Immune Checkpoint Inhibitor based Efficacy Modeling & Pharmacodynamics Service
Introduction
Immune Checkpoint Inhibitors (ICIs) have revolutionized tumor immunotherapy by restoring the immune system's ability to eliminate cancer cells, fundamentally altering the treatment landscape for various malignancies, yet only 15%-30% of patients respond, and concerns remain regarding toxicity risks and high costs. Efficacy modeling and pharmacodynamics services offer a comprehensive solution for the precise application of ICIs by integrating mathematical models, biomarkers, and clinical data. Creative Biolabs leverages deep, unique insights into the pharmacological efficacy, toxicology (e.g., immune-related adverse events (irAEs)), and pharmacokinetics (PK/PD) of ICIs, coupled with extensive experience in tumor model establishment, to accelerate your research and development pipeline. By strategically matching the animal model and the pharmacodynamic readout to the specific checkpoint pathway (Inhibitory vs. Co-stimulatory), Creative Biolabs is uniquely positioned to assist in the precise mechanism research and successful clinical translation of your next-generation ICI candidates.
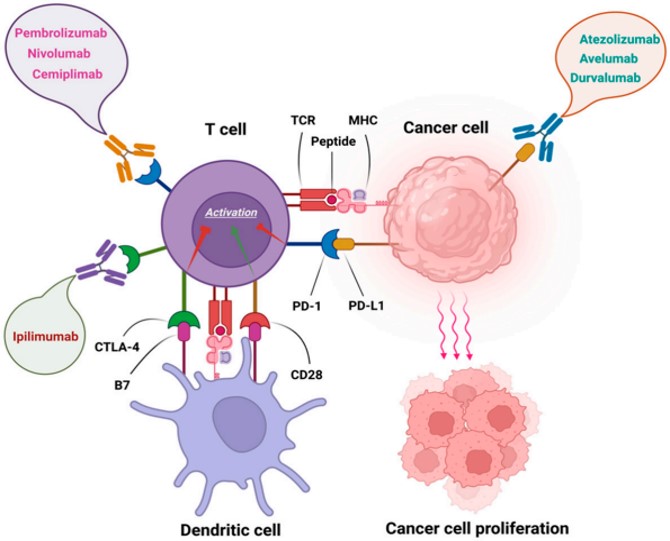 Fig.1 Immune checkpoint inhibitors.1,4
Fig.1 Immune checkpoint inhibitors.1,4
 Fig.2 The common stimulatory and inhibitory immune checkpoint receptors.
Fig.2 The common stimulatory and inhibitory immune checkpoint receptors.
Available ICIs based on Efficacy Services
| Animal Models | Modeling Methods and Core Evaluation Values |
| Syngeneic Mouse Models | Transplanting mouse tumor cell lines (e.g., B16) or tumor fragments subcutaneously or orthotopically into immune-competent mice. Gold standard for ICI efficacy & TME analysis, essential for assessing the anti-tumor effect and T cell exhaustion reversal within a fully competent immune system. |
| Genetically Engineered Mouse Models (GEMMs) | Inducing spontaneous tumor development by introducing specific human oncogenes (e.g., Kras mutation) into the germline of immunocompetent mice. High-fidelity mechanism research & resistance modeling, used to study ICI primary resistance and long-term efficacy in naturally arising tumors. |
| Humanized Mice | Transplanting human immune cells (PBMCs or HSCs) into severely immunodeficient mice. Tumor is typically a human cell line derived xenograft (CDX) or patient derived xenografts (PDX). Translational assessment of human-specific ICIs, necessary for evaluating ICIs (e.g., human anti-PD-1 antibodies) that require human-to-human immune component interaction. |
| PDX Models | Transplanting fresh human tumor tissue fragments subcutaneously or orthotopically into severely immunodeficient mice. ICIs' combination with targeted agents screening, vital for evaluating the efficacy of non-immune drugs used in combination with ICIs in the clinic. |
