Protein Degrader based Efficacy Modeling & Pharmacodynamics Service
Introduction
Proteolysis-Targeting Chimeras (Protein Degrader) technology represents a transformative new paradigm in oncology, operating as a bifunctional molecular entity that harnesses the cell's own ubiquitin-proteasome system by recruiting E3 ubiquitin ligases to induce the directed degradation of specific onco-proteins; this mechanism offers a significant advantage by effectively overcoming previously inaccessible targets and existing drug resistance pathways, thereby greatly expanding the therapeutic modalities available for malignant disease. Leveraging a deep understanding of the Protein Degrader mechanism and critical evaluation parameters, Creative Biolabs' service provides specialized preclinical models (e.g., PDX models, humanized E3 Ligase models) and customized pharmacokinetics (PK) / pharmacodynamics (PD) assessment to ensure the most rigorous validation of these novel degraders.
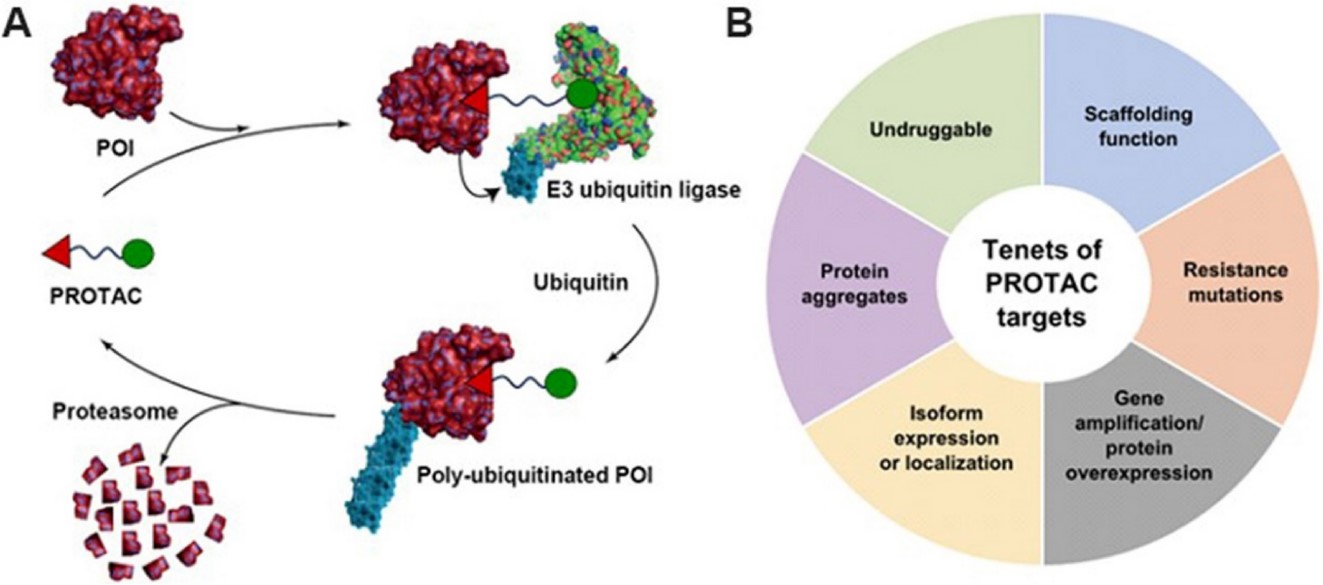 Fig.1 A Protein Degrader-mediated degradation of target proteins through the UPS; B The tenets of Protein Degrader targets.1,3
Fig.1 A Protein Degrader-mediated degradation of target proteins through the UPS; B The tenets of Protein Degrader targets.1,3
Available Protein Degrader based Efficacy Services
Model selection is the critical first step in Protein Degrader pharmacodynamic assessment, customized based on the specific E3 ligase recruited and the anticipated clinical application of the Protein Degrader molecule, ensuring high scientific value and translational potential.
