Testosterone induced Benign Prostatic Hyperplasia Modeling & Pharmacodynamics Service
Creative Biolabs offers a range of well-established animal models to evaluate drug efficacy for Benign Prostatic Hyperplasia (BPH). These models help assess the effectiveness of various treatments, including hormone-based therapies and pharmaceutical agents targeting prostate growth and urinary symptoms.
Introduction
Benign Prostatic Hyperplasia (BPH) is a common condition in aging men characterized by the non-cancerous enlargement of the prostate gland. As the prostate enlarges, it can cause urinary symptoms such as frequent urination, nocturia (waking up at night to urinate), weak urine flow, and incomplete bladder emptying. These symptoms arise due to the prostate's compression of the urethra, obstructing urine flow. The exact cause of BPH is not entirely understood, but it is believed to be influenced by hormonal changes associated with aging, particularly the balance between testosterone and estrogen. BPH is common in men over 50, with symptoms becoming more severe as men age. If left untreated, BPH can lead to complications like urinary retention, bladder stones, or kidney damage. Treatment options include medications like alpha-blockers and 5-alpha reductase inhibitors, or surgical interventions in more severe cases. The condition is not cancerous, but its symptoms can significantly impact quality of life.
Testosterone-Induced Benign Prostatic Hyperplasia Model and Applications
The Testosterone-Induced Benign Prostatic Hyperplasia Model involves the administration of testosterone to rodents, typically rats or mice, to induce BPH-like symptoms. This model is characterized by prostate enlargement and histopathological changes such as glandular hyperplasia and stromal growth. The process typically includes the subcutaneous or intramuscular injection of testosterone for several weeks, leading to the development of symptoms resembling human BPH. One of the main advantages of this model is its ability to replicate the key features of human prostate enlargement, making it ideal for studying the pathophysiology of BPH and testing pharmacological agents. However, it is also limited by the fact that it may not fully mimic all aspects of the disease, particularly in terms of age-related progression.
- Simulates: This model simulates benign prostatic hyperplasia, particularly the androgen-driven enlargement of the prostate.
- Evaluates Drugs: It is used to evaluate drugs that target prostate enlargement, urinary dysfunction, or hormonal regulation, including androgen receptor antagonists and 5-alpha reductase inhibitors.
Evaluation Platform
- Animals: Mouse, Rat, Hamster, Rabbit, Cat, Dog, NHPs.
-
Measurements
For evaluating drug efficacy in the Testosterone-Induced Benign Prostatic Hyperplasia Model, we employ various advanced techniques, such as:- General Observations: Changes in body weight, urinary patterns, and overall animal health.
- Histological Examination: Prostate tissue analysis via H&E staining to assess glandular hyperplasia and stromal changes.
- Biomarker Analysis: Measurement of serum testosterone, DHT levels, and other hormone markers.
- Immunohistochemistry: Evaluation of androgen receptor expression in prostate tissue.
- Cytokine Profiling: Detection of inflammation markers such as TNF-α, IL-6, and IL-1β in prostate tissue.
- Gene and Protein Expression Analysis: Using RT-qPCR and Western blot to assess the expression of genes and proteins involved in prostate growth and inflammation.
Additionally, our experienced team helps with study design, model selection, and data analysis to ensure precise results and tailored approaches.
Our advantages
- Expertise: We bring years of specialized experience in developing and optimizing animal models for drug efficacy testing across various therapeutic areas, including BPH.
- Customized Solutions: Our team works closely with clients to tailor research models and protocols that best suit specific project needs.
- Advanced Technology: We utilize cutting-edge technologies and techniques, including histology, immunohistochemistry, gene expression profiling, and cytokine analysis, ensuring precise and reliable results.
- Comprehensive Support: From experimental design to data analysis, we provide end-to-end support, guiding you at every step of the research process.
- High-Quality Models: Our models are validated, reproducible, and offer a reliable foundation for evaluating new therapies.
- Commitment to Innovation: We continuously innovate and improve our models to meet the evolving needs of the research community.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
1. What is the Testosterone-Induced Benign Prostatic Hyperplasia Model?
This model simulates the enlargement of the prostate induced by testosterone in rodents, making it ideal for studying prostate growth and testing potential therapies for BPH.
