CD19⁺ Humanized Patient derived Xenograft (PDX) Model based CAR-T Efficacy Modeling & Pharmacodynamics Service
Introduction
A CD19 Positive Patient derived Xenograft (PDX) in a Humanized Mouse Model represents the pinnacle of clinically relevant preclinical platforms for CAR-T validation. Creative Biolabs has mastered the integration of the patient's unique tumor complexity (PDX) with a functional human immune system (Humanized Mouse Model). This powerful combination allows researchers to assess the efficacy and safety of human CAR-T cells against a patient derived malignancy under near-clinical conditions. This platform is essential for accelerating drug development and securing IND approval by providing comprehensive data on CAR-T cell persistence and clinical efficacy, predicting human-specific toxicities like cytokine release syndrome (CRS), and offering crucial translational insight into resistance mechanisms for optimizing next-generation constructs.
Available CD19 Positive PDX in Humanized Mouse Model for CAR-T Efficacy Service
CD19 Positive PDX models represent the most predictive platform for preclinical CAR-T evaluation. Sourced directly from human B-cell malignancies, they faithfully retain the original tumor's genetic complexity, signaling pathways, and microenvironment characteristics.
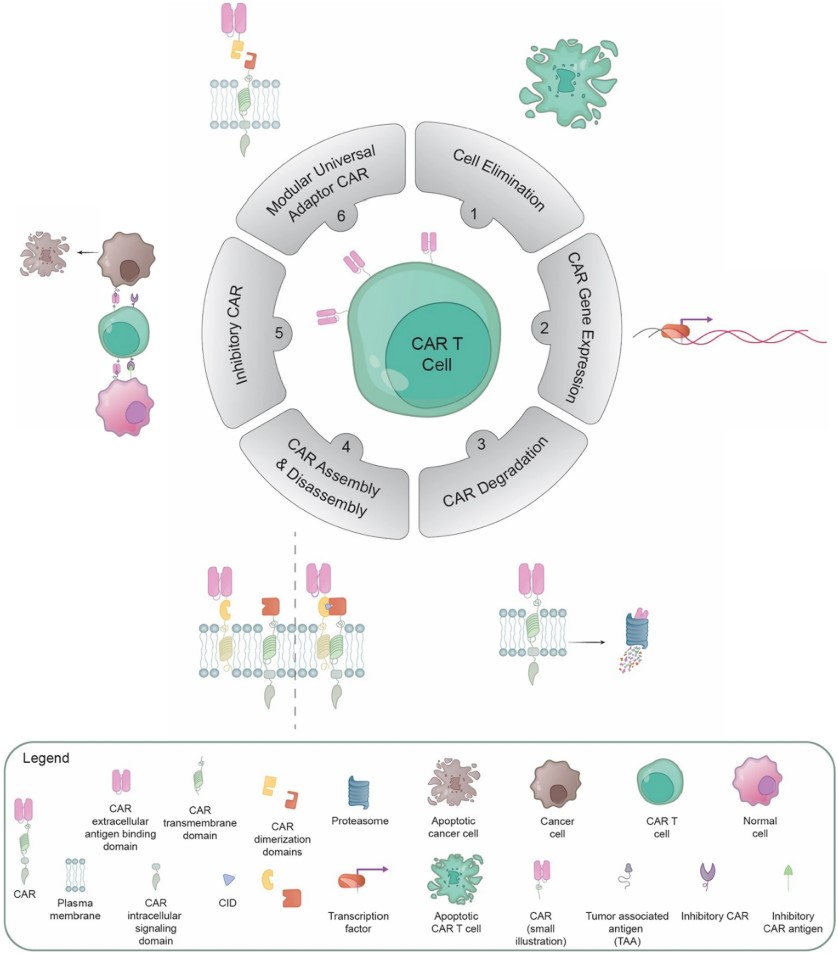 Fig.1 Schematic overview of the various approaches developed to modulate CAR T cell activities.1,4
Fig.1 Schematic overview of the various approaches developed to modulate CAR T cell activities.1,4
| Clinical Category | Model Type | Model Characteristics | Key Evaluation Values |
| Acute Lymphoblastic Leukemia (ALL) | B-ALL PDX | Systemic disease modeling: originates from b-all blasts, establishing systemic disease in the bone marrow and peripheral blood of humanized mice. | Gold standard for systemic CAR-T efficacy and minimal residual disease (MRD) assessment, crucial for studying relapse mechanisms like antigen escape (CD19 loss). |
| B Cell Non-Hodgkin Lymphoma (B-NHL) | DLBCL PDX | Aggressive solid tumor modeling: establishes aggressive solid tumor masses in soft tissue/nodes. | Evaluating solid tumor penetration and CAR-T trafficking. Key for testing strategies to mitigate CAR-T exhaustion within the complex TME. |
| Solid/Nodal Disease | FL PDX | Indolent lymphoma modeling: derived from indolent follicular lymphoma tissue. | Investigating long-term CAR-T persistence and the mechanisms driving transformation to aggressive NHL, as well as efficacy against low-proliferative disease. |
