CD19⁺ Humanized Cell Line derived Xenograft (CDX) Model based CAR-T Efficacy Modeling & Pharmacodynamics Service
Introduction
A CD19 positive cell line derived xenograft (CDX) model involves the transplantation of human cancer cell lines expressing the CD19 antigen into immunodeficient mice. Creative Biolabs offers a sophisticated, highly translational CD19 positive CDX model established in humanized mouse models to provide comprehensive preclinical validation for CD19-targeted therapies, particularly CAR-T cells. This platform uniquely overcomes traditional limitations by incorporating a functional human immune system, allowing for the rigorous evaluation of efficacy and mechanism-of-action (MoA) assessments. We concurrently track critical pharmacodynamics (PD) metrics, such as CAR-T persistence, tumor killing rate, and cytokine release profiles, alongside crucial safety indicators, including monitoring for cytokine release syndrome (CRS), neurotoxicity (ICANS), and systemic organ toxicity.
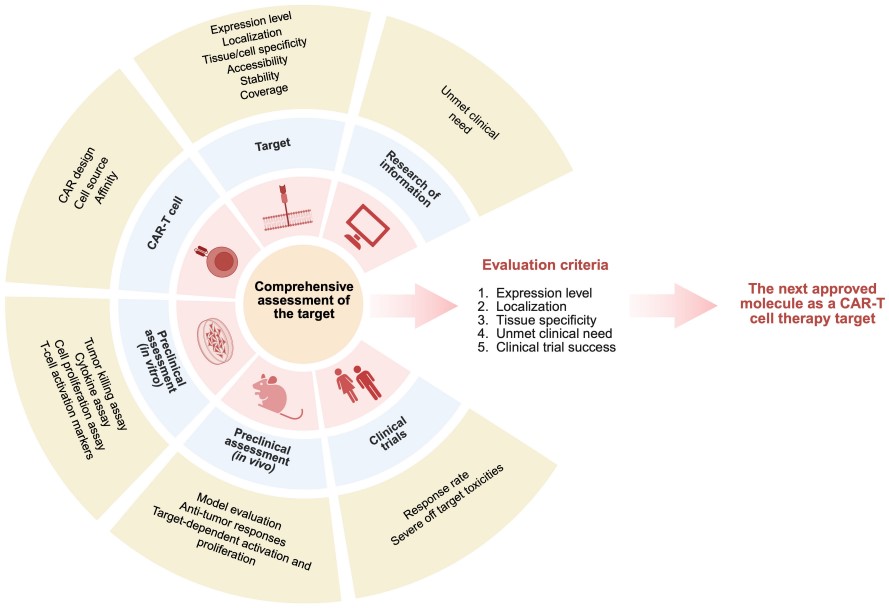 Fig.1 Comprehensive criteria for evaluating potential targets across all stages of CAR‑T development.1,4
Fig.1 Comprehensive criteria for evaluating potential targets across all stages of CAR‑T development.1,4
Available CD19 Positive CDX in Humanized Mouse Model for CAR-T Efficacy Service
The triple-immunodeficient mouse serves as the foundation for human cell engraftment. We utilize PBMC engraftment for rapid, short-term efficacy evaluation and acute toxicity (CRS proxy) assessment. Conversely, CD34+ HSC engraftment is deployed for chronic studies requiring long-term CAR-T persistence, detailed PD analysis, and the monitoring of critical long-term safety endpoints like B-cell aplasia.
