RNA Therapy based Efficacy Modeling & Pharmacodynamics Service
Introduction
The unique ability of RNA therapies to precisely control gene expression, be developed rapidly, and hit previously undruggable targets has made them a central focus for breakthroughs in oncology, rare diseases, infectious diseases, and cardiovascular disease. The selection and customization of animal models are the most critical steps in RNA therapy evaluation, as the model must precisely match the drug's mechanism of action to ensure scientific rigor and translational accuracy. Drawing on years of experience in tumor modeling, pharmacology, efficacy, and toxicology assessment, Creative Biolabs is well-equipped to evaluate these advanced RNA therapies.
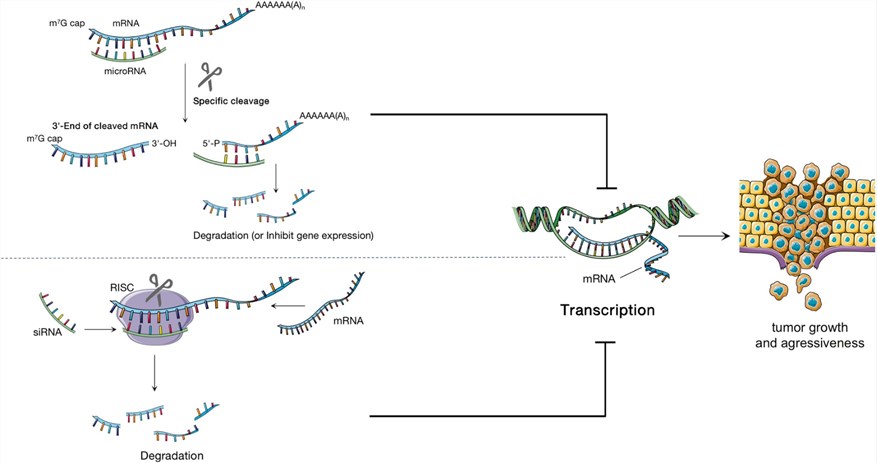 Fig.1 Small non-coding RNA influences tumor development.1
Fig.1 Small non-coding RNA influences tumor development.1
Available RNA based Efficacy Services
Our service provides mechanism-driven pharmacological and efficacy services that are highly customized according to the mechanism of action (MoA) and molecular type (e.g., mRNA, siRNA, ASO) of the RNA therapeutic. This specialized advantage rigorously addresses the inherent delivery and stability challenges of RNA molecules, ensuring the precise quantification of key PD parameters like gene silencing efficiency, immune activation, and target enrichment, thereby maximizing clinical translation rates and accelerating your drug development pipeline.
| Classification | Characteristics | Animal Modeling & Key Evaluation Values |
| mRNA therapy | Transient expression of antigens or immunostimulatory proteins, aimed at activating the host immune system. | Immuno-Competent Tumor Models (Syngeneic Models) or Humanized Immune System Models. Assessing Immunogenicity, T cell response, and TME modulation (e.g., cytokine secretion, T cell infiltration). |
| siRNA therapy | Mediates degradation of target mRNA via the RISC complex to achieve oncogene silencing. | CDX/PDX Tumor Models overexpressing the target. Quantification of Target mRNA Knockdown Efficiency (RNA and protein levels) and correlation with tumor growth inhibition (TGI). |
| miRNA therapy | Modulates gene expression by inhibiting translation or degrading mRNA, often used for tumor suppression. | PDX Models with miRNA deletion/overexpression. Studying the Modulation of Complex Gene Regulatory Networks and validation of miRNA as novel tumor suppressors or oncomiRs. |
| saRNA therapy | Possesses self-replicating ability, allowing for sustained protein expression at lower doses. | CDX Tumor Models with low dose administration. Assessing Sustained Efficacy and Lower Dosing Regimens. Crucial for comparing durability and toxicity versus conventional mRNA at equivalent doses. |
| Ribozymes | Catalytically active RNA molecules that directly cleave the target oncogenic mRNA. | CDX Models overexpressing the target mRNA. Assessing Targeted Catalytic Cleavage Efficiency and determining the required intracellular concentration to achieve therapeutic target elimination. |
| sgRNA | Guides the Cas enzyme to specific gene loci for gene knockout or repair (used for tumor therapy/prevention). | Gene Editing Reporter Models, PDX Models. Precise Genomic Editing Fidelity and Off-Target Analysis. Essential for validating the therapeutic sgRNA sequence and assessing the long-term safety of the edit. |
| Antisense Oligonucleotide (ASO)therapy | Single-strand DNA/RNA analogs that regulate oncogenic mRNA via RNase H or steric hindrance. | CDX/PDX Models overexpressing the target mRNA. Assessing Target Splicing Modulation and RNase H Degradation Capacity. Key for evaluating systemic ASO delivery efficiency and mRNA processing alteration. |