| Humanized Target Mice | Knocking in the human gene sequence for a specific checkpoint protein (e.g., PD-1) into the corresponding mouse locus in an immunocompetent host. Evaluation of human-specific ICIs in an intact immune system, ideal for testing human therapeutic antibodies without the variability of human immune cell engraftment. |
| irAEs Specific Models | Utilizing specific genetically modified mice (e.g., single or double immune gene knockouts, or transgenic mice) that are prone to developing systemic inflammation when treated with high doses of ICIs (e.g., anti-CTLA-4, anti-PD-1 combination). Prediction and mechanistic study of clinical irAEs. Essential for assessing the drug's therapeutic window, identifying organ-specific toxicity (e.g., colitis, myocarditis), and elucidating the underlying immune mechanisms driving toxicity. |
| Tumor Organoids | Derived from patient tissue (PDX or primary tumor) and grown as self-assembling, three-dimensional cultures, often incorporating TME components. High-throughput drug screening and mechanistic MoA studies, providing a personalized platform for rapid combination screening and controlled mechanistic studies. |
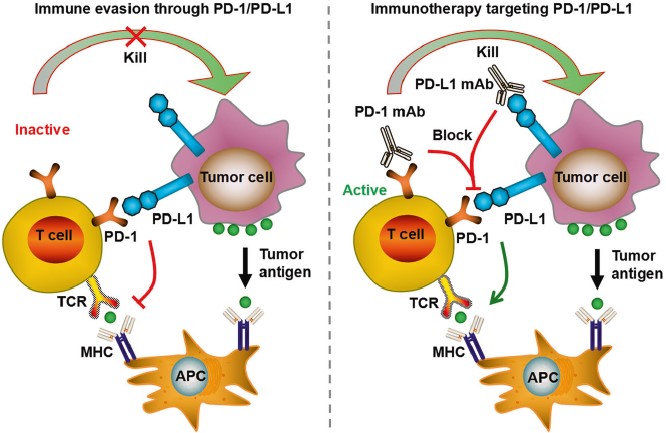
|
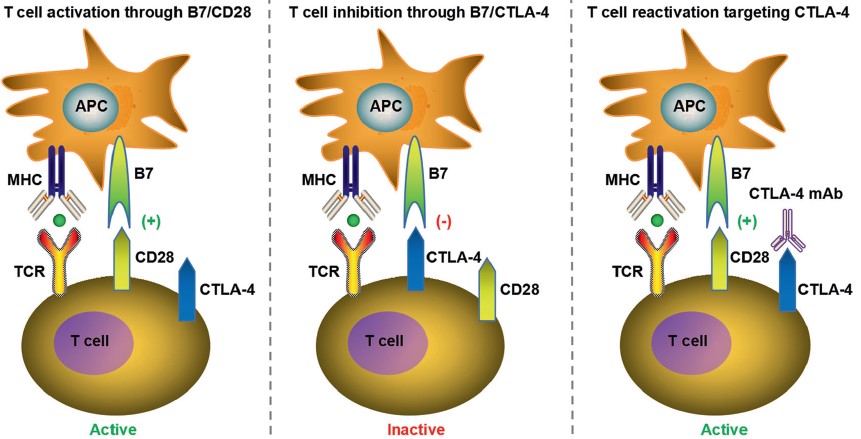
|
| Fig.3 PD-1/PD-L1 pathway contributes to tumor immune escape, enabling tumors to be resistant to immune response.2,4 | Fig.4 CTLA-4 negative regulates T cell responses through several ways, such as attenuating T cell receptor (TCR) and CD28 signaling by competitively binding to B7 with CD28.2,4 |
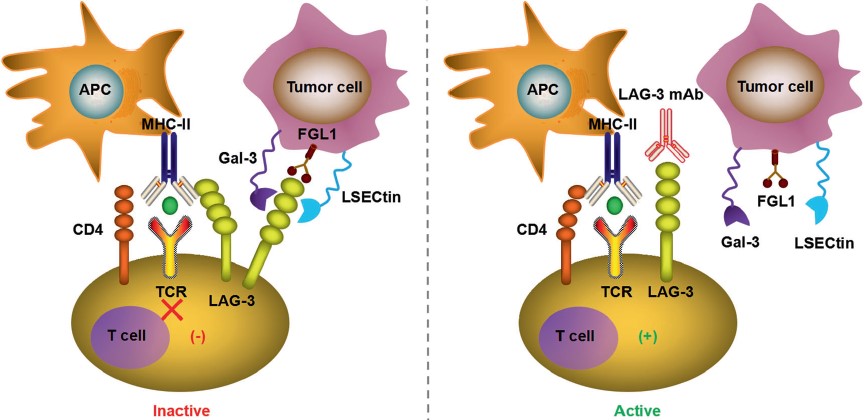
|
|
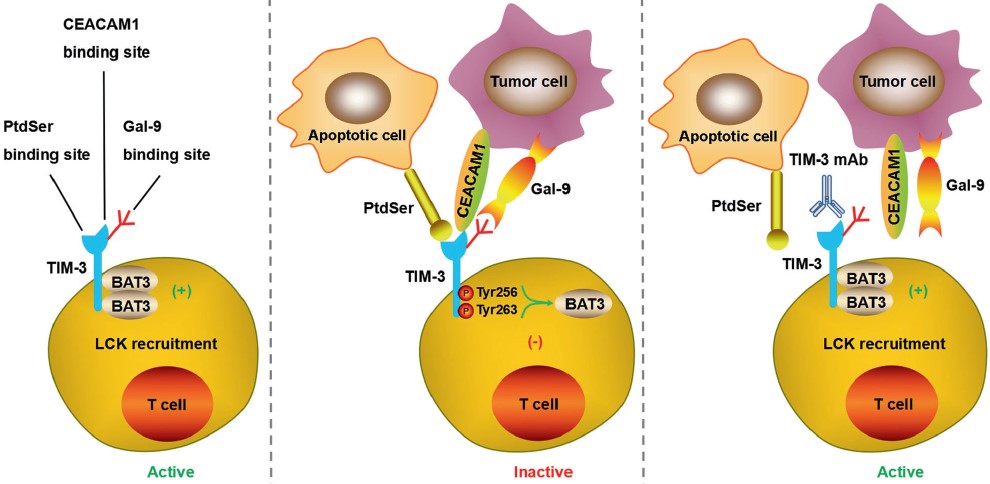
| Fig.5 The typical ligand of LAG-3 is MHC class II (MHC-II), which interacts stably with LAG-3 through the D1 domain and has a significantly higher affinity than CD4.2,4 | Fig.6 TIM-3 has multiple ligands, such as Gal-9, CEACAM1, PtdSer, and HMGB1, and each of them binds to different regions of the extracellular IgV domain.2,4 |
Evaluation Platform
The Creative Biolabs platform for ICI efficacy evaluation is built around functionally intact immune systems, aiming to comprehensively elucidate drug anti-tumor activity, MOA, and combination potential through a combination of gold-standard models and deep molecular analysis. We utilize cutting-edge, precise instrumentation and validated advanced analytical methods to guarantee superior data accuracy and trustworthiness, driving the rapid acceleration of your drug development pipeline.
- Tumor Growth Inhibition (TGI): Longitudinal monitoring of tumor volume changes using calipers and in vivo imaging techniques, providing the most basic evidence of efficacy.
- Overall Survival (OS): Quantification of potential survival benefit using Kaplan-Meier survival curves.
- Immune Cell Phenotype & Function: Utilizing FACS to analyze the number, subtypes (CD4+, CD8+, Treg), and functional status of tumor-infiltrating lymphocytes (TILs) in peripheral blood, spleen, and tumor tissue.
- TME Spatial Analysis: Utilizing Immunohistochemistry (IHC) and Multiplex Immunofluorescence (mIF) to visualize the spatial distribution and density of immune cells in situ, assessing the ICI's ability to convert "cold" tumors into "hot" tumors.