| Special CD19 Malignancies | MM PDX | Bone marrow niche modeling: multiple myeloma cells (low CD19) established in the bone marrow. | Developing CAR-T therapies that overcome the protective bone marrow microenvironment and validating constructs targeting specific multiple myeloma antigens (e.g., BCMA, CD38). |
Evaluation Platform
Creative Biolabs leverages patient PDX heterogeneity and humanized systems to generate the most predictive CAR-T data for clinical translation. By employing world-class precision instruments and scientifically robust detection processes, we ensure the delivery of high-fidelity results to fully support and expedite your research and development decisions.
-
Core Efficacy & CAR-T Dynamics Tracking
- Tumor Clearance & Survival: Non-invasive Bioluminescence Imaging (BLI) is used for real-time tracking of tumor burden and analysis of overall survival (OS), quantifying CAR-T clearance of complex PDX tumors. PET-CT Imaging provides a more precise evaluation of a tumor's location and size, particularly for deep or disseminated tumors.
- CAR-T Persistence: High-precision ddPCR performs absolute quantification of CAR gene copy numbers in tissues, accurately tracking CAR-T expansion and long-term persistence.
- Infiltration & Function: Multiplex immunofluorescence (mIF) provides deep spatial analysis of CAR-T density and functional status within heterogeneous PDX solid tumors.
- Critical Safety & Toxicity Signals Analysis
- CRS Risk Proxy: High-throughput Luminex/MSD monitor human cytokines (e.g., IL-6, IFN-γ), providing a critical safety signal predictive of CRS severity.
- B Cell Aplasia: Multi-Color FACS tracks normal human B cell clearance in blood and marrow, assessing on-target/off-tumor toxicity and CAR-T durability.
-
Mechanism & Resistance Exploration
- Antigen Loss Analysis: Post-treatment analysis validates the loss or down-modulation of CD19 antigen expression in residual PDX cells, directly reflecting clinical resistance mechanisms.
- T Cell Exhaustion & Profiling: We analyze CAR-T exhaustion markers (e.g., PD-1, LAG-3) and use RT-qPCR or RNA-Seq to identify resistance pathways, guiding next-generation CAR-T optimization.
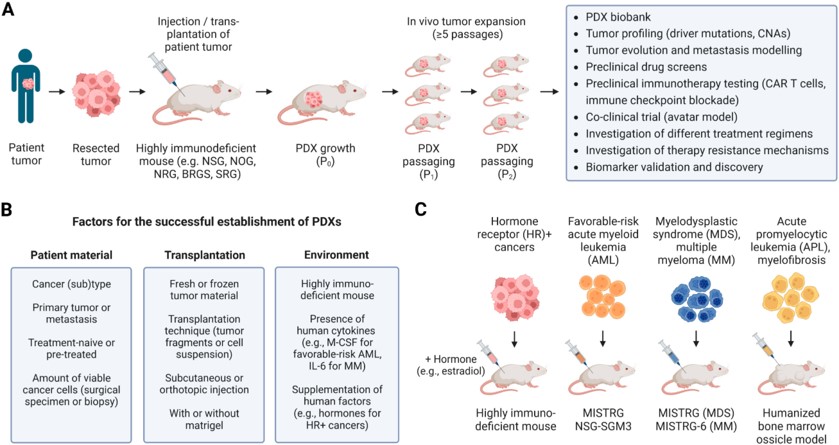 Fig.2 Modeling patient derived tumors in immunodeficient mice.2,4
Fig.2 Modeling patient derived tumors in immunodeficient mice.2,4
Applications
- Overcoming CDX Limitations: By retaining the patient tumor's genetic heterogeneity and complex TME features, the PDX model enables the assessment of CAR-T clearance of MRD and its penetration and functional persistence against solid tumors (e.g., DLBCL).
