validation by creating Humanized Receptor Models for virus tropism studies, or by Modeling Tumor Genetic Defects to test OVs engineered for tumor-specific pathway defects. They are also essential for Off-Target Toxicity Assessment by expressing viral antigens on normal, healthy tissues.
II. Advanced Detection Methods & Key Metrics:
Our platform employs a comprehensive suite of Advanced Detection Methods to evaluate key efficacy metrics with precision and depth:
- Advanced Imaging (BLI, PET, MRI, CT): We employ these non-invasive tools for longitudinal tracking of key metrics, including Tumor Volume, Viral Replication and Spread, and Survival Analysis. This ensures continuous, real-time monitoring throughout the study.
- Cellular & Histological Analysis (Flow Cytometry, IHC/IF): This suite of methods delivers detailed insights into the cellular landscape. By providing highly specific spatial and phenotypic quantification, we analyze Immunological Markers (cell phenotypes, infiltration) and perform comprehensive TME and Abscopal Effect Evaluation.
- Molecular Analysis (qPCR, dPCR, etc): These methods provide crucial mechanistic depth. We quantitatively measure viral copy numbers and serum cytokine levels (via multiplex analysis), while simultaneously conducting in-depth mechanistic investigations into gene expression and pathway alterations at the cellular level.
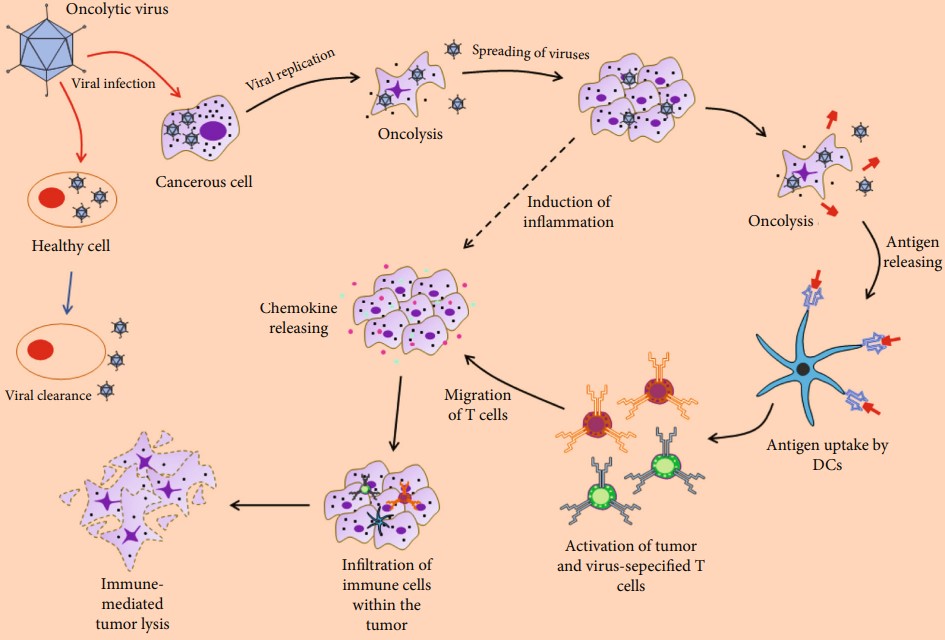 Fig.3 The modes of action of oncolytic viruses in killing tumor cells and their different effects on various cells within the tumor microenvironment.2,5
Fig.3 The modes of action of oncolytic viruses in killing tumor cells and their different effects on various cells within the tumor microenvironment.2,5
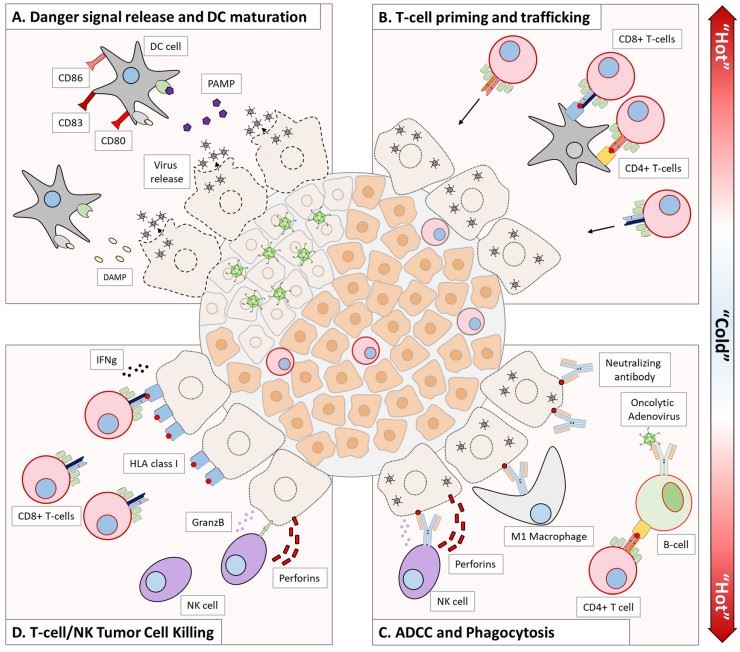 Fig.4 Activating the immune system for cancer rejection with oncolytic virus therapy.3,5
Fig.4 Activating the immune system for cancer rejection with oncolytic virus therapy.3,5
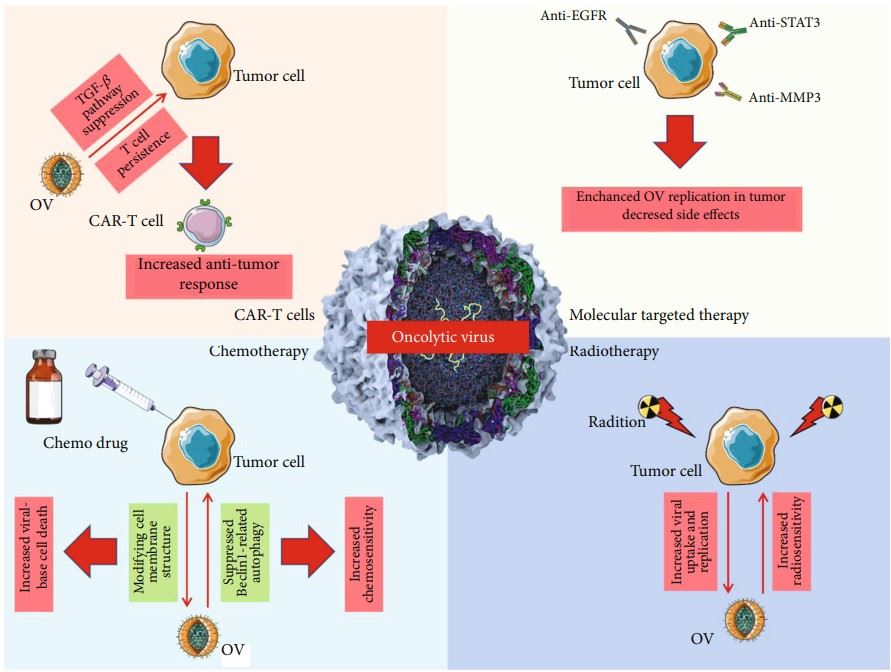 Fig.5 The combination of OV therapy with other therapeutic approaches.2,5
Fig.5 The combination of OV therapy with other therapeutic approaches.2,5
Applications
- Viral Design and Optimization: We utilize a model-driven approach to optimize OV design across Targeting, Replication, Combination, and Immune Enhancement. We predict the best Targeting Strategy (promoters/ligands) to reduce off-target risk, and quantify the Replication Capability of viral mutants to balance lytic efficiency with safety. We design Combination Strategies by simulating synergy with other therapies (ICIs/CAR-T), predicting optimal dosing sequences and ratios. Finally, we verify Immune Enhancements (e.g., IL-12/anti-PD-L1 expression) to predict the synergistic strength of the "oncolysis + immune activation" effect.