- Cytokine and Molecular Pathway Analysis: Quantification of systemic and local cytokines and chemokines (IFN-γ, IL-6) using ELISA / Luminex platforms. Analysis of gene expression changes in the TME via RNA-seq or RT-qPCR to elucidate molecular mechanisms of drug action and resistance pathways.
- PK/PD Correlation Studies: Linking drug exposure (PK) with changes in immune markers (PD) to establish the therapeutic window and guide safe clinical dosing.
Applications
- Preclinical Efficacy Assessment: We provide the gold-standard in vivo data (TGI and OS) in immunocompetent models, definitively proving the ICI's capacity to reverse T cell exhaustion and generate a durable anti-tumor immune response.
- Resistance Mechanism Research: We analyze molecular and immune evasion pathways in treated animal model tumors to identify the root causes of PD-1 resistance and pinpoint effective targets for next-generation or combination therapies.
- Drug Development and Dose Optimization: We utilize PK/PD animal models to precisely correlate drug exposure with immune marker changes, establishing the therapeutic window and informing the safe and effective clinical starting dose.
- Combination Therapy Strategy Development: We systemically evaluate synergistic effects by combining ICIs with other treatments (e.g., co-stimulatory agonists) directly within Syngeneic Models, optimizing future clinical combination protocols.
- Biomarker Development and Validation: We use RNA-seq and multiplex in vivo Imaging (e.g., mIF) on treated tumors from relevant animal models to identify and validate predictive molecular markers and guide patient selection.
- Personalized Precision Medicine: Our services employ PDX or GEMMs to model patient-specific tumor characteristics in vivo, allowing for the evaluation of ICI efficacy against diverse tumor subtypes and guiding individualized treatment strategies.
- Regulatory and Clinical Decision Support: We generate the necessary scientific evidence concerning MOA and irAEs risk prediction, utilizing specific irAEs animal models to significantly support the IND submission and accelerate clinical entry.
Advantages
- Expertise in Immuno-Oncology: Deep knowledge and experience in immuno-oncology and immune checkpoint pathways.
- Diverse Animal Models: Access to a wide range of validated immunocompetent models, including syngeneic and GEMMs, as well as advanced humanized models for testing human-specific therapeutics.
- Advanced Technological Capabilities: Cutting-edge laboratory facilities equipped with advanced technologies for genomic, proteomic, flow cytometry, imaging technologies, and high-throughput screening and metabolic analysis.
- Experienced Scientific Team: A team of skilled scientists and researchers with expertise in oncology, immunology, and pharmacology who can design and conduct complex studies efficiently.