| Category | Animal Modeling | Characteristics | Key Evaluation Values |
| von Hippel-Lindau (VHL) | Subcutaneous/Orthotopic Xenograft Models, PDX Models | Recruits VHL to mediate target protein degradation; highly efficient and common. | Degradation efficiency (Dmax), degradation concentration (DC50), and impact of linker length optimization on degradation kinetics. |
| Cereblon (CRBN) | Orally dosed Xenograft Models, PDX Models | Recruits CRBN for degradation; mature ligands and high oral feasibility. | Oral bioavailability (PK) correlation with PD, duration of catalytic effect (critical for dosing frequency). |
| Inhibitor of Apoptosis Protein (IAP) | Immunocompetent or Humanized Immune System Models | Recruits IAP for degradation, potentially inducing simultaneous apoptosis. | Assessment of the ratio between the degradation effect and the apoptotic effect; impact on immune function in relevant models. |
| Heterobifunctional Protein Degraders | CDX/PDX Models | Standard Protein Degrader design: three components (ligand-linker-E3 ligand). | Efficiency of ternary complex formation, chemical stability of the linker, and overall in vivo distribution. |
| Molecular Glues | CDX Models | No discernible linker; small molecules directly mediate novel protein-protein interaction (PPI). | In vitro selectivity (off-target risk), in vivo MoA confirmation (verifying the induction of a novel PPI), distinction from Protein Degraders. |
| Lysosome-Targeting Chimeras (LYTACs) | Orthotopic Models (for membrane protein targets) | Targets cell membrane/secreted proteins for degradation via the lysosomal pathway. | Cellular internalization efficiency, lysosomal escape rate, and lysosomal degradation kinetics. |
| Autophagy-Targeting Chimeras (AUTOTACs) | Models involving large molecular aggregates or difficult-to-clear proteins | Utilizes the autophagy-lysosome pathway for degradation. | Verification of autophagosome formation, co-localization analysis of autophagosomes and the target, and assessment of autophagic flux. |
| Ribosome-Targeting Chimeras (RIBOTACs) | Cell Line Models (primarily for in vitro validation) | Targets and degrades non-coding RNA by recruiting E3 to the RNA molecule. | RNA degradation efficiency (non-protein endpoint), RNA half-life, and impact on mRNA translation. |
Evaluation Platform
Leveraging world-class, cutting-edge technology platforms and precision instrumentation, we specialize in providing high-standard preclinical pharmacology and efficacy (PK/PD) assessments. We are committed to delivering superior, high-precision research results and excellent service, comprehensively accelerating the transition of your innovative drugs from the lab to the clinic.
-
In vivo models:
- Subcutaneous Cell Line Derived Xenografts (CDX) Models: CDX models are the foundation for early screening and dose finding. Initial screening and dose exploration for standard VHL/CRBN Protein Degraders. These models are cost-effective, rapid, and feature high tumor take rates. This facilitates the quick acquisition of efficacy data and the easy collection of tissue samples for rapid target degradation analysis.
- Patient derived Xenograft (PDX) Models: PDX models are essential for increasing clinical translational success. PDX models better preserve the heterogeneity and biology of the original human tumor. The efficacy data derived from these models are more predictive of the Protein Degrader's potential success in complex human clinical settings.
- Orthotopic Models: Orthotopic models focus on the Protein Degrader's behavior within its native physiological environment. By implanting tumor cells in their organ of origin, these models accurately simulate the authentic tumor microenvironment and vasculature. This is vital for assessing the Protein Degrader's distribution, barrier penetration (e.g., Blood-Brain Barrier), and efficacy against spontaneous metastasis.
- Immuno-Competent/ Genetically Engineered Mouse Models (GEMMs): Immuno-competent models are the gold standard for complex mechanism evaluation. These models are crucial for Protein Degraders that influence immune components or require an intact host immune system (e.g., IAP-Protein Degraders) for full pharmacodynamic effect. They assess bystander effects on immune cells and systemic tolerability in a native setting.