| Nucleic Acid Aptamer (Aptamer) | Acts as chemical antibodies, binding to cell surface receptors or tumor factors with high affinity. | Cancer Targeting Models, circulating target models. Evaluating Target Binding Affinity, Tumor-Specific Accumulation, and PK/PD Correlation. Crucial for assessing internalization mechanisms and therapeutic factor neutralization. |
Evaluation Platform
Leveraging our deep, long-standing expertise in in vivo oncology models and efficacy assessment, our customized model platform provides the precision required to dissect the unique mechanisms of RNA therapeutics: from quantifying the direct inhibitory effect on oncogene expression, to evaluating the core function of activating the host immune system, and verifying the ability to overcome physiological barriers and achieve precise targeting, ensuring the most reliable translational validation of your RNA drug's efficacy.
- Subcutaneous Xenograft Models (CDX/PDX): CDX models are used for the quick, high-sensitivity assessment of basic gene silencing efficacy in an environment with high target mRNA expression. PDX models are used to evaluate silencing efficacy and therapeutic response within the context of tumor heterogeneity, improving clinical relevance. The tumor tissue must ensure sufficient target mRNA expression to accurately quantify the RNA molecule's silencing effect.
- Immuno-Competent Syngeneic Models: These serve as the foundational model for assessing immune effects, allowing for the measurement of the Cytotoxic T Lymphocyte (CTL) response induced by the mRNA-expressed antigen and the indirect anti-tumor effects within a fully functional host immune system.
- Humanized Immune System Models: Used to assess human-specific immune recognition and response to the RNA product, including cytokine release and immune cell activation. These models provide the highest level of translational data for clinical application.
- Orthotopic Models: Function in Assessment: Used to verify the penetration and targeting efficiency of the RNA molecule's delivery vector (e.g., LNP) within the tumor's natural organ environment. This is crucial for assessing efficacy against tumors that require Blood-Brain Barrier (BBB) penetration or organ-specific delivery (e.g., liver or lung). Must accurately mimic the histological and microenvironmental characteristics of the tumor's organ of origin.
Applications
- Early Candidate Screening & Optimization: This application focuses on rapid in vitro screening to determine RNA stability and initial knockdown/expression efficiency (EC50), guiding the optimization of the RNA sequence and chemical modifications.
- RNA Delivery System Efficacy Adaptability Assessment: This critical step tracks the pharmacokinetics (PK) of the complex using live in vivo imaging and qPCR tissue analysis to quantify vector accumulation and functional RNA enrichment at the target site.
- Preclinical Efficacy Assessment: This service provides definitive in vivo data by quantifying TGI, CR, and Overall Survival in customized animal models (PDX, Syngeneic), and establishing the crucial PK-PD correlation to optimize clinical dosing.
Our Advantages
- Expertise and Experience: We have extensive experience and specialized knowledge in RNA therapies and oncology research, particularly in areas like RNA interference or mRNA vaccines.
- Multidisciplinary Team: A skilled team comprising molecular biologists, pharmacologists, toxicologists, and biostatisticians is critical for providing comprehensive services and consultancy.
- Advanced Technology Platforms: We have cutting-edge technology and equipment, such as high-throughput sequencing, RNA analysis tools, and bioimaging technologies, to conduct in-depth molecular and cellular analyses.
- Flexibility and Adaptability: The ability to tailor study protocols to client needs and to adjust experimental designs flexibly during the research process is crucial.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q. Why do RNA therapeutics require a specialized preclinical evaluation platform?
