iPSC based Efficacy Modeling & Pharmacodynamics Service
Introduction
Induced pluripotent stem cells (iPSCs) are derived by genetically reprogramming mature somatic cells to achieve a pluripotent state comparable to embryonic stem cells. This grants them the ability to differentiate into many different cell types. The technology possesses extensive potential for applications, particularly in the areas of regenerative medicine, disease modeling, drug screening, and tissue engineering. Creative Biolabs' iPSC Disease Modeling Platform offers a powerful solution for regenerative medicine and drug development by leveraging the process of genetically reprogramming mature somatic cells back to a pluripotent state. The core strength of this platform lies in its high level of integration and technical sophistication: we utilize the inherent advantages of iPSCs, including their near-infinite proliferative capacity and ease of genetic engineering, to ensure a stable, high-quality cell supply. Crucially, this is combined with our extensive experience in stem cell culture, translating into a stable iPSC induction system, precise gene-editing capabilities, and integration with highly biofidelic advanced humanized mouse models. This unique integration allows us to construct highly predictive oncology models, facilitating the accurate evaluation of immuno-oncology drug efficacy and accelerating the safe and efficient transition of your innovative therapies from the lab to the clinic.
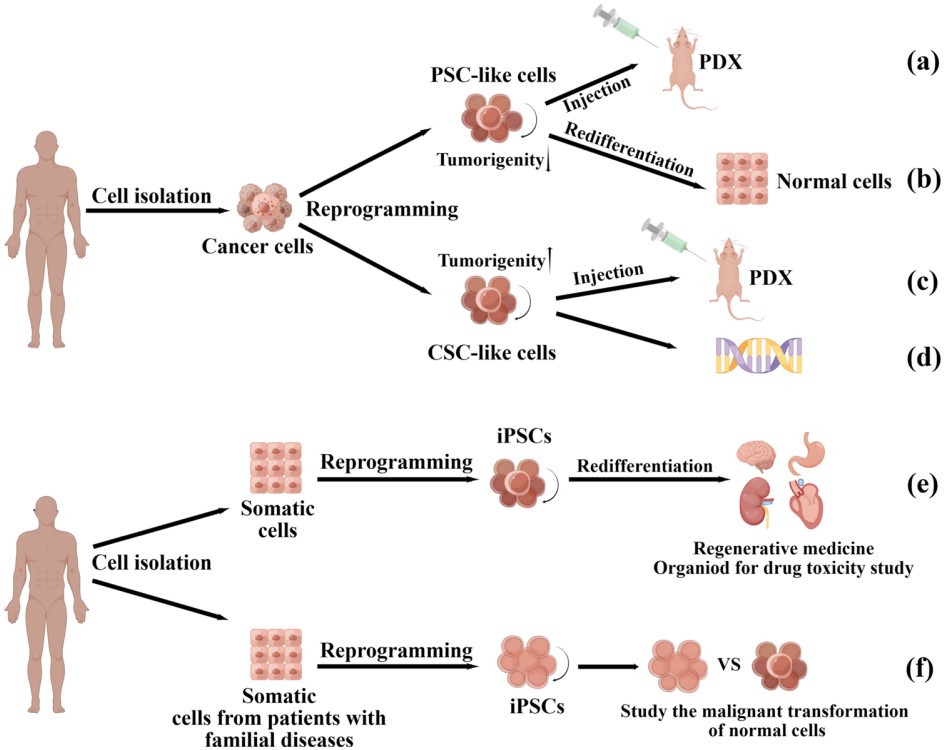 Fig.1 The applications of cancer cell and somatic cell reprogramming.1,5
Fig.1 The applications of cancer cell and somatic cell reprogramming.1,5
Available iPSC based Efficacy Services
In vitro Services
Our platform ensures the highest quality and versatility by utilizing diverse sources of somatic cells, ranging from healthy donors to patient cohorts with specific diseases, while actively creating advanced disease models and isogenic controls through precise CRISPR/Cas9 gene editing. Crucially, every single iPSC line we develop undergoes rigorous quality control (QC), including stringent assays that validate pluripotency, karyotypic stability, and purity, thereby guaranteeing the standardization and superior reproducibility essential for all downstream therapeutic and research applications.
