Syngeneic Modeling & Pharmacodynamics Services
Introduction
Syngeneic models have an irreplaceable role and research significance in cancer research. Due to their core advantage of a complete immune system and a homologous tumor microenvironment, syngeneic models have become a crucial bridge connecting basic research and clinical translation. Creative Biolabs offers a wide range of well-validated syngeneic models, which hold irreplaceable value, especially in the development of immunotherapy, the analysis of tumor-immune interaction mechanisms, and the evaluation of combination therapies.
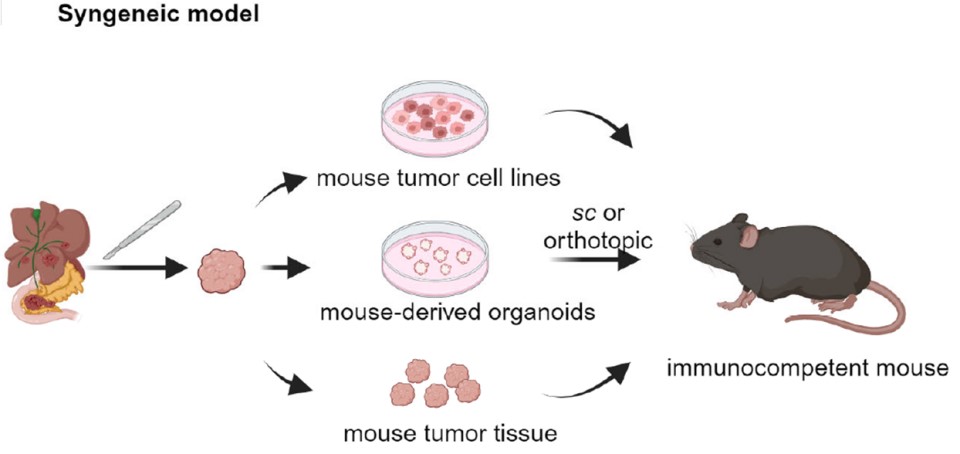 Fig. 1 Syngeneic mouse models are generated by injecting mouse tumor materials subcutaneously or orthotopically.1,5
Fig. 1 Syngeneic mouse models are generated by injecting mouse tumor materials subcutaneously or orthotopically.1,5
Available Syngeneic Models
The syngeneic models provided by Creative Biolabs ensure the genetic consistency of the tumor, immune system, and microenvironment by implanting tumors into genetically matched, immunocompetent hosts. This enables researchers to deeply investigate tumor-immune interactions and accurately assess the efficacy of various therapeutics (such as checkpoint inhibitors) within a fully functional immune background.
| Syngeneic Models | Modeling Method | Relevant Evaluation | Animal Species |
| Syngeneic Cell Line derived Tumor Models | Injecting immortalized mouse tumor cell lines (e.g., B16F10, CT26) subcutaneously, orthotopically, or intravenously into immunocompetent mice of the same genetic background. | Foundational and High-throughput Screening for Immunotherapies (e.g., Checkpoint Inhibitors, vaccines). Studying the complete TME and host immune response. | Immunocompetent Mouse (e.g., C57BL/6, Balb/c, FVB) |
| Syngeneic Mice derived Tumor Models | Harvesting fresh tumor tissue or cells from GEMM models or spontaneous tumors and transplanting them back into immunocompetent mice of the same strain. | High fidelity simulation of human tumor molecular pathology and histology. Used to study specific gene-driven tumor immune microenvironments. | Immunocompetent Mouse (Matching the background strain of the GEMM or spontaneous tumor) |
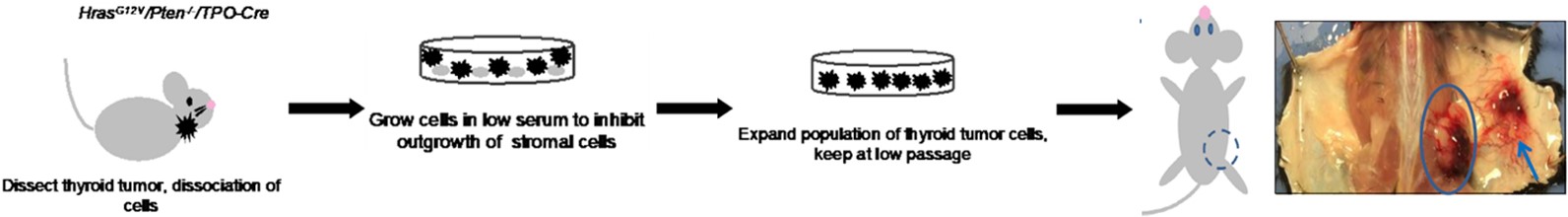 Fig. 2 Gross image of subcutaneous tumor and neovascularization on the right hind flank of a wild-type recipient.2
Fig. 2 Gross image of subcutaneous tumor and neovascularization on the right hind flank of a wild-type recipient.2
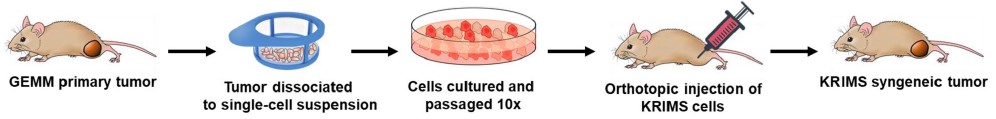 Fig. 3 The construction of the K-Ras induced murine sarcoma (KRIMS) tumor model.3,5
Fig. 3 The construction of the K-Ras induced murine sarcoma (KRIMS) tumor model.3,5
Evaluation Platform
To translate the full potential of our models into reliable clinical data, we have built an industry-leading Pharmacodynamics (PD) evaluation platform. This platform integrates cutting-edge, multi-modal detection technologies, achieving comprehensive coverage from macro-level in vivo imaging down to single-cell molecular fidelity. Leveraging these sophisticated analytical tools, we ensure the depth and rigor of drug mechanism-of-action and efficacy assessments, which are key to accelerating your success in oncology drug development. Our advanced assessment metrics include, but are not limited to, the following core areas:
