Carcinogen induced Mouse Tumor Modeling & Pharmacodynamics Service
Introduction
Creative Biolabs' well-established Carcinogen induced (CI) Mouse Tumor Model platform employs diverse chemical agents to reliably and reproducibly induce in vivo neoplasia. This methodology facilitates the investigation of tumorigenesis mechanisms, the subsequent screening of novel therapeutics, and the rigorous assessment of cancer prevention strategies.
Available Carcinogen induced Mouse Tumor Models
Creative Biolabs leverages its decades of specialization in oncology model development to offer state-of-the-art CI Mouse Tumor Models. These sophisticated platforms stand apart because the tumors are generated in situ within immunocompetent hosts, faithfully mimicking the natural process of human carcinogenesis and ensuring the presence of an intact tumor microenvironment and native neoantigens.
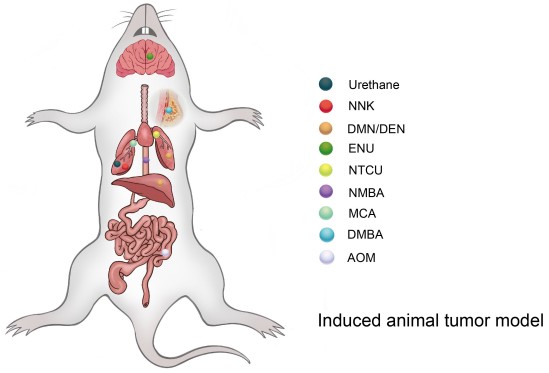 Fig1. Different types of compounds can induce tumors in different organs of mice (rats).1
Fig1. Different types of compounds can induce tumors in different organs of mice (rats).1
| Model Type | Carcinogen | Modeling Method | Relevant Evaluation | Animal Species |
| Colon Cancer | AOM + DSS | Mimics the inflammation-driven tumorigenesis pathway, similar to colitis-associated cancer in humans. | Immuno-oncology efficacy (e.g., Checkpoint Inhibitors), Inflammation-cancer link studies, and Toxicity of new drugs. | C57BL/6, BALB/c Mouse |
| Liver Cancer (HCC) | DEN | Efficiently induces HCC linked to human hepatitis, suitable for studying early lesions and cirrhosis. | Fibrosis and cirrhosis mechanism studies, Evaluation of small molecule drugs targeting early HCC stages. | C3H/HeN, C57BL/6 Mouse |
| Liver Adenoma | DEN + CCl4 | Combines chemical initiation (DEN) with inflammation/fibrosis promotion (CCl4), accelerating tumor development. | Evaluation of anti-fibrotic and anti-tumor agents, Mechanistic studies of chemically accelerated hepatocarcinogenesis. | B6C3F1/J Mouse |
| Lung Cancer | NNK, DEN, Urethan | Induces orthotopic lung tumors (adenomas and adenocarcinomas), simulating smoking-related lung pathology. | Pulmonary pathology progression, Efficacy of immune based therapies and vaccines against lung tumors. | A/J, C57BL/6, BALB/c, Swiss Mouse |
| Bladder Cancer | BBN | Highly specific induction of bladder transitional cell carcinoma (TCC), easy to assess drug activity in the urinary system. | Efficacy of intravesical therapies, Studies on urinary system drug disposition, and TCC mechanistic research. | C57BL/6, BALB/c Mouse |
| Skin Cancer | DMBA + TPA | Two-stage carcinogenesis model (initiation and promotion), suitable for studying early mechanisms of cell proliferation and inflammation. | Research on early carcinogenesis, Inflammation signaling pathways, and topical/dermatological oncology treatments. | SKH-1, CD-1 Mouse |
| Breast Cancer | DMBA | Induces mammary tumors, often simulating hormone-dependent or triple-negative human breast cancer subtypes. | Evaluation of hormone therapy, Chemotherapy efficacy, and Studies on mammary gland tumorigenesis mechanisms. | SD rat |
| Esophageal Tumors | NMBA | Specifically induces squamous cell carcinomas in the esophagus, mirroring human esophageal cancer. | Efficacy assessment for therapies targeting esophageal SCC, Mechanistic study of nitrosamine induced cancer. | A/J or FVB/N Mouse |
| Lung Squamous Cell Carcinoma | NTCU | Specifically induces squamous cell carcinoma (SCC) in the lung, a common aggressive human subtype. | SCC-specific therapy development, Investigation of carcinogen induced lung SCC development. | SWR/J, NIH Swiss, A/J, FVB/J, BALB/cJ Mouse |
| Nervous System Tumor | ENU | Highly potent mutagen inducing tumors in the central and peripheral nervous system (e.g., gliomas). | Development of neuro-oncology drugs, Mechanistic studies of nervous system tumor development. | SD rat (offspring) |
| Respiratory Tract Squamous Cell Carcinoma | MCA | Induces squamous cell carcinoma (SCC) in the respiratory tract. | Studies on respiratory tract carcinogenesis, Evaluation of preventative agents. | BC3F1, DBA/2 Mouse |
| Oral squamous cell carcinoma | 4NQO | Induces oral mucosal lesions and SCC, mirroring the human pathology sequence (dysplasia to SCC). | Efficacy of local/systemic therapies for oral cancer, Mechanistic research on oral carcinogenesis. | Mouse |
Abbreviations: AOM: Azoxymethane, DSS: Dextran Sulfate Sodium, DEN: Diethylnitrosamine, NNK: Urethane or 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone, BBN: N-Butyl-N-(4-hydroxybutyl)nitrosamine, DMBA: 7,12-Dimethylbenz[a]anthracene, TPA: 12-O-tetradecanoylphorbol-13-acetate, DMBA: 7,12-Dimethylbenz[a]anthracene, NMBA: N-nitrosomethylbenzylamine, CCl4: Carbon tetrachloride, NTCU: N-nitroso-trichloroethylurea, ENU: Ethylnitrosourea, MCA: 3-Methylcholanthrene, 4NQO: 4-Nitroquinoline 1-oxide.