| Tumor Cell Lines (CDX Model) | Characteristics & Preclinical Value |
| NALM-6 | Highly aggressive, human Pre-B Acute Lymphoblastic Leukemia (ALL) (CD19+), often inoculated intravenously (IV) to establish systemic leukemia. Ideal for rapid efficacy testing, dose-titration studies, and evaluating CAR-T cell capacity to clear systemic leukemia before significant tumor burden develops. |
| Raji | Derived from human Burkitt's Lymphoma (CD19+). Can be inoculated intravenously or subcutaneously (SC) to form solid/lymphoma masses. Used for assessing CAR-T efficacy against aggressive solid/lymphoma masses and for monitoring key safety signals like CRS proxies and CAR-T persistence. |
| Daudi | Derived from human Burkitt's Lymphoma (CD19+). Often inoculated SC or IV. Provides an alternative lymphoma genetic background to Raji, offering a different profile for comprehensive resistance testing and confirmation of broad CAR-T applicability. |
| KMS-11 | Human Multiple Myeloma (MM) cell line (CD19+ low), which is present in MM precursor cells and some relapsed/refractory MM. Used to evaluate CAR-T therapy efficacy against malignancies expressing low levels of CD19 and to explore strategies targeting early-stage or minimal residual disease in Multiple Myeloma. |
| Pfeiffer | Human Diffuse Large B Cell Lymphoma (DLBCL) cell line (CD19+). DLBCL is the most common aggressive Non-Hodgkin Lymphoma (NHL). This model assesses CAR-T performance against aggressive, high-tumor-burden solid/lymphoma masses, testing CAR-T infiltration capacity and persistence required for DLBCL treatment. |
| Sup-B15 | Human B Cell Acute Lymphoblastic Leukemia (B-ALL) cell line (CD19+). Serves as an alternative B-ALL model to NALM-6, used to validate the universality (generalizability) of the therapeutic agent and its efficacy against ALL subtypes with diverse genetic backgrounds. |
Evaluation Platform
Our platform employs a multi-faceted approach, combining gold-standard efficacy metrics with advanced molecular tracking, ensuring comprehensive CAR-T validation. Leveraging international-class, state-of-the-art instrumentation and scientifically rigorous, advanced assay protocols, we are dedicated to generating highly reliable, high-precision research results, comprehensively accelerating your R&D decision-making process.
-
Primary Efficacy Endpoints
- Tumor Load Monitoring: We provide precise, longitudinal measurement of tumor volume using calipers and high-throughput non-invasive imaging techniques, primarily bioluminescent imaging (BLI), complemented by computed tomography (CT) and magnetic resonance imaging (MRI) when needed.
- Survival Rate Analysis: We record and analyze the survival time of the treatment group versus the control group using Kaplan-Meier curves, providing the definitive endpoint for therapeutic success.
-
CAR-T Kinetics & Dynamics
- CAR-T Cell Kinetics: We use highly sensitive techniques, including quantitative PCR (qPCR) or digital droplet PCR (ddPCR) on DNA from blood and tissues, to measure CAR gene copy numbers, allowing for precise absolute quantification and tracking of CAR-T cell persistence over time.
- Distribution and Persistence: We track the non-invasive distribution and persistence of CAR-T cells (often engineered with a reporter gene) using BLI.
- Gene Expression Analysis: We utilize RT-qPCR to measure the mRNA expression levels of the CAR gene itself or other relevant pharmacodynamic genes, reflecting the CAR-T product's in vivo activity status.
-
Mechanistic, Safety & Toxicity Analysis
- Immunological Analysis (Micro-Environment): We analyze changes in immune cell subsets in tumor tissue and peripheral blood using multi-parameter flow cytometry and advanced spatial techniques like Multiplex Immunofluorescence (mIF), allowing for deep mechanistic insight into the TME.
- Cytokine Analysis: We employ ELISA or high-throughput multiplex cytokine assays (Luminex, MSD) to measure systemic cytokine levels in serum, enabling the critical preclinical assessment of the risk of CRS.
- Histopathological Analysis: We collect and fix organ and tumor tissues for pathological analysis (H&E, IHC) to evaluate the toxicity of CAR-T cells to normal tissues (on-target/off-tumor safety) and the extent of immune infiltration.
