Tumor Organoid Modeling & Pharmacodynamics Service
Introduction
Tumor Organoids are a cutting-edge in vitro three-dimensional (3D) cell culture model, formed through the self-assembly of stem cells or primary cells, capable of highly miniaturizing and simplifying native tissues and organs. They maintain a high degree of consistency with the source tissue in terms of structure, cell types, self-renewal, and function, successfully replicating complex spatial morphology and cell-matrix interactions. Creative Biolabs' Organoid Models, owing to their unique biological characteristics, exhibit vast application potential in both basic research and clinical practice: They not only provide a multi-level experimental system essential for in-depth elucidation of tumor mechanisms and disease pathogenesis, but also serve as an ideal platform for developing novel therapies (such as high-throughput drug screening and target validation). Crucially, because organoids retain the genetic diversity and heterogeneity of the original tissue, they are particularly well-suited for precision medicine research, enabling prediction and guidance for clinical medication.
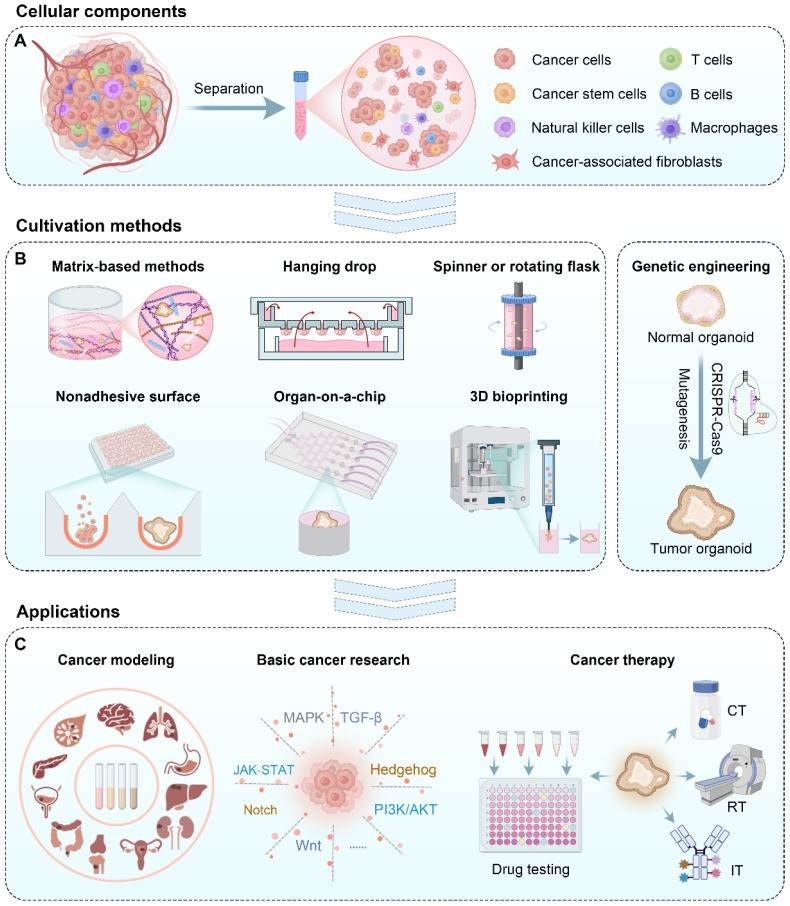 Fig.1 Cellular materials, various tumor organoid construction methods, and their applications to cancer research and therapy. CT: Chemotherapy; RT: radiotherapy; IT: immunotherapy.1,3
Fig.1 Cellular materials, various tumor organoid construction methods, and their applications to cancer research and therapy. CT: Chemotherapy; RT: radiotherapy; IT: immunotherapy.1,3
Available Tumor Organoid Models
Creative Biolabs is dedicated to constructing complex tissue simulations (organoids) and delivering high-standard services. Our organoid library features models meeting application-grade standards, with model diversity and quantity ensured through rigorous QC procedures. We guarantee our models possess the capacity for on-demand revival, expansion, and cryopreservation, providing stable and reliable experimental systems and critical tools for cancer and oncology research.