- Tumor Growth and Progression Assessment: Longitudinal in vivo imaging, tumor volume measurement, TGI calculation, and histological validation of tumor morphology.
- Tumor Microenvironment (TME) Assessment: Immune cell profiling, spatial distribution analysis, cytokine/checkpoint quantification, and immune-related gene expression analysis.
- Metastatic Potential Assessment: Nodule counting, histological detection of micrometastases, and molecular analysis of disseminated tumor cells (DTCs) in distant organs.
Applications
- Drug Screening: Used for high-throughput initial screening of immunotherapies, rapidly establishing foundational efficacy and PK/PD relationships.
- Disease Simulation: Faithfully simulate the tumor's Immune Microenvironment (TME) and molecular pathology, crucial for studying immune evasion and progression.
- Efficacy Assessment: The gold standard for all Immunotherapies, focusing on Mechanism of Action (MoA) validation (e.g., T cell activation) and optimal combination therapy evaluation.
- New Target Identification: The complete immune environment allows for the discovery and validation of novel immune checkpoint targets and tumor antigens using advanced single-cell analysis.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
Our Advantages
- Extensive Model Portfolio: We have curated a high-quality library of syngeneic models, covering a diverse range of common and rare cancer types. This extensive portfolio serves as a valuable resource to help clients select the optimal model for their research.
- Advanced Genetic Engineering: Our team can construct custom, gene-edited syngeneic models to support combination studies involving targeted therapies and immunotherapies.
- Standardized and Reliable Operations: We follow rigorous SOPs that guarantee a tumor take rate of over 90%. Our advanced evaluation platforms provide comprehensive assessments of therapeutic response and efficacy.
- Proven Track Record: We have an extensive portfolio of successful case studies in preclinical drug evaluation, demonstrating our robust capabilities and expertise.
- Custom Model Development: We offer tailored model creation services to meet the unique requirements of specific therapeutic agents or research questions.
- Integrated Preclinical Services: We provide a comprehensive suite of services for your oncology program, from model selection and study design to in-depth analysis, including pharmacokinetics and toxicology assessments.
FAQs
-
Q: What are the key characteristics of syngeneic models?
A: They retain a complete immune system, so they are a core tool for evaluating the efficacy of immunomodulatory drugs.
-
Q: Do you support humanized target modification?
A: Yes. We can use gene editing to create humanized immune checkpoint models, which enhances the clinical relevance of antibody drug evaluations.
-
Q: What are the key efficacy endpoints for drug evaluation?
