Tumor Seeding Types Modeling & Pharmacodynamics Services
Introduction
The success of modern oncology research hinges on bridging the gap between preclinical data and clinical outcomes. Traditional in vivo modeling often fails to capture the true complexity of human cancer, specifically, its organ-specific microenvironment and metastatic potential. Our Tumor Seeding Types Modeling & Pharmacodynamics Services platform directly addresses this translational challenge. We move beyond simple subcutaneous screening, offering Orthotopic models to ensure high-fidelity simulation of the native Tumor Microenvironment (TME) and local invasion, and Systemic/Disseminated models, which serve as the scientific benchmark for anti-metastatic drug evaluation. By precisely controlling the seeding environment, we ensure that every Pharmacodynamic (PD) endpoint is clinically relevant, delivering the robust, predictive data required to accelerate your drug candidate toward regulatory success.
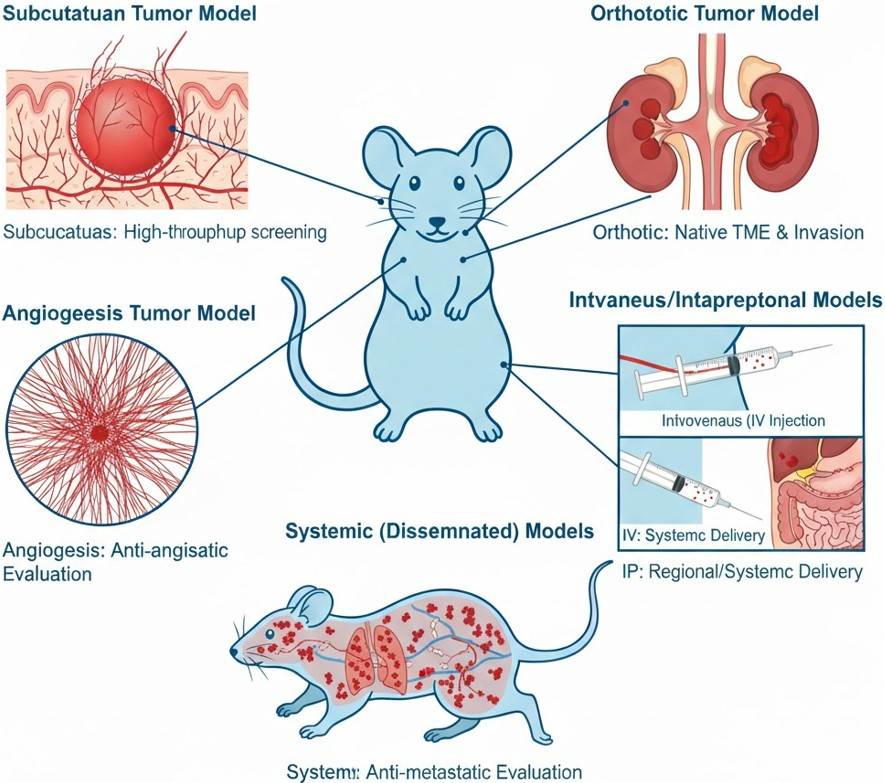 Fig. 1 Tumor seeding types & modeling strategies.
Fig. 1 Tumor seeding types & modeling strategies.
Available Tumor Seeding Types
Creative Biolabs' tumor Seeding Types serve as the critical translational platform that accelerates the oncology drug R&D pipeline. We provide the robust in vivo environment essential for validating complex tumorigenesis mechanisms and generating high-fidelity efficacy data. Our models are specifically validated for the precise evaluation of hot therapeutic modalities, including Immune Checkpoint Blockade (e.g., PD-1/PD-L1), next-generation Antibody-Drug Conjugates (ADCs), and novel targeted agents against hard-to-treat mutations like KRAS and EGFR, thereby ensuring successful clinical transition. Our portfolio of successfully established tumor models encompasses:
| Tumor Seeding Types | Modeling Method | Relevant Evaluation | Animal Species |
| Subcutaneous Tumor Models | Inject tumor cell suspension or tissue fragment subcutaneously into the flank or back. | Suitable for high-throughput, rapid screening of drug candidates. As the foundational model, it is mainly used to assess basic efficacy (e.g., TGI), PK/PD relationships, and maximum tolerated dose, making it the most economical and efficient choice for early preclinical stages. | Mouse (Nude, SCID, NOG, Balb/c, etc.), Rat |
| Orthotopic Tumor Models | Directly inject tumor cell suspension into the organ of natural origin (e.g., mammary fat pad, subcapsular liver, or lung lobe). | Possesses the highest clinical relevance. By establishing the model in the tumor's organ of origin, it faithfully simulates the Tumor Microenvironment (TME) and the process of local invasion. It is crucial for evaluating TME-targeting therapies, cell therapies, and studying the mechanisms of metastasis initiation. | Mouse, Rat, Dog |
| Angiogenesis Models | Subcutaneous injection of tumor or endothelial cells mixed with Matrigel, or utilizing the corneal micropocket assay. | Dedicated to mechanism validation. The focus is on assessing the drug's capacity and potency to block tumor neovascularization. It is the core tool for validating all drugs targeting pathways like Vascular Endothelial Growth Factor (VEGF). | Mouse, Rat |
| Intraperitoneal / Intraorgan Injection Models | Inject tumor cell suspension intravenously (IV) or intraperitoneally (IP). | IV injection is suitable for studying systemic drug distribution and the organ colonization capacity of tumor cells, making it key for assessing early metastasis. IP injection is used for efficacy evaluation of regional therapies (e.g., ovarian cancer, peritoneal pseudomyxoma). | Mouse, Rat |
| Systemic (Disseminated) Models | Intravenously inject tumor cell suspension into a specific site (e.g., left ventricle, femoral artery) to simulate systemic dissemination. | The gold standard for evaluating anti-metastatic drugs. By simulating the circulation of tumor cells and their dissemination and colonization in distant organs (e.g., brain, bone), it provides the highest level of data support for predicting the clinical efficacy of drugs blocking advanced disease. | Mouse |