| Model Type | Modeling Method | Relevant Evaluation |
| Tumor Organoids / Patient derived Organoids (PDOs) | Establishment from fresh or cryopreserved patient tumor tissue (biopsy or surgical resection) and culture in 3D matrix (Matrigel/BME) with specific growth factors. |
Precision Medicine: Individualized Drug Sensitivity Testing (DST), prediction of clinical response, and establishment of drug resistance profiles. Drug Development: High-throughput screening (HTS) of novel compounds, target validation, and disease modeling. |
| Tumor Organoid and Immune Cell Co-culture | Integrating autologous or allogeneic immune components (e.g., T cells, PBMCs, macrophages) with established TOs in the 3D matrix. |
Immunotherapy Efficacy: Evaluation of immune checkpoint inhibitors (ICIs), CAR-T/CAR-NK cell activity, and bispecific antibody efficacy in vitro. TME Study: Investigation of immune cell infiltration, tumor-immune evasion mechanisms, and cytokine release profiles. |
| Gene-Edited Organoids | Utilizing gene editing technology to introduce or correct specific genetic mutations (e.g., p53 knockout, KRAS mutation) in normal or tumor organoids. |
Mechanistic Research: Studying the functional role of key oncogenes or tumor suppressors in transformation and progression. Drug Response Validation: Creating isogenic pairs to confirm the dependency of a drug on a specific mutation. |
| Organoids-on-a-Chip (Microfluidics) | Culturing TOs within microfluidic devices that mimic fluid flow, shear stress, and nutrient gradients found in vivo. |
Advanced TME Simulation: High-fidelity modeling of drug delivery kinetics, metastasis, and interaction with vasculature (e.g., blood-brain barrier models). PK/PD: Establishing more accurate in vitro concentration-response relationships. |
Evaluation Platform
The Creative Biolabs evaluation platform implements strict SOPs and cutting-edge analytical technologies, ensuring all PD and mechanistic results are highly accurate, reproducible, and clinically predictive. By integrating high-throughput instruments and specialized assays, we deliver multi-dimensional analysis. This technical advantage provides clients with mechanistically clear, highly reliable data, directly supporting critical drug development decisions, successful IND submissions, and accelerated clinical translation.
- High Content Screening (HCS) System: Observes and analyzes the growth, morphological changes, and expression of specific markers in cell spheroids and organoids.
- Multispectral Flow Cytometer: Detects and analyzes the relative proportions of different cell types and the expression of cell surface markers.
- Fluorescence and Confocal Microscopes: Used for resolution imaging to observe cellular structures and cell-to-cell interactions.
- Laser Capture Microdissection (LCM) System: Isolates specific cell populations from spheroids or organoids for subsequent analysis.
- Real-time Quantitative PCR (qPCR): Detects gene expression levels to analyze biological changes in cells.
- Mass Spectrometer: Used for metabolomics and proteomics analysis, helping to understand metabolic pathways and protein expression.
- Microplate Reader: Quantitatively analyzes the concentration of cytokines or other soluble factors in cell culture media.
- Label-free Cell Analyzer: Real-time monitors cell proliferation, adhesion, and migration.
- Gene-Editing Tools: Perform specific gene editing in 3D structures to study gene function.
- Bioreactor: Provides a well-controlled environment for large-scale culture of spheroids and organoids.
Applications
- Precision Medicine & Drug Screening: Tumor Organoids, as rapidly established models, enable large-scale Drug Sensitivity Testing to predict individual patient response to chemotherapy or targeted agents, thereby guiding clinical decisions; concurrently, they also allow for the real-time tracking in vitro of phenotype drift and the emergence of drug resistance mechanisms.
- Tumor Biology & Disease Modeling: Tumor Organoids provide authentic modeling by retaining the tumor's native cellular heterogeneity, making them ideal for studying cancer mechanisms and the functional role of hereditary mutations. Furthermore, they facilitate immune interaction studies through co-culture assays, allowing researchers to investigate the complex interplay between the tumor and essential Tumor Microenvironment (TME) components, such as immune cells.
- Drug Development & Preclinical Evaluation: Tumor Organoids significantly boost drug development Efficiency as they are used for both novel target validation and early toxicity assessment against normal tissues. Furthermore, they can be adapted as an HTS Platform by being miniaturized for automated High-Throughput Screening, substantially enhancing cost-effectiveness.
- Metastasis and Drug Resistance Mechanisms: Tumor Organoids are ideal for Mechanistic Study as they allow for the modeling of acquired resistance mechanisms and facilitate the screening of combination therapies to overcome drug refractoriness. Furthermore, they are valuable for Potential Assessment, being used to simulate invasion and migration to evaluate the tumor's metastatic potential.
Our Advantages
- Biological Relevance: These systems are closer to the in vivo environment, authentically simulating cell-to-cell interactions, the tumor microenvironment, and tissue structure, which makes research results more biologically relevant.
- Complex Cell-to-Cell Interactions: Co-culture systems allow for the study of interactions between different cell types, revealing mechanisms that influence tumor growth and metastasis.
- Three-Dimensional Structure: Cancer spheroids and organoids offer a 3D culture system that can better simulate the spatial structure and heterogeneity of tumors, improving the accuracy of drug screening and cancer research.