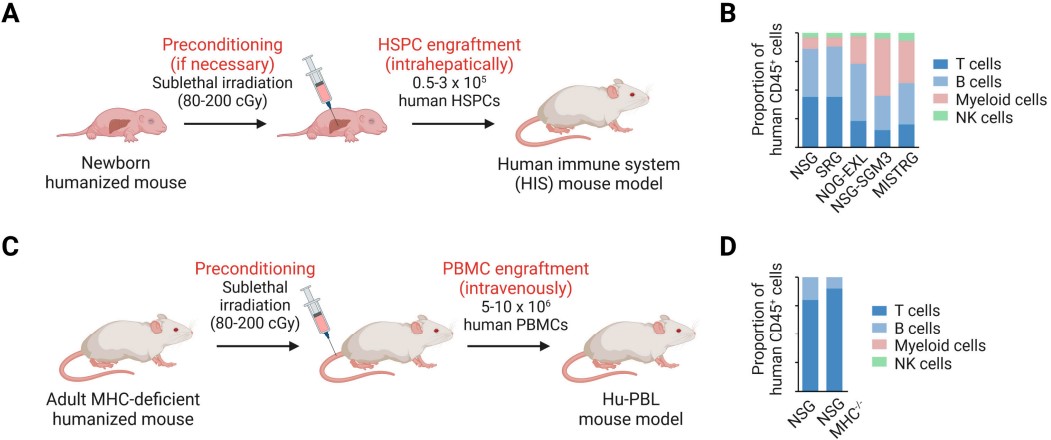 Fig.2 Establishment of human immune system (HIS) mouse models.2,4
Fig.2 Establishment of human immune system (HIS) mouse models.2,4
Applications
- Targeted Efficacy Assessment: The model serves as the definitive platform for the preclinical evaluation of therapies explicitly designed to target CD19-expressing cancer cells, such as CAR-T therapies.
- Mechanism of Resistance Insights: We provide crucial insights into treatment efficacy, PD, and the underlying mechanisms of resistance, enabling data-driven optimization of your therapeutic candidate.
- Guiding Clinical Translation: By offering robust, quantifiable in vivo data, this specialized CDX model directly supports the clinical translation of CD19-targeted cellular and biologic therapies, accelerating your path toward IND filing and clinical success.
Our Advantages
- Expertise in Immuno-Oncology: Deep understanding and experience in immuno-oncology and the development of CAR-T cell therapies, including expertise in designing and conducting preclinical studies in relevant models.
- Advanced Animal Facilities: State-of-the-art animal facilities equipped to house and monitor humanized mouse models. This includes the ability to maintain sterility and provide specialized care to immunocompromised animals.
- Comprehensive Analytical Capabilities: A wide range of analytical techniques for evaluating CAR-T cell function and efficacy, including flow cytometry, PCR, bioluminescent imaging, and histopathology.
- Experienced Scientific Team: A team of experienced scientists and technicians skilled at executing complex study designs and interpreting results accurately to provide meaningful insights into CAR-T cell efficacy and safety.
- Collaborative Approach: A collaborative approach to project management, working closely with clients to customize study designs and provide regular updates and strategic advice on study progress.
- Proven Track Record: A solid track record of successful CAR-T cell preclinical evaluations, including published research and case studies.
- Innovative Technologies: Access to the latest technologies and methods to improve the precision and reliability of preclinical assessments, such as advanced imaging technologies and next-generation sequencing.
- Scalability and Flexibility: The ability to scale up studies efficiently and offer flexible solutions tailored to the specific needs of different CAR-T cell projects.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q. Why use a humanized mouse for CAR-T evaluation?
A: Because CAR-T cells are human-specific, a standard immunodeficient mouse lacks a human immune system, so it can't fully replicate how the CAR-T cells would behave in a patient.
-
Q: What are the applications of the humanized mouse model?
A: The applications of the platform include assessing CAR-T cell expansion and persistence, evaluating off-target effects and potential toxicities to healthy human immune cells, and modeling CRS, a severe side effect caused by an overactive immune response.
-
Q: What is the typical timeframe for a CAR-T efficacy study?
A: The overall project timeline is typically 14–20 weeks. This includes the 8–12 week period required for the human immune system to fully engraft and mature in the host, followed by a 6–8 week intensive efficacy study period.