| Cell Sourcing & iPSC Reprogramming | Features and advantages |
 iPSC Reprogramming Service |
Flexible Starting Cells: Simply provide your fibroblasts or PBMCs. Footprint-Free Technology: We utilize non-integrating episomal vectors or Sendai virus for safe and reliable reprogramming. Comprehensive Validation: Each line undergoes rigorous, multi-dimensional quality control to verify pluripotency and stability. Guaranteed Clones We deliver 2 fully characterized and ready-to-use iPSC clones. Rapid Turnaround: Project completion in as fast as 10-14 weeks. |
 Custom iPSC Gene Editing Services |
Multiple Editing Types: We expertly handle a full range of edits, from complete knockouts to precise knock-ins and point mutations. Professional Gene Editing Platform: Our team utilizes advanced CRISPR/Cas9 technology to ensure high-precision results. High-Efficiency Monoclonal Screening: We employ robust methods to rapidly isolate pure, single-cell derived clones. Comprehensive Deliverables: You receive 2 fully validated edited clones plus the original wild-type parental line for your experiments. Industry-Leading Speed Complete your gene knockout project in as fast as 8 weeks. |
In vivo model Services
Our advanced in vivo model platform serves as the core accelerator for the clinical translation of iPSC therapies. These models provide highly accurate personalized efficacy validation for iPSC derived cells via advanced biomimetic environments. Crucially, they act as the critical hub for assessing the immunogenicity and rejection of universal (allogeneic) products, ensuring long-term safety. Furthermore, the platform strictly enforces the regulatory-mandated teratoma formation gold standard test, providing the most reliable safety clearance for your iPSC products entering clinical trials.
| iPSC derived Cell Transplantation Models | Modeling & Application Value |
| PDX & iPSC Combination Models | Patient tumor tissue is surgically implanted, and iPSC derived therapeutic cells (e.g., CAR-NK cells, tumor-infiltrating lymphocytes (TILs)) are co-transplanted to model a personalized treatment environment. High Fidelity and Personalization. Used to assess the efficacy of iPSC derived cell therapies against a patient's individual tumor. |
| Genetically Humanized Mouse Models | Immunodeficient mice are injected with human CD34+ HSCs to reconstitute a functional human immune system or are gene-edited to express human targets. Translational Relevance and Immune Response Assessment. Essential for monitoring immunogenicity and rejection of allogeneic iPSC derivatives (e.g., cardiomyocytes, neurons) in a human immune context. |
| Immunodeficient Mouse Models | iPSC derived products (e.g., neural progenitors, retinal pigment epithelial cells (RPEs)) are directly injected into the recipient mouse (e.g., NSG) for in vivo monitoring. Fundamental Safety and Regulatory Compliance. The mandatory safety check to confirm that iPSC derivatives do not form teratomas before human clinical trials. |
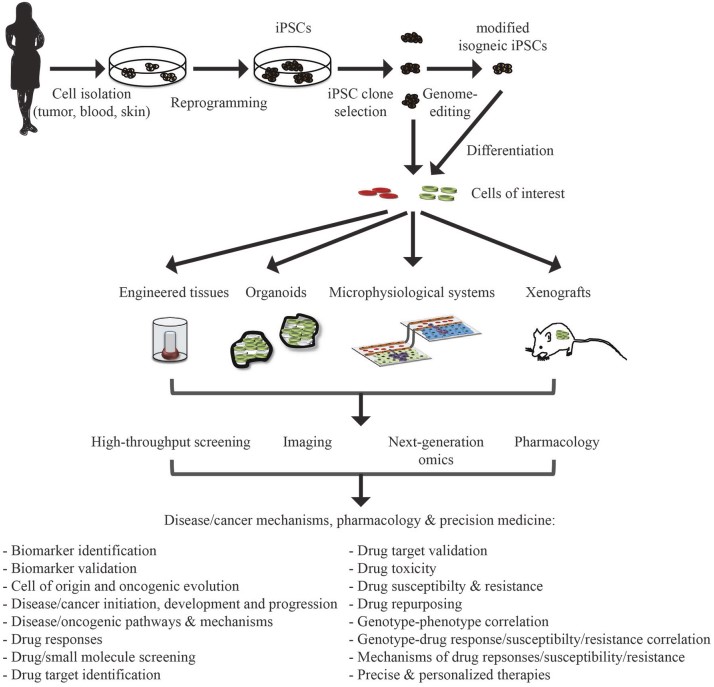 Fig.2 Application of iPSC in disease or cancer modeling, pharmacology, and precision medicine.2,5
Fig.2 Application of iPSC in disease or cancer modeling, pharmacology, and precision medicine.2,5
Evaluation Platform
Ensuring the safety, efficacy, and functionality of iPSC derived products is paramount to achieving clinical success. Our iPSC therapy evaluation and translation platform integrates the world's most cutting-edge biomedical technologies and stringent quality control systems, designed to provide your iPSC products with comprehensive, accurate, and highly reliable preclinical assessment services.