Evaluation Platform
To ensure true translational success, our platform moves beyond traditional efficacy testing. We deploy a cutting-edge, multi-modal detection suite designed to overcome the complexity of the TME. This integrated system provides unparalleled detection resolution, linking non-invasive, real-time in vivo imaging to single-cell molecular fidelity. This rigorous approach yields highly reliable and clinically relevant data, delivered through four specialized pillars:
- In Vivo Imaging (e.g., BLI, CT, PET-CT) provides non-invasive, dynamic monitoring of tumor growth, metastasis, and therapeutic response, linking real-time cellular tracking to quantitative structural and metabolic data (e.g., osteolytic lesions).
- The molecular and Cellular Level Detection platform provides high-fidelity quantification of TME heterogeneity. Flow Cytometry isolates rare cell populations, while qPCR/Western Blot clarifies signaling pathways.
- The Histopathological Detection platform ensures morphological and phenotypic validation. Conventional staining assesses tissue damage and structure, while Advanced Immunostaining (e.g., IHC, mIHC) detects markers and enables precise co-localization analysis of TME components.
- The Functional and Efficacy Evaluation platform validates clinical outcomes through quantitative assessment of tumor growth and metastasis, complemented by survival analysis and essential evaluation of tumor microenvironment remodeling.
Applications
- Anti-Tumor Drug R&D and Screening: Leveraging the efficiency of Subcutaneous Tumor Models for high-throughput, rapid screening, this stage yields foundational efficacy (e.g., TGI) and early-stage Pharmacokinetics/Pharmacodynamics (PK/PD) correlation data, ensuring the efficient identification and progression of promising drug candidates.
- Basic Tumor and Mechanism Research: Orthotopic Seeding models' high TME fidelity is used, combined with molecular PD detection, to dissect mechanisms of local invasion. Systemic Disseminated Models are utilized to track and reveal the key molecular pathways involved in tumor cell circulation and distant metastasis.
- Drug Evaluation: Model selection is matched to therapy type: Syngeneic/Humanized Models are used for Immune PD assessment (e.g., T-cell infiltration) of immunotherapies. Systemic Disseminated Models are specifically utilized for the highest standard of anti-metastatic efficacy validation.
- Clinical Translation Research and Precision Medicine: We establish PDX models at the most clinically relevant Orthotopic or Systemic sites. Drug sensitivity testing (DST) at these sites provides site-specific Pharmacodynamic (PD) evidence, directly guiding clinical drug choices and enabling individualized precision medicine.
Our Advantages
- Comprehensive Tumor Model Portfolio (1000+ Resources): We offer over 3400 leading PDX models (covering 25 cancer types and supporting drug resistance studies), complemented by 200+ CDX models and 17 syngeneic models, providing market-leading coverage for all research scenarios.
- Integrated Multi-Omic Technology Platform: Our platform integrates Genomics, Proteomics, and Efficacy Data to support target-disease association analysis, significantly enhancing translational efficiency and the scientific rigor of the data.
- Precision Model Selection Strategy: We ensure optimal experimental design by recommending syngeneic models for immunotherapies, genetically matched PDX models for targeted therapies, and orthotopic models for microenvironment-targeting drugs.
- Expertise in Complex & Customized Efficacy Studies: Having completed many efficacy evaluations, we specialize in complex models, such as tumor bone metastasis and multidrug resistance, offering comprehensive solutions that integrate mechanism and efficacy research.