A: Due to their inherent molecular instability, dependency on specialized delivery vehicles, and potential immunogenicity, RNA therapeutics fundamentally differ from traditional drugs, thus necessitating a specialized platform to simultaneously evaluate the RNA molecule, the delivery vehicle, and the combined complex for both efficacy and safety.
-
Q. In evaluating an RNA cancer therapy, how can we prove it reaches the tumor?
A: Proving that an RNA therapeutic accurately reaches the tumor is a critical part of efficacy assessment, typically achieved by real-time tracking of drug distribution using fluorescent or radioactively labeled vectors combined with in vivo imaging, and by quantifying the RNA molecule's tumor enrichment and half-life using highly sensitive techniques like qPCR on tissue samples collected at different time points.
-
Q. How do you assess the impact of an RNA therapeutic on the tumor immune system?
A: Assessing the immune effect of many RNA cancer therapies designed to activate the host's immune system requires co-culturing the RNA therapeutic with immune cells in vitro to measure activation markers and cytokine secretion using flow cytometry, and analyzing the types and proportions of immune cells infiltrating the tumor by collecting tumor tissue, spleens, or lymph nodes from in vivo animal models.
-
Q. What is an off-target effect of an RNA therapeutic, and how is it evaluated?
A: An off-target effect occurs when an RNA therapeutic (such as siRNA) unintentionally binds to sequences similar to the intended target, leading to the silencing of these off-target genes and potential toxicity; this is evaluated by comparing gene expression differences between treated and control groups to identify all affected genes and assess potential toxicity.
Published Data
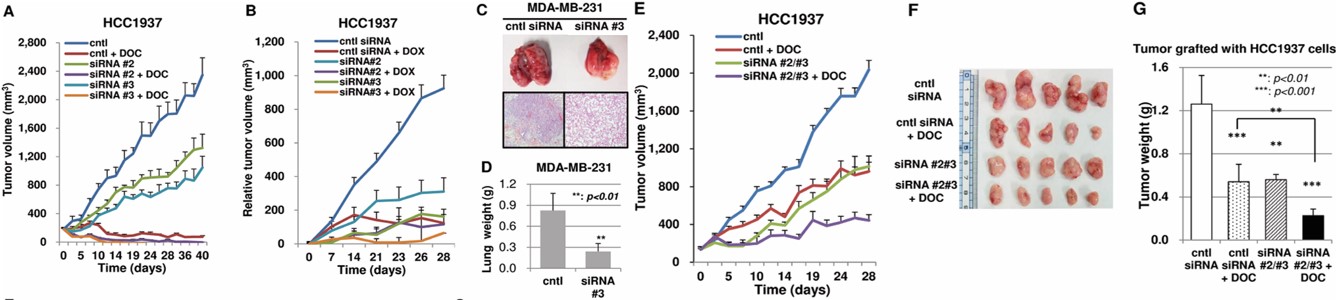 Fig.2 PRDM14-targeted siRNA demonstrates successful local and systemic delivery, with safe and synergistic combination efficacy in breast cancer.2
Fig.2 PRDM14-targeted siRNA demonstrates successful local and systemic delivery, with safe and synergistic combination efficacy in breast cancer.2
In the nude mouse HCC1937 orthotopic breast cancer model, RNAi targeting PRDM14 successfully inhibited tumor growth following both local (intratumoral siRNA/PEI) and systemic (intravenous chimeric RNAi/CaP micelles) delivery. Combination with Docetaxel showed safe and synergistic enhancement, whereas the combination with Doxorubicin highlighted safety risks, confirming the RNAi delivery effectiveness and guiding rational combination therapy design.
References
- Zheng, Yiyang et al. "The role of mRNA in the development, diagnosis, treatment and prognosis of neural tumors." Molecular cancer vol. 20,1 49. https://doi.org/10.1186/s12943-021-01341-7. Distributed under Open Access license CC BY 4.0, without modification.
- Taniguchi, Hiroaki et al. "Silencing PRDM14 expression by an innovative RNAi therapy inhibits stemness, tumorigenicity, and metastasis of breast cancer." Oncotarget vol. 8,29 (2017): 46856-46874. https://doi.org/10.18632/oncotarget.16776. Distributed under Open Access license CC BY 4.0, with modification.
For Research Use Only.