- Preclinical Efficacy Assessment and Screening: We establish In Vitro-In Vivo Correlation (IVIVC) to link in vitro lysis with in vivo regression, accelerating the R&D cycle and reducing animal experiments by over 40%. Our platform enables rapid, broad screening of responsive indications and facilitates individualized prediction and biomarker development. Furthermore, we assess anti-tumor immune memory, validating the "oncolysis + long-term immune protection" dual effect.
- Safety Assessment and Risk Prediction: We rigorously define the safety profile by constructing Normal Tissue Viral Replication-Toxicity models to predict potential organ damage (liver, kidney, lung) and establish the safe dose window. We predict Immune-Related Toxicity by simulating the risk of cytokine storms (IL-6, IFN-γ, TNF-α), guiding the design of pre-treatment regimens. Finally, we assess Viral Spread Risk by monitoring in vivo dissemination to evaluate the probability of transmission, providing essential data for clinical containment protocols.
- Combination Therapy Design and Validation: We quantify synergy of OVs with Immunotherapies (PD-1/PD-L1, CTLA-4) to predict efficacy gain and determine optimal sequence/dosage. We optimize synergy with Chemo/Radiotherapy by simulating the "sensitization effect" and predicting combined toxicity risks. Finally, we model Cell Therapy combinations (CAR-T/CAR-NK), evaluating the synergistic effect (TME remodeling) and optimizing staggered administration timing to maximize benefit.
Our Advantages
- Diverse Animal Model Repository: We offer immunodeficient mouse models for assessing direct oncolytic activity, immunocompetent mouse models for evaluating the immune response, and humanized mouse models for simulating the interaction with the human immune system.
- Specialized Dosing Techniques: We have expertise in various administration routes, such as intratumoral, intravenous, and intraperitoneal injection, and we can optimize these methods based on study requirements.
- Experienced Research Team: Our team has a deep understanding of virology, immunology, and the unique mechanisms of OVs to design scientifically sound and relevant experimental protocols.
- Quality Control & SOPs: We implement strict quality control (QC) to ensure data reliability and reproducibility.