A: The fundamental evaluation of anti-tumor drug efficacy relies on two core metrics: Tumor Growth Inhibition (TGI) and Survival Analysis. TGI is quantitatively calculated by measuring the difference in tumor volume between the treatment group and the control group, serving as the primary short-term indicator of drug effectiveness. This is critically complemented by Survival Analysis, which measures the drug's long-term impact on the disease progression, often reported through the median survival time and visualized via Kaplan-Meier survival curves.
-
Q: How do you analyze the immune microenvironment?
A: We use multi-parameter flow cytometry to quantify tumor-infiltrating lymphocytes (TILs), including the proportion and activation markers of T cells, NK cells, and macrophages.
-
Q: Do you offer multi-omics integrated analysis?
A: Yes, we support the joint analysis of transcriptomics (RNA-seq), metabolomics, and TILs data to gain a deeper understanding of drug mechanisms.
-
Q: What is the typical experimental timeline?
A: A standard preclinical study typically spans 6 to 12 weeks. This process begins with Model Construction (1–2 weeks), involving cell line resuscitation, expansion, and inoculation. This is immediately followed by the intensive Efficacy Study phase (3–6 weeks) for drug treatment and tumor monitoring. The project concludes with Data Analysis (2–4 weeks), which encompasses flow cytometry, pathology, and comprehensive statistical analysis to deliver the final insights.
Published Data
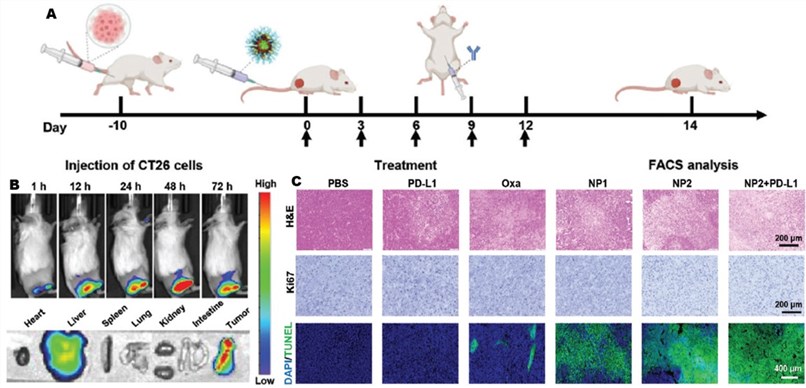 Fig. 4 Results of fluorescence imaging, H&E staining, TUNEL staining, and Ki67 staining.4
Fig. 4 Results of fluorescence imaging, H&E staining, TUNEL staining, and Ki67 staining.4
In the CT26 syngeneic mouse model, the NP2@Cy7 nanoparticle demonstrates robust tumor-targeting properties. Combination therapy utilizing NP2 and an anti-PD-L1 antibody effectively promotes tumor cell killing and significantly inhibits proliferation. This data confirms the model's significant application potential in the research and development of novel cancer therapeutics.
References
- Yan, Bin et al. "From Genes to Environment: Elucidating Pancreatic Carcinogenesis Through Genetically Engineered and Risk Factor-Integrated Mouse Models." Cancers vol. 17,10 1676. https://doi.org/10.3390/cancers17101676.
- Caperton, Caitlin O et al. "Development of Novel Follicular Thyroid Cancer Models Which Progress to Poorly Differentiated and Anaplastic Thyroid Cancer." Cancers vol. 13,5 1094. https://doi.org/10.3390/cancers13051094. Distributed under Open Access license CC BY 4.0, with modification.
- Gutierrez, Wade R et al. "Divergent immune landscapes of primary and syngeneic Kras-driven mouse tumor models." Scientific Reports vol. 11,1 1098. https://doi.org/10.1038/s41598-020-80216-1
- Gao, Xiangjie et al. "Encapsulation of Platinum Prodrugs into PC7A Polymeric Nanoparticles Combined with Immune Checkpoint Inhibitors for Therapeutically Enhanced Multimodal Chemotherapy and Immunotherapy by Activation of the STING Pathway." Advanced science (Weinheim, Baden-Wurttemberg, Germany) vol. 10,4 (2023): e2205241. https://doi.org/10.1002/advs.202205241. Distributed under Open Access license CC BY 4.0, with modification.
- Distributed under Open Access license CC BY 4.0, without modification
For Research Use Only.