- Customized Study Design: Capability to tailor study designs to meet specific client needs, including flexibility in protocol development and adjustment.
- Quality Assurance: Robust quality assurance processes in place to ensure accuracy, reliability, and reproducibility of research data.
- Data Analysis and Reporting: Strong capabilities in data analysis and interpretation, with clear and comprehensive reporting to support scientific findings and decision-making.
- Collaborative Approach: Willingness to work closely with clients to understand their project goals and provide insights and recommendations throughout the study process.
- Strong Track Record: A proven history of successful projects and satisfied clients in the field of ICIs and immune-oncology research.
- Scalability and Flexibility: Ability to scale operations and adapt quickly to project demands or changes in study design, volume, and timelines.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: What are the main goals of preclinical evaluation?
A: The primary goal for advancing an immuno-oncology drug to clinical trials is to build a strong scientific case by validating its anti-tumor efficacy, elucidating its mechanism of action, assessing its immune-related toxicity to inform safe dosing, and exploring beneficial synergistic combination therapies.
-
Q: How is a drug's efficacy assessed?
A: Efficacy is a multi-dimensional assessment that extends beyond tumor growth inhibition and survival by utilizing flow cytometry and immunohistochemistry for detailed immune cell analysis (e.g., TILs subsets), and employing ELISA/multiplex assays and RNA-seq for Molecular and Cytokine Analysis to elucidate the drug's mechanism of action.
-
Q: How is drug safety evaluated?
A: Safety evaluation for ICIs requires a special focus on immune-related toxicities, involving standard general toxicology to determine the maximum tolerated dose (MTD), utilizing irAEs models to predict specific adverse events, and performing PK/PD studies to connect drug exposure with changes in immune markers for safe clinical dosing.
Published Data
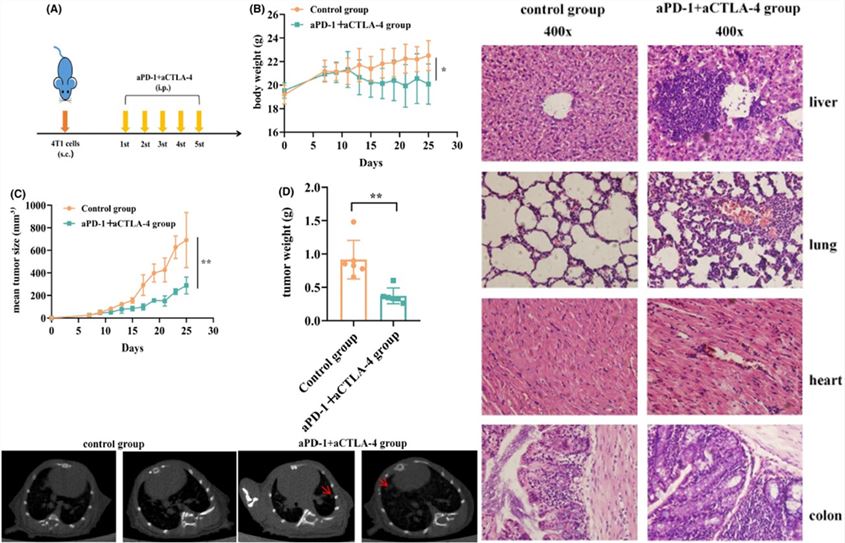 Fig.7 Dual ICI combination demonstrates potent anti-tumor efficacy counterbalanced by significant irAEs toxicity.3
Fig.7 Dual ICI combination demonstrates potent anti-tumor efficacy counterbalanced by significant irAEs toxicity.3
In the 4T1 mouse mammary tumor model, the combination of anti-PD-1 and anti-CTLA-4 antibodies resulted in a significant reduction in tumor volume (efficacy). However, this therapeutic benefit was accompanied by severe irAEs, evidenced by significant weight loss, multi-organ inflammatory infiltration, and lung inflammatory patches confirmed by CT and histopathology, highlighting the critical need for preclinical toxicity prediction.
References
- Shiravand, Yavar et al. "Immune Checkpoint Inhibitors in Cancer Therapy." Current oncology (Toronto, Ont.) vol. 29,5 3044-3060. https://doi.org/10.3390/curroncol29050247.
- Meng, Lingjiao et al. "Mechanisms of immune checkpoint inhibitors: insights into the regulation of circular RNAs involved in cancer hallmarks." Cell death & disease vol. 15,1 3. https://doi.org/10.1038/s41419-023-06389-5.
- Meng, Yuan et al. "Establishment of an animal model of immune-related adverse events induced by immune checkpoint inhibitors." Cancer medicine vol. 13,14 (2024): e70011. https://doi.org/10.1002/cam4.70011. Distributed under Open Access license CC BY 4.0, with modification.
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