-
Q: What is the success rate for human immune cell engraftment?
A: Utilizing highly optimized protocols with triple-immunodeficient strains, we achieve human immune cell engraftment success rates of over 90% for robust study initiation.
-
Q: Do you support CAR-NK or TCR-T evaluation in this platform?
A: Yes. This platform is flexible and also suitable for evaluating other human-specific adoptive cell therapies, including CAR-NK cells and TCR-T cells, using appropriate CD19 expressing targets.
-
Q: Why use a CDX model instead of a PDX model for early CAR-T validation?
A: The CDX model (using cell lines like NALM-6) offers high stability, standardization, and predictable CD19 expression, ideal for establishing proof-of-concept and reliable basic efficacy/toxicity (PK/PD) metrics during early screening.
-
Q: What are the core PD efficacy endpoints assessed in this model?
A: Core PD endpoints include CAR-T cell persistence and trafficking (quantified in blood/tissue), target cell killing rate (CD19+ tumor elimination), and the cytokine release profile (e.g., quantifying IL-6, IFN-γ).
-
Q: How do you assess CAR-T cell safety and toxicity?
A: Safety is assessed by monitoring clinical signs associated with CRS (e.g., body weight, temperature, activity levels) and ICANS (e.g., tremors, ataxia), complemented by serum biochemistry for systemic organ toxicity.
Published Data
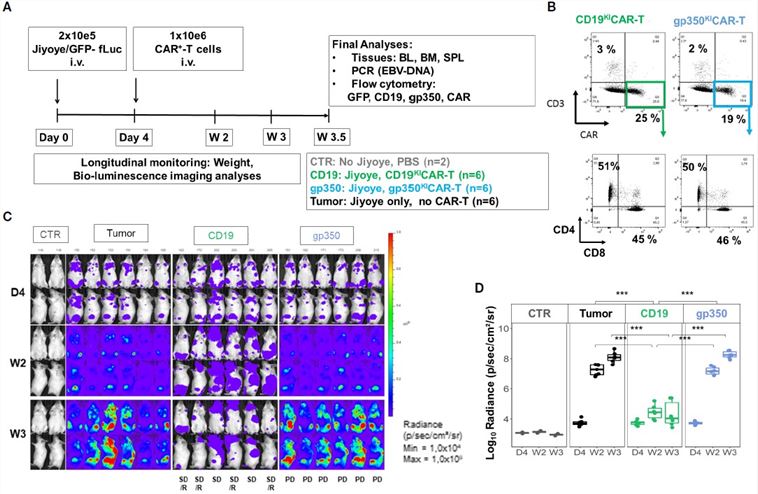 Fig.3 In vivo efficacy of CAR-T in the Daudi model.3,4
Fig.3 In vivo efficacy of CAR-T in the Daudi model.3,4
The in vivo efficacy study, utilizing CAR-T cell phenotyping and quantitative BLI in an EBV Type II Jiyoye lymphoma model, demonstrated that the CD19 KI CAR-T significantly inhibited tumor growth and dissemination, while the gp350 KI CAR-T exhibited no therapeutic activity, providing direct in vivo evidence for the differential efficacy of the two constructs.
References
- Ershova, Alina et al. "Future perspectives on novel CAR-T therapeutics beyond CD19 and BCMA in onco-hematology." Frontiers in immunology vol. 16 1592377. https://doi.org/10.3389/fimmu.2025.1592377
- Chen, Anna et al. "Modeling the Tumor Microenvironment and Cancer Immunotherapy in Next-Generation Humanized Mice." Cancers vol. 15,11 2989. https://doi.org/10.3390/cancers15112989
- Braun, Tobias et al. "Non-viral TRAC-knocked-in CD19KICAR-T and gp350KICAR-T cells tested against Burkitt lymphomas with type 1 or 2 EBV infection: In vivo cellular dynamics and potency." Frontiers in immunology vol. 14 1086433. https://doi.org/10.3389/fimmu.2023.1086433
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