-
2. How long does it take to establish the model?
The model typically takes 3-4 weeks to induce prostate enlargement following testosterone administration, though the timeline may vary depending on the specific protocol used.
-
3. What kind of analysis can be performed using this model?
We offer various analyses, including histopathological evaluation, biomarker testing, immunohistochemistry for androgen receptors, and gene/protein expression profiling, among others.
-
4. Can you help design the experiment and select the right model?
Yes, our team can assist with experimental design, model selection, and data analysis to ensure the approach aligns with your research objectives.
-
5. What are the advantages of using animal models for BPH research?
Animal models like the Testosterone-Induced BPH Model offer a controlled environment to study disease mechanisms, test therapies, and evaluate drug safety and efficacy before clinical trials.
Published Data
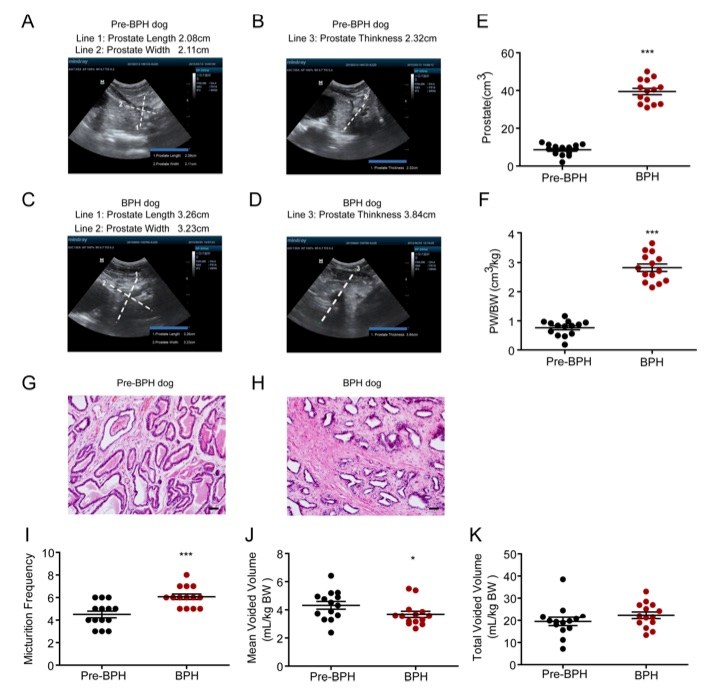 Fig. 1 Voiding behavior in testosterone-induced BPH dogs.1
Fig. 1 Voiding behavior in testosterone-induced BPH dogs.1
To establish a BPH dog model, young adult male Beagle dogs (1 year old, 9–13 kg) were castrated and then subcutaneously injected with testosterone propionate (10 mg/kg per day) for a duration of 8 weeks. Prostate volume was measured using an ultrasonic diagnostic instrument both prior to castration (pre-BPH group, Fig. 1A and 1B) and 8 weeks after testosterone administration (BPH group, Fig. 1C and 1D). Prostate size was calculated using the formula: volume = length × width × thickness, based on measurements obtained from the ultrasound. The results showed that testosterone propionate treatment significantly increased both the prostate volume and the prostate volume-to-body weight ratio (Fig. 1E and 1F). Histological analysis revealed that testosterone treatment induced hyperplasia in the stromal tissue of the prostate, which is composed of collagen and smooth muscle, but not in the epithelial tissue (Fig. 1G and 1H). Additionally, micturition behaviors were assessed before and after BPH onset. Like the testosterone-induced BPH rat model, the BPH dogs exhibited an increased micturition frequency (Fig. 1G) and a decreased mean voided volume (Fig. 1H) compared to the pre-BPH group. However, no significant differences were observed in the total voided volume between the BPH and pre-BPH groups (Fig. 1I).
Reference
- Li, Jing et al. "Testosterone-induced benign prostatic hyperplasia rat and dog as facile models to assess drugs targeting lower urinary tract symptoms." PloS one vol. 13,1 e0191469. 19 Jan. 2018. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1371/journal.pone.0191469
For Research Use Only.