Evaluation Platform
At Creative Biolabs, the predictive power of our CI Mouse Tumor Models is ensured by our scientifically rigorous Evaluation Platform. This platform integrates advanced, multi-modal technologies and standardized procedures to deliver accurate, reproducible, and translationally relevant results for your therapeutic candidates.
- Multimodal Imaging: Imaging technologies provide non-invasive in vivo data. Magnetic Resonance Imaging (MRI) analyzes tumor anatomical structure and visceral metastases; Photoacoustic Imaging (PAI) detects tumor oxygen saturation and hemoglobin concentration; In Vivo Fluorescence Imaging tracks tumor growth and metastasis dynamics; Ultrasound (US) monitors changes in tumor volume; and Infrared Thermal Imaging assesses body surface temperature.
- Functional Assessment: Functional assessments delve into physiological activity. Metabolic Monitoring is achieved by using Micro-CT/PET to detect tumor glucose metabolic activity, and Angiogenesis Analysis quantifies tumor vascular density using techniques like LSCI.
- Histopathological Analysis: Pathological analysis verifies morphology and biomarkers. H&E Staining is used to identify tumor grading and malignancy; Immunohistochemistry (IHC) is used to detect specific tumor markers, such as protein expression levels.
- Molecular Mechanism Analysis: Molecular analysis reveals the regulatory basis: Genomic Sequencing (NGS) is used to identify driver mutations, and Methylation Analysis is used for an in-depth study of epigenetic regulation mechanisms.
- Immune Microenvironment Analysis: Immune analysis is core to immunotherapy assessment. Flow Cytometry is used to quantify key immune cell subsets like Tumor-Infiltrating Lymphocytes (TILs) and Myeloid derived Suppressor Cells (MDSCs), and Spatial Transcriptomics is used to precisely locate tumor-immune cell interaction regions.
Applications
- Study of Tumorigenesis Mechanisms: Faithfully models the human tumor initiation process, tracking and analyzing the temporal sequence and mechanism of key molecular events (e.g., signaling pathways, gene alterations) following carcinogen exposure, and identifying early drivers and therapeutic targets.
- Carcinogenic Risk Assessment: Utilized for the carcinogenicity testing and risk assessment of new chemical substances in food and the environment. It establishes dose-response relationships, providing scientific data for setting regulatory safety standards.
- Screening of Cancer Prevention Strategies: Serves as an effective platform for screening novel chemopreventive agents, nutritional interventions, or lifestyle modifications, assessing their inhibitory or blocking effects on the carcinogenic process at the very early stages of tumor initiation.
- Pre-clinical Evaluation of Anti-cancer Drugs: Performs Pharmacodynamics (PD) evaluation of drugs in orthotopic tumor models, including tumor growth inhibition (TGI) rates and survival analysis, and integrates with Pharmacokinetics (PK) to optimize dosing regimens.
- Preliminary Study of Drug Resistance and Metastasis Mechanisms: Induces acquired drug resistance through long-term drug administration and utilizes the model's spontaneous metastatic capability to initially explore the molecular mechanisms underlying tumor recurrence and metastasis.
The core applications include:
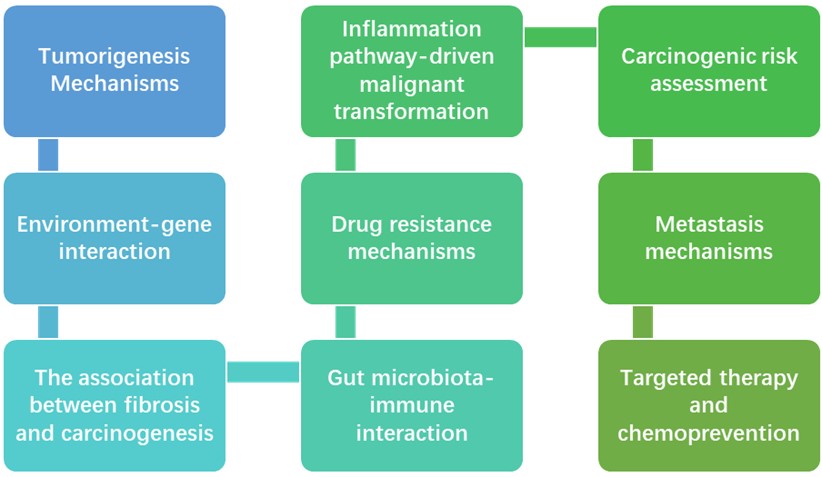
Our Advantages
- Tumor Model Library: Possesses a mature carcinogen - induced model library, covering multiple cancer types and subtypes.