- Personalized Medicine: 3D culture technology allows for the cultivation of individualized tumor models from patient samples, aiding in precision medicine research by enabling the tailoring of personalized treatment plans through drug screening and genetic analysis.
- Improved Drug Screening: These systems can more effectively predict in vivo drug responses, thereby increasing the success rate of drug development and reducing clinical trial costs.
- Reduction of Animal Experiments: As these systems can better simulate in vivo conditions in an in vitro environment, they can reduce the reliance on animal experiments, which aligns with ethical requirements.
- Innovative Research Platforms: As cutting-edge technology platforms, these services can drive the development of basic research and translational medicine, and contribute to the publication of high-impact scientific papers and patent applications.
- One-Stop Service: We offer a seamless one-stop service, allowing you to validate findings from our advanced in vitro models with follow-up in vivo tumor model studies, complete with pharmacokinetic (PK) and toxicological (TOX) evaluations.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q. What are the advantages of providing organoid model services?
A: They more accurately simulate the complex in vivo environment. They provide more physiologically relevant data than traditional 2D cultures. They can be used for drug screening and disease modeling, enhancing the ability to predict drug efficacy and safety.
-
Q. Can you customize your services?
A: Yes, we offer multiple customization options to ensure we meet our clients' specific research needs. Please contact our project manager to discuss your detailed requirements.
-
Q. What types of cells and tissues can your company handle?
A: We can handle a wide range of cell and tissue types, including human and animal cells. Please contact us to confirm specific types.
-
Q. How are the timeline and costs determined?
A: The timeline and costs depend on the project's complexity and customization needs. Generally, basic projects are completed within a few weeks.
-
Q. What technologies do you use for organoid culture?
A: We use the latest biotechnological tools, including matrix gels, microfluidic technology, and bioreactors, to support 3D culture.
-
Q. How do you validate the effectiveness of my experimental models and results?
A: We provide detailed experimental reports, including culture conditions, validation methods, and results analysis. We also offer follow-up support to assist with data interpretation.
-
Q. How do you ensure my data is safe and confidential?
A: We follow strict security protocols and data management policies to ensure your data is protected. We sign a Non-Disclosure Agreement (NDA) with every client to ensure information security.
-
Q. Do you provide experimental data analysis and reporting services?
A: Yes, we provide comprehensive data analysis and customized reporting services to ensure you can make the most of your experimental results.
Published Data
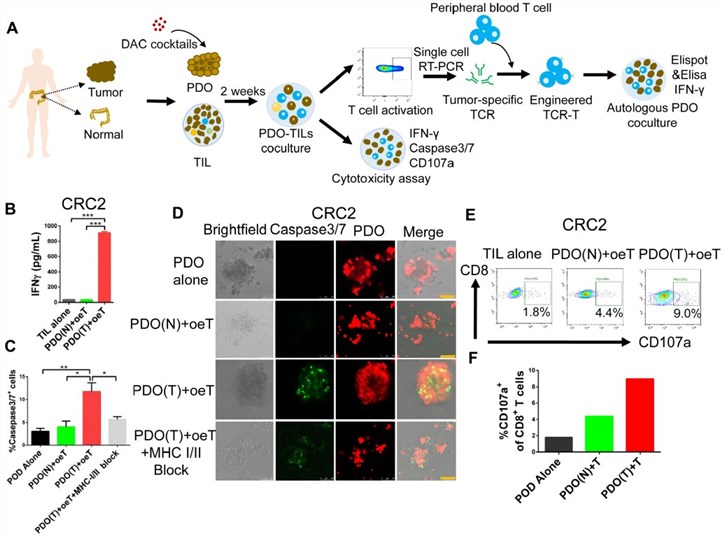 Fig.2 Cytotoxic activity of tumor oeT.2,3
Fig.2 Cytotoxic activity of tumor oeT.2,3
In a co-culture system of tumor organoids and autologous tumor-infiltrating lymphocytes (TILs), organoid enriched TILs (oeT) cells can specifically recognize and respond to autologous tumor antigens. They have a selective killing effect on tumor cells, and this killing activity is dependent on MHC molecule-mediated antigen recognition.
References
- Lv, Jiajing et al. "Construction of tumor organoids and their application to cancer research and therapy." Theranostics vol. 14,3 1101-1125. https://doi.org/10.7150/thno.91362
- Li, Zhilang et al. "Identification and validation of tumor-specific T cell receptors from tumor infiltrating lymphocytes using tumor organoid co-cultures." Cancer immunology, immunotherapy: CII vol. 73,9 164. 2 Jul. 2024, doi:10.1007/s00262-024-03749-8
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