- One-stop service: We offer a full suite of services for oncolytic viruses, including pharmacology, efficacy, toxicology, and pharmacokinetics.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: How do you choose the right animal model for OV evaluation?
A: The optimal choice of animal model is dictated by specific research objectives. To evaluate a virus's direct oncolytic ability, researchers should use immunodeficient mouse models bearing human xenograft tumors. Conversely, assessing the immune-activating effect and systemic efficacy requires immunocompetent mouse models with syngeneic tumors. For results with the highest clinical relevance, the recommended models include humanized mice or studies in comparative oncology using companion animals, such as dogs, with naturally occurring tumors.
-
Q: Why doesn't an increase in tumor volume necessarily mean treatment failure?
A: Unlike conventional therapies, Oncolytic Viruses (OVs) function through both direct cell killing and subsequent immune system activation. The essential phenomenon of "pseudoprogression" occurs because the necessary influx and activation of immune cells (like T cells) attracted by the virus induced lysis temporarily increase the tumor volume appearance on imaging. Therefore, judging OV efficacy requires moving beyond tumor size alone. Key indicators used to confirm efficacy include improved clinical symptoms, a decrease in tumor metabolic activity seen on PET/CT imaging, and tumor biopsies confirming significant immune cell infiltration and cancer cell death.
-
Q: What are the routes of administration for oncolytic viruses in animal models?
A: Common administration routes include intratumoral injection, intravenous injection, intraperitoneal injection, and oral administration. The route should be selected based on the virus properties and the location of the target tumor.
-
Q: How is the safety and toxicity of oncolytic viruses assessed?
A: Safety and toxicity are assessed by monitoring changes in animal body weight, behavioral changes, pathological examination of major organs, and biochemical analysis.
-
Q: How to address immunogenicity issues of viruses within animal bodies?
A: For Oncolytic Viruses (OVs), immunogenicity is a critical part of the mechanism of action, not a factor to be suppressed. We address this by utilizing the right models for proper study: Syngeneic Models (using mouse-specific viruses in immunocompetent mice) allow us to study the complete virus-immune system interaction, while Humanized Models provide the most clinically relevant data for human-specific viruses, enabling the simultaneous assessment of both efficacy and potential immunogenicity within a human immune context.
-
Q: How long does it take to see significant efficacy results?
A: Preliminary efficacy changes are usually observed within a few weeks, but the specific timeframe may vary depending on the type of virus and the specific research conditions.
Published Data
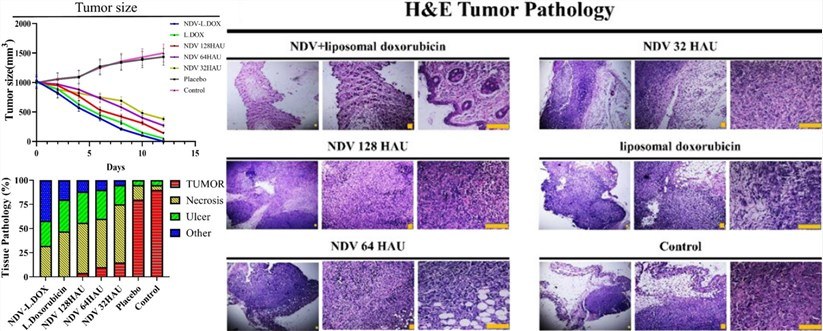 Fig.6 Comparison of tumor size changes and histopathology results in each mouse group.4
Fig.6 Comparison of tumor size changes and histopathology results in each mouse group.4
The oncolytic Newcastle Disease Virus (NDV) LaSota strain strongly killed 4T1 breast cancer cells in vitro by inducing apoptosis and ROS. This effect was synergistically enhanced with liposomal Doxorubicin. This synergy translated in vivo, leading to strong tumor inhibition against 4T1 xenografts, with the combination achieving tumor-free status in mice within 10 days, significantly outperforming monotherapy.
References
- Wu, Xiaoke, and Shaokuan Fang. "Comparison of differences in immune cells and immune microenvironment among different kinds of oncolytic virus treatments." Frontiers in immunology vol. 15 1494887. https://doi.org/10.3389/fimmu.2024.1494887.
- Hemminki, Otto et al. "Oncolytic viruses for cancer immunotherapy." Journal of hematology & oncology vol. 13,1 84. https://doi.org/10.1186/s13045-020-00922-1.
- Garmaroudi, Ghazal Asadi et al. "Therapeutic Efficacy of Oncolytic Viruses in Fighting Cancer: Recent Advances and Perspective." Oxidative medicine and cellular longevity vol. 2022 3142306. https://doi.org/10.1155/2022/3142306.
- Faranoush, Pooya et al. "Evaluation of the in vitro and in vivo effect of liposomal doxorubicin along with oncolytic Newcastle disease virus on 4T1 cell line: Animal preclinical research." Veterinary medicine and science vol. 9,3 (2023): 1426-1437. Distributed under Open Access license CC BY 4.0, with modification. https://doi.org/10.1002/vms3.1109.
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