- Capability to Construct Multistage Carcinogenesis Models: Can design and validate multistage carcinogenesis processes, such as the DEN + CCl4 - induced hepatitis, fibrosis, and liver cancer progression.
- Multimodal Dynamic Monitoring Technology: Integrates technologies like in vivo imaging and photoacoustic imaging to real-time monitor tumor growth, metastasis, and changes in the microenvironment's oxygenation.
- In-depth Analysis of Biomarkers and Mechanisms: Analyzes changes in the immune microenvironment, identifies mutations, and studies drug resistance mechanisms. Identifies driver mutations, such as Trp53 and Kmt2d mutations, in the BBN bladder cancer model. Researches drug resistance mechanisms, such as the FGFR3 mutation leading to erdafitinib resistance.
- One-Stop Support: Provides IND-filing-level data packages, including integrated reports for pharmacodynamics (PD), pharmacokinetics (PK), and toxicology (TOX).
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: How to control model fluctuations caused by individual differences?
A: To reduce variability, genetic background control is required, necessitating the use of inbred mouse strains and SPF-level animal facilities with standardized procedures.
-
Q: Can personalized model customization be provided?
A: We support customized solutions, allowing us to adjust the carcinogen dosage, such as optimizing the N-methyl-N-nitrosourea (MNU) concentration for the gastric cancer model, based on the specific cancer type's requirements.
-
Q: Can the model construction cycle be optimized?
A: To meet the need for accelerated modeling techniques, we employ strategies such as the application of aged mice (e.g., in the 4NQO oral cancer model, cancerization rate increases to 92% using mice >65 weeks old) and combined induction strategies (e.g., DEN + CCl4 shortens the liver cancer model creation cycle to 22 weeks).
Published Data
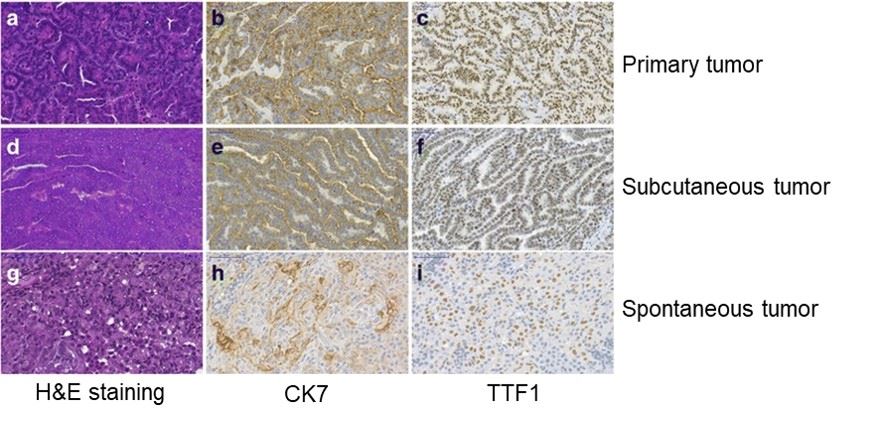 Fig.2 Histological analysis of primary, subcutaneous, and spontaneous lung adenocarcinoma. CK7: Immunostaining of cytokeratin-7, TTF1: Thyroid Transcription Factor-1.2
Fig.2 Histological analysis of primary, subcutaneous, and spontaneous lung adenocarcinoma. CK7: Immunostaining of cytokeratin-7, TTF1: Thyroid Transcription Factor-1.2
In FVB/N mice, DEN successfully induces lung adenocarcinoma whose primary, subcutaneously transplanted, and spontaneous tumors, including their papillary structures, exhibit histological morphology highly similar to that of human lung adenocarcinoma. This impressive fidelity demonstrates the model's significant application potential in translational oncology research and drug development.
References
- Zhou, Yinxi et al. "Experimental mouse models for translational human cancer research." Frontiers in immunology vol. 14 1095388. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3389/fimmu.2023.1095388.
- Mervai, Zsolt et al. "Diethylnitrosamine induces lung adenocarcinoma in FVB/N mouse." BMC cancer vol. 18,1 157. Distributed under Open Access license CC BY 4.0, with modification. https://doi.org/10.1186/s12885-018-4068-4.
For Research Use Only.