- Precise Resistance Modeling: We use PDX heterogeneity to accurately monitor CD19 antigen loss and molecular pathway activation (e.g., RNA-Seq) under therapeutic pressure. This capability provides essential mechanistic insight to guide next-generation construct design.
- Functional Exhaustion Study: Analysis of CAR-T functional exhaustion (e.g., PD-1, LAG-3) guides the rational design of combination strategies (e.g., with ICIs) and the development of more persistent CAR structures.
- Optimized Safety Assessment: In humanized PDX models, we monitor human cytokines (e.g., IL-6, IFN-γ) as a reliable CRS risk proxy and track normal B cell clearance to assess CD19's on-target/off-tumor toxicity, delivering critical safety data required for IND submission.
- Supporting Next-Gen Design: PDX models are utilized to validate the superiority of dual/multi-targeted CAR-T strategies (e.g., CD19/BCMA) against heterogeneous tumors and to screen novel CAR constructs for enhanced persistence and reduced exhaustion features.
Our Advantages
- Advanced and Experienced Humanized Mouse Platform: We have mature technology for building a humanized mouse model that reliably reconstitutes the human immune system. This ensures the mice develop sufficient numbers and activity of human T cells, B cells, and other immune components, which accurately reflects CAR-T cell behavior in a human body. We also have a robust PDX model library with well-characterized CD19-positive B-cell lymphoma or leukemia models, providing a range of options with clear genetic and molecular backgrounds for various research needs.
- Comprehensive Efficacy and Safety Assessment Capabilities: Advanced CAR-T evaluation integrates in vivo BLI and FACS qPCR for precise tracking of persistence and tumor clearance, alongside dedicated safety monitoring using cytokine assays and histopathology to predict CRS and other adverse effects.
- Strong Project Management and Data Handling: We provide dedicated project managers to ensure smooth experimental execution and timely data updates, maintaining transparent and efficient communication with the client. We deliver high-quality, reproducible data with professional statistical analysis and clear, concise final reports.
- One-stop service: In addition to our comprehensive tumor model services, we offer a full suite of preclinical oncology solutions, including dedicated teams for pharmacokinetics and toxicology assessment.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q. What is a CD19-Positive PDX Humanized Mouse Model?
A: This is a preclinical model established by implanting CD19-positive, patient derived xenograft (PDX) tumor tissue into mice engrafted with a humanized immune system (HIS). The model is designed to simultaneously simulate the patient's tumor characteristics, including its heterogeneity and microenvironment, as well as a human immune environment.
-
Q. What are the advantages of this model compared to traditional models (CDX or standard PDX)?
A: Traditional preclinical models like CDX, despite being low-cost, are limited by their lack of tumor heterogeneity and human immune components. While Standard PDX preserves tumor heterogeneity, it still lacks a human immune system. The CD19-Positive PDX Humanized Model is the most advanced platform because it integrates both the patient's tumor and a functional human immune system, making it essential for accurately evaluating immune-mediated effects, monitoring critical toxicities like CRS, and modeling clinical resistance mechanisms.
-
Q. What is the workflow for constructing the model?
A: The CD19 Positive PDX Humanized Model is established by transplanting patient derived CD19+ tumors into immunodeficient mice reconstituted with a human immune system (via CD34+ or PBMC engraftment), enabling the comprehensive evaluation of CAR-T efficacy (tumor regression), persistence, immune response (e.g., cytokine levels, immune cell activation), and toxicity (e.g., CRS-associated weight loss, inflammatory cytokine release) under clinically relevant conditions.
-
Q. In which scenarios is this model primarily applied?
A: The CD19-targeted CAR-T research platform is utilized for comprehensive preclinical assessment, focusing on testing clearance capability against B-cell malignancies, predicting and mitigating clinical toxicities (like CRS), analyzing tumor escape mechanisms, and developing synergistic combination therapies.