- High-Content Screening (HCS) System: Employs automated fluorescence microscopy for imaging and analysis, enabling quantitative, high-throughput assessment of cell morphology and molecular markers.
- Electrophysiology Systems: Uses multi-electrode arrays (MEA) or patch-clamp techniques to precisely monitor the functional metrics (action potentials, ion channel activity) of iPSC derived cardiomyocytes or neurons.
- Flow Cytometry (FCM): Used for analyzing the surface markers and intracellular proteins of iPSC derived cells, quickly identifying purity and differentiation status.
- In Vivo Imaging System (IVIS): Uses fluorescence/bioluminescence markers for non-invasive, longitudinal, real-time tracking of transplanted cell homing and survival.
- High-Resolution Ultrasound/MRI: Used to assess the recovery of cardiac function or brain structure after transplanting iPSC derived cells.
- Behavioral Analysis Systems: Used to quantify the recovery of motor function and cognitive function after neural cell transplantation.
- Histopathology and Immunohistochemistry (IHC): Provides the highest standard for safety checks (e.g., teratoma detection) and analysis of cell integration.
Applications
- Oncology Modeling & Mechanistic Studies: Integrating patient-specific iPSC differentiation and gene editing, often combined with 3D organoid culture and PDX models, we comprehensively investigate mechanisms of tumorigenesis, the impact of genetic mutations, the TME, and differential drug responses.
- Drug Screening & Toxicity Assessment: Using iPSC derived cancer cells, organoids, and iPSC derived cell transplantation models to accelerate iPSCs drug development via high-throughput screening (HTS), drug repurposing, and evaluating combination therapies, alongside essential toxicity and safety testing.
- Immunotherapy & Cell Therapy: Leveraging large-scale iPSC differentiation and gene editing technology, the iPSC based efficacy services platform is highly capable of developing and enhancing next-generation immune cell therapies (e.g., CAR-T, CAR-NK, CAR-Mac), specifically targeting critical functions such as the ADCC effect and persistence.
- Disease Mechanisms & Translational Research: Leveraging the reprogramming of cancer cells into cancer-iPSCs and the subsequent differentiation of iPSCs into Cancer Stem Cells (CSCs), along with studying shared molecular signatures that maintain stemness (e.g., OCT4, SOX2, c-MYC), we investigate the origins of CSCs, tumor cell plasticity, hereditary cancer mechanisms, and drug resistance.
Our Advantages
- Specialized Expertise and Experience: Strong expertise and extensive experience in iPSCs technology and oncology research, particularly in the generation and handling of iPSCs derived animal models.
- Diverse Animal Model Library: Availability of a wide range of animal models suitable for cancer research, including xenograft models, humanized mouse models, and genetically engineered mouse models. Ability to custom-develop specific animal models based on research requirements.
- Advanced Laboratory Facilities: Equipped with state-of-the-art laboratory facilities supporting complex operations such as iPSC culture, differentiation, and gene editing. Capability for high-throughput screening and advanced imaging technologies to monitor tumor growth and drug distribution.
- Efficient Project Management: A professional project management team ensures efficient project completion, with the ability to adjust experimental designs according to client needs and deliver results on schedule. Regular communication with clients, providing transparent project progress reports.
- Data Analysis and Management Capabilities: Possession of advanced data analysis platforms capable of handling and interpreting complex datasets. Provision of detailed analytical reports, elucidating key results such as drug efficacy and biomarker changes.