- Humanized Animal Models: Through gene editing technologies, models are engineered to express humanized E3 ligases (replacing murine Crbn or Vhl with human CRBN or VHL) or humanized target proteins (when the target displays significant species divergence) to ensure appropriate Protein Degrader recognition and binding affinity, thereby guaranteeing preclinical relevance.
-
PD Readouts:
- Efficacy: Tumor growth inhibition (TGI) and overall survival (OS) analysis conducted in high-translatability animal models. Monitoring of body weight changes (a key indicator of overall health), organ toxicological analysis (histopathology), and assessment of potential immune-related toxicities in humanized E3 ligase models.
- Mechanism Confirmation and Target Modulation: To confirm the drug engages its target and triggers the intended molecular pathway. Using techniques like Western Blot or immunohistochemistry (IHC) to quantify target protein degradation (e.g., Protein Degrader's DC50 or Dmax) or measure the phosphorylation status of downstream signaling proteins.
- Functional and Cellular Response: To evaluate the drug's direct impact on cell behavior (proliferation, apoptosis, immune activity). Assessing proliferation with markers like Ki-67 and apoptosis with markers like cleaved Caspase-3 via IHC. Using flow cytometry (FACS) to analyze the number, subtypes (e.g., CD4+, CD8+), and activation status of tumor-infiltrating lymphocytes (TILs) in various tissues.
- Clinical Translational Markers and Safety Proxy: To quantify systemic immune responses, predict potential toxicity, and guide clinical dosing. Employing highly sensitive platforms like Luminex or ELISA to quantify systemic or local cytokine release (e.g., IL-6, IFN-γ). These markers are crucial for predicting the risk of cytokine release syndrome (CRS) and assessing overall immune activation.
Applications
- Early Screening & Structural Optimization: Utilizing DC50, ternary complex formation assays, and CDX models, unsuitable Protein Degrader candidates are rapidly screened and eliminated in the early phase, thereby effectively guiding the design of critical structures like linkers and ligands.
- Preclinical Efficacy Assessment: TGI and survival analyses are conducted on high-translatability platforms like PDX and Orthotopic models, complemented by validation using humanized E3 models, to quantify the Protein Degrader's in vivo efficacy and predict clinical success rates.
- Mechanism Elucidation: The key PK/PD correlation is established, linking drug concentration to the sustained degradation effect of the target protein, while targeted proteomics and histopathology are used to comprehensively confirm the degradation mechanism and safety profile.
Our Advantages
- Specialized Expertise: We have deep expertise in working with Protein Degraders, including knowledge of their unique mechanism of action and the technical skills required for handling these compounds.
- Comprehensive Animal Model Platform: Access to a wide range of relevant animal models. Capability to custom develop models specific to the needs of Protein Degrader evaluation.
- Advanced Analytical Capabilities: Equipped with state-of-the-art analytical tools for PK, PD, and biomarker analysis. Capability to perform high-throughput screening and sophisticated imaging techniques to assess tumor response and drug distribution.
- Strong Project Management: A dedicated project management team to ensure streamlined study design, execution, and reporting. Flexibility and responsiveness to adjust study protocols as needed based on interim data.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q. Which animal model should I prioritize for evaluating Protein Degrader efficacy?
A: There is no one-size-fits-all model: Model selection for prodrug degraders must be customized according to the research purpose, wherein Subcutaneous Xenograft Models are preferred for initial screening and MoA validation due to their cost-effectiveness, shorter timelines, and high tumor take rates, allowing for rapid efficacy data acquisition and degradation analysis; PDX models, which better preserve the heterogeneity and biology of the original tumor, are utilized for translational and predictive research as their efficacy data is more predictive of clinical success; finally, humanized immune system models are the gold standard for assessing immune-related efficacy and toxicity.