-
Q. What are the limitations of the model?
A: The limitations of humanized mouse models include the inherent challenge of the incomplete immune system, where murine non-hematopoietic tissues affect immune signaling, the high technical complexity due to the long and costly construction period requiring specialized facilities, and discrepancies in toxicity prediction, as human-specific toxicities like high-level IL-6-mediated CRS may not be fully replicated.
-
Q. Are there alternative models available?
A: While syngeneic mouse models are suitable for studying immunological mechanisms due to their murine-origin tumors and immune systems, they cannot directly test human CAR-T therapies; conversely, non-human primates (NHPs) Models, though highly similar to humans and primarily used for late-stage safety verification, are extremely expensive and subject to significant ethical restrictions.
Published Data
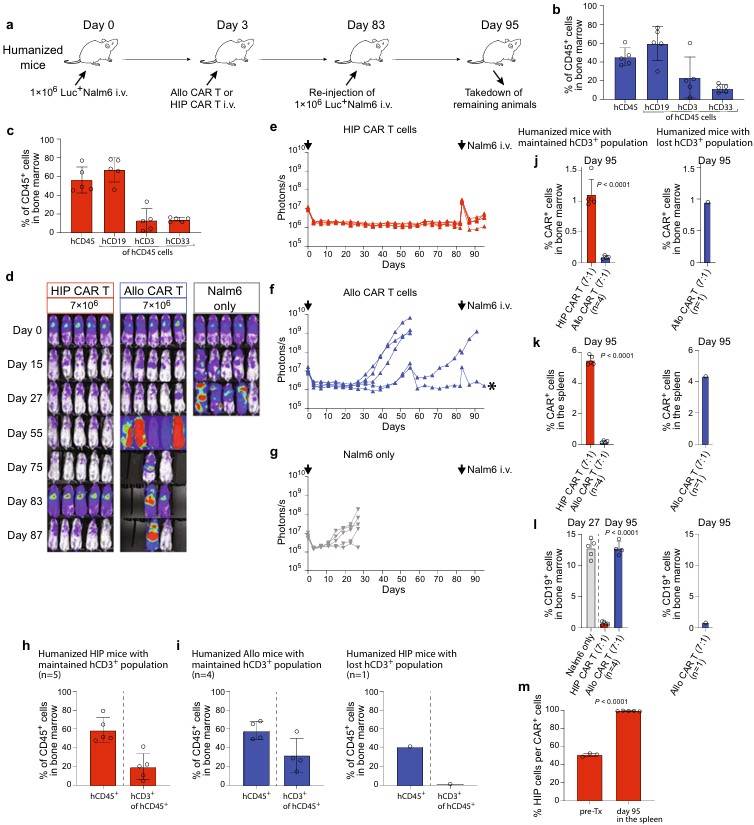 Fig.3 HIP CAR T cells quickly clear tumor cells re-injected after 83 days in fully immunocompetent allogeneic humanized mice.3,4
Fig.3 HIP CAR T cells quickly clear tumor cells re-injected after 83 days in fully immunocompetent allogeneic humanized mice.3,4
A comparison between the humanized NSG-SGM3 mouse leukemia model and the standard NSG mouse leukemia model demonstrates that hypoimmunogenic (HIP) CD19 CAR-T cells exhibit greater persistence and faster tumor regression. Furthermore, a CD47-targeted fusion protein can specifically eliminate these HIP CAR-T cells, confirming the model's potential for evaluating CD19-positive CAR therapies.
References
- Celichowski, Piotr et al. "Tuning CARs: recent advances in modulating chimeric antigen receptor (CAR) T cell activity for improved safety, efficacy, and flexibility." Journal of translational medicine vol. 21,1 197. https://doi.org/10.1186/s12967-023-04041-6
- Chen, Anna et al. "Modeling the Tumor Microenvironment and Cancer Immunotherapy in Next-Generation Humanized Mice." Cancers vol. 15,11 2989. https://doi.org/10.3390/cancers15112989
- Hu, Xiaomeng et al. "Hypoimmune anti-CD19 chimeric antigen receptor T cells provide lasting tumor control in fully immunocompetent allogeneic humanized mice." Nature communications vol. 14,1 2020. https://doi.org/10.1038/s41467-023-37785-2
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