- Collaboration and Flexibility: Keeps close collaboration with clients, effectively responding to changes and challenges during research. Offers personalized services and recommendations, supporting research needs at different stages.
- Integrated PK/PD Analysis: Our services include biodistribution tracking of PSC derived cells or drugs and the establishment of quantitative exposure-response (PK/PD) relationships to scientifically inform clinical dosing strategies.
- One-Stop Solution: Our one-stop service spans from iPSC generation, gene editing, and in vitro differentiation to in vivo efficacy and safety evaluation. This greatly minimizes vendor management costs and administrative burden for our clients, accelerating the development process.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: Why choose iPSC technology to build tumor animal models? What are its advantages over traditional cell line models?
A: iPSC technology offers unparalleled capabilities for personalized cancer modeling and physiological relevance. iPSCs, reprogrammed directly from a patient's somatic cells, retain the unique genomic background essential for accurately simulating tumor heterogeneity. They possess unlimited expansion and differentiation potential, allowing for the construction of cancer models from virtually any cell type (e.g., neurons, epithelial, and immune cells), overcoming the limitations of primary tumor cells. Furthermore, iPSCs are highly amenable to precise gene editing (e.g., using CRISPR/Cas9) to introduce specific driver mutations (e.g., TP53 or KRAS) or to create isogenic controls. Crucially, this technology allows for modeling the entire process of tumorigenesis, enabling the dynamic study of cancer progression from the initial accumulation of mutations to malignant transformation by introducing oncogenic mutations into "normal" differentiated iPSC derived cells.
-
Q: What is the biggest challenge of iPSC models? How can the experimental success rate be ensured?
A: The core challenges in iPSC technology revolve around stringent QC and efficient differentiation. The risk of teratoma formation from residual undifferentiated cells necessitates stringent QC before transplantation, involving assays like flow cytometry (confirming negative pluripotency marker expression, e.g., SSEA-4, OCT-4), karyotype analysis, and embryoid body formation tests to ensure genomic stability and purity. Furthermore, to overcome the issue of low cell yield caused by inefficient conversion, it is essential to employ optimized, highly efficient directed differentiation protocols and confirm the identity and purity of the resulting target cells using functional marker identification methods.
-
Q: Can iPSC models be used to evaluate cancer immunotherapy?
A: Yes, iPSC models have enormous potential for evaluating cancer immunotherapies.
This type of model can simulate the human immune environment, allowing for the assessment of the in vivo persistence and anti-tumor efficacy of iPSC derived immune cells, as well as potential toxic side effects (such as cytokine release syndrome).
-
Q: What is the typical timeline and cost for a project?
A: Timelines and costs are project-specific, depending on factors like the iPSC line source (existing vs. de novo), the complexity of the differentiation protocol, and the scope of the efficacy endpoints. A standard project typically ranges from 3 to 6 months.
-
Q: Can we provide our own patient samples for model development?
A: Absolutely. We specialize in creating custom, patient derived models. This "patient avatar" approach is at the heart of precision medicine, allowing you to test therapeutic hypotheses on the specific genetic backgrounds you are targeting in the clinic.
Published Data
Case 1:
HPV16 E6-rejTs (derivation of iPSCs by reprogramming) demonstrated potent anti-tumor activity in a NOG cervical cancer mouse model, significantly inhibiting tumor growth, reducing tumor burden, and prolonging survival. The cells maintained chromosomal stability throughout the entire process (CTL -> iPSC -> rejT), as confirmed by CGH analysis, validating their dual-target efficacy and safety.