- Accelerated One-Stop Preclinical Solution: We offer a bundled service integrating efficacy, pharmacokinetics (PK), and toxicology assessments to streamline the R&D process and shorten the total development cycle by 30%.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: What is the core advantage of the subcutaneous tumor xenograft model?
A: The advantage of the subcutaneous model lies in its simple operation, high tumor formation rate (>80%), and ease of visual observation and measurement of tumor size, making it suitable for early-stage drug screening.
-
Q: What are the limitations of the subcutaneous model?
A: The limitation is that the tumor microenvironment is significantly different from the primary site, lacking organ-specific stroma, blood vessels, and immune cells, and rarely undergoing spontaneous metastasis.
-
Q: What types of studies are not suitable for the subcutaneous model?
A: It is not suitable for studying tumor invasion, metastasis mechanisms, or drugs that rely on the microenvironment.
-
Q: What are the methods for constructing orthotopic models? What are the technical challenges?
A: Constructed by direct cell injection or tissue block implantation into the corresponding organ, orthotopic models present significant technical hurdles, including complex surgical procedures, the need for imaging assistance for localization, and high variability in tumor formation rates.
-
Q: Which studies must use the orthotopic model?
A: It is suitable for studying tumor invasion mechanisms, organ-specific metastasis, drug efficacy dependent on the microenvironment, or evaluating the effects of local treatments such as surgery or radiotherapy.
-
Q: How is tumor angiogenesis quantified?
A: Common indicators include: microvessel density, vessel length/branching, vascular permeability, and levels of pro-angiogenic factors.
-
Q: What is the relationship between the angiogenesis model and tumor growth?
A: Tumor growth depends on blood vessels for nutrients. The angiogenesis model focuses on the tumor's ability to induce the formation of new blood vessels.
-
Q: What is the core purpose of the intravenous and intraperitoneal injection models? What is the difference between them?
A: Both the IP model and the IV model are essential for studying tumor metastasis, though they simulate distinct routes of tumor spread. The IP model simulates peritoneal metastasis (or peritoneal seeding), making it suitable for cancers like ovarian or gastric cancer. In contrast, the IV model simulates hematogenous metastasis via tail vein or intracardiac injection, and is primarily used to study distant organ colonization, such as metastasis to the lungs, bones, or brain.
-
Q: What is the difference between a dissemination model and a metastasis model?
A: The dissemination model focuses on the early survival and dormancy of microscopic disseminated tumor cells (DTCs), simulating a state where cancer cells have disseminated but not yet formed macroscopic metastatic lesions. The metastasis model, in contrast, focuses on the formation of macroscopic metastatic lesions.
-
Q: What types of studies are suitable for the dissemination model?
A: It is suitable for exploring tumor dormancy mechanisms, predicting recurrence risk, or developing drugs that target minimal residual disease (MRD).
Published Data
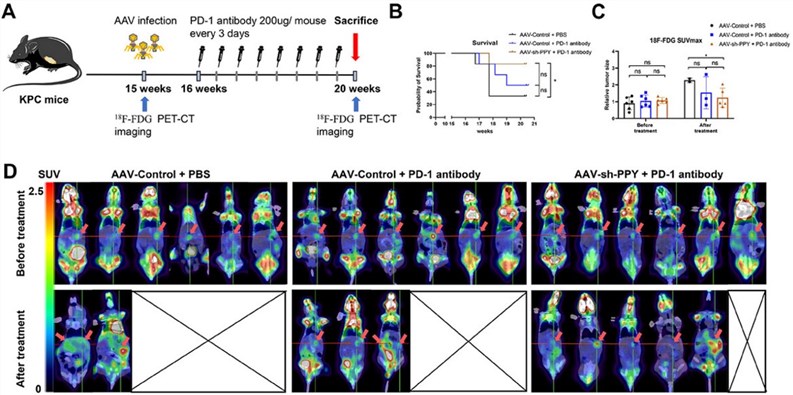 Fig.2 Targeting pancreatic polypeptide (PPY) can enhance the efficacy of anti-PD-1 antibodies in pancreatic cancer.1
Fig.2 Targeting pancreatic polypeptide (PPY) can enhance the efficacy of anti-PD-1 antibodies in pancreatic cancer.1
Treatment with AAV-sh-PPY combined with anti-PD-1 significantly prolonged survival (resulting in an 83.3% survival rate) and reduced tumor volume (SUVmax detected by PET-CT) compared to anti-PD-1 monotherapy or the control treatment.
Reference
- Cao, Mengdie et al. "PPY-Induced iCAFs Cultivate an Immunosuppressive Microenvironment in Pancreatic Cancer." Advanced science (Weinheim, Baden-Wurttemberg, Germany) vol. 12,20 (2025): e2413432. https://doi.org/10.1002/advs.202413432. Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