-
Q. Why is it necessary to develop drug-resistant models?
A: A key advantage of Protein Degraders is overcoming drug resistance, which is their core value proposition.
-
Q. What are the irreplaceable advantages of orthotopic models over subcutaneous models?
A: Orthotopic models, which implant tumor cells in their organ of origin, more closely mimic the clinical microenvironment, allowing for a better assessment of the Protein Degrader's tissue penetration and distribution, its inhibitory effects on spontaneous metastasis, and bystander effects on other cells within the authentic tumor microenvironment.
-
Q. Why is the durability of efficacy important?
A: The catalytic nature of Protein Degraders causes their degradative effect to long outlast their PK half-life, and demonstrating that a single dose yields sustained degradation for days provides a key rationale for determining clinical dosing regimens, improving patient compliance, and scientifically estimating the first-in-human dose.
-
Q5. What are the key metrics for the efficacy of Protein Degraders in tumor models?
A: Key metrics encompass anti-tumor activity, measured by tumor regression, TGI%, and overall survival; mechanism of action validation, achieved by quantifying the percentage and duration of target protein degradation, downstream pathway inhibition, and apoptosis/proliferation markers; and safety and tolerability, determined by monitoring body weight change, clinical signs of toxicity, and histopathology of major organs.
Published Data
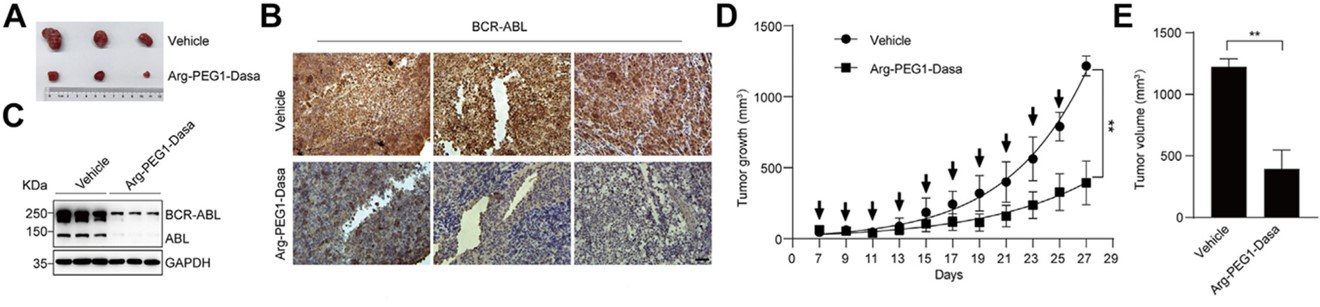 Fig.2 Novel Protein Degrader Arg-PEG1-Dasa achieves significant tumor growth inhibition via BCR-ABL degradation in vivo.2,3
Fig.2 Novel Protein Degrader Arg-PEG1-Dasa achieves significant tumor growth inhibition via BCR-ABL degradation in vivo.2,3
The in vivo anti-tumor efficacy was validated in the nude mouse K562 Chronic Myeloid Leukemia (CML) xenograft model. Treatment with the novel degrader Arg-PEG1-Dasa achieved highly efficient degradation of the key oncoprotein BCR-ABL, resulting in statistically significant tumor growth inhibition. Detailed evidence from growth curves, volume statistics, and immunohistochemistry collectively confirms the complete in vivo mechanism-of-action chain.
References
- Wang, Chao et al. "New-generation advanced Protein Degraders as potential therapeutic agents in cancer therapy." Molecular cancer vol. 23,1 110. https://doi.org/10.1186/s12943-024-02024-9
- Zhang, Jianchao et al. "Single amino acid based Protein Degraders trigger degradation of the oncogenic kinase BCR-ABL in chronic myeloid leukemia (CML)." The Journal of biological chemistry vol. 299,8 (2023): 104994. https://doi.org/10.1016/j.jbc.2023.104994
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