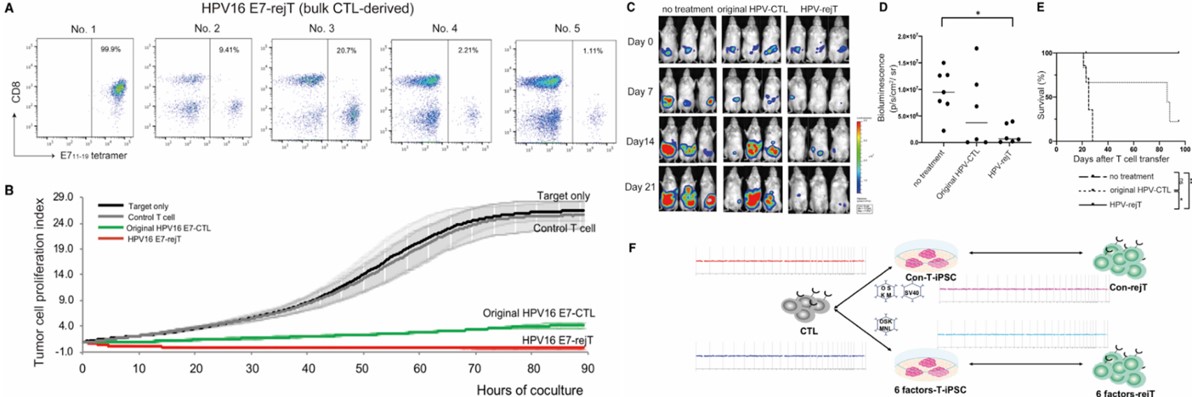 Fig.3 Tumorigenic assessment of iPSC products and in vivo efficacy validation.3
Fig.3 Tumorigenic assessment of iPSC products and in vivo efficacy validation.3
Case 2:
Through intrahepatic orthotopic transplantation experiments, it was demonstrated that both miPS-Huh7cm cells and primary miPS-Huh7cmP1 cells can form tumors with a malignant phenotype in the livers of immunodeficient mice.
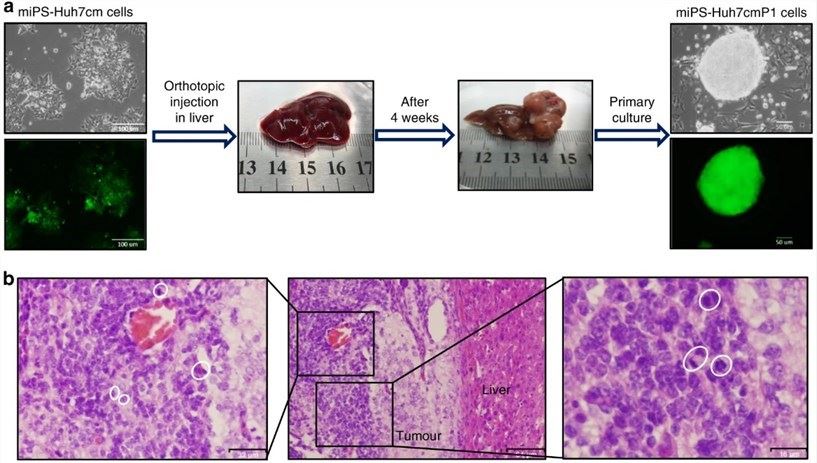 Fig.4 Representative scheme of the orthotopic injection of miPS-Huh7cm cells (miPS treated with Huh7cm for 4 weeks) into the liver and the formed tumor after 4 weeks of injection. Scale bars represent 100 and 50 µm.4,5
Fig.4 Representative scheme of the orthotopic injection of miPS-Huh7cm cells (miPS treated with Huh7cm for 4 weeks) into the liver and the formed tumor after 4 weeks of injection. Scale bars represent 100 and 50 µm.4,5
References
- He, Rong et al. "Application of Induced Pluripotent Stem Cells in Malignant Solid Tumors." Stem cell reviews and reports vol. 19,8 (2023): 2557-2575. https://doi.org/10.1007/s12015-023-10633-y.
- Kim, Huensuk, and Christoph Schaniel. "Modeling Hematological Diseases and Cancer With Patient-Specific Induced Pluripotent Stem Cells." Frontiers in immunology vol. 9 2243. https://doi.org/10.3389/fimmu.2018.02243.
- Honda, Tadahiro et al. "Sustainable Tumor-Suppressive Effect of iPSC derived Rejuvenated T Cells Targeting Cervical Cancers." Molecular therapy: the journal of the American Society of Gene Therapy vol. 28,11 (2020): 2394-2405. https://doi.org/10.1016/j.ymthe.2020.07.004. Distributed under Open Access license CC BY 4.0, with modification.
- Afify, Said M et al. "A novel model of liver cancer stem cells developed from induced pluripotent stem cells." British journal of cancer vol. 122,9 (2020): 1378-1390. https://doi.org/10.1038/s41416-020-0792-z.
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
