Prodrug based Efficacy Modeling & Pharmacodynamics Service
Introduction
Prodrugs, defined as pharmacologically inert derivatives requiring in vivo conversion to active cytotoxic agents, represent a strategic oncology approach designed to enhance therapeutic index by maximizing tumor-specific activation. By utilizing advanced and highly predictive tumor models, Creative Biolabs ensures your prodrug achieves optimal targeted distribution and minimal systemic toxicity. Our dedicated platform is equipped to precisely validate the tumor-specific activation mechanism, whether it is enzyme-driven, hypoxia-dependent, or pH-sensitive within the tumor microenvironment. We offer integrated pharmacokinetics (PK) and pharmacodynamics (PD) modeling services that rigorously connect active drug generation and disposition with cytotoxicity at the tumor site.
Available Prodrug based Efficacy Services
Prodrugs can improve bioavailability, target drug delivery, enhance the taste or stability of the drug, prolong the duration of action, and reduce toxicity. Based on these characteristics, we optimize animal models specifically for the pharmacological and efficacy evaluation of prodrug molecules, accelerating the R&D process. Tailored efficacy services driven by the unique chemical structure, activation mechanism, and carrier/delivery system of the prodrug molecule.
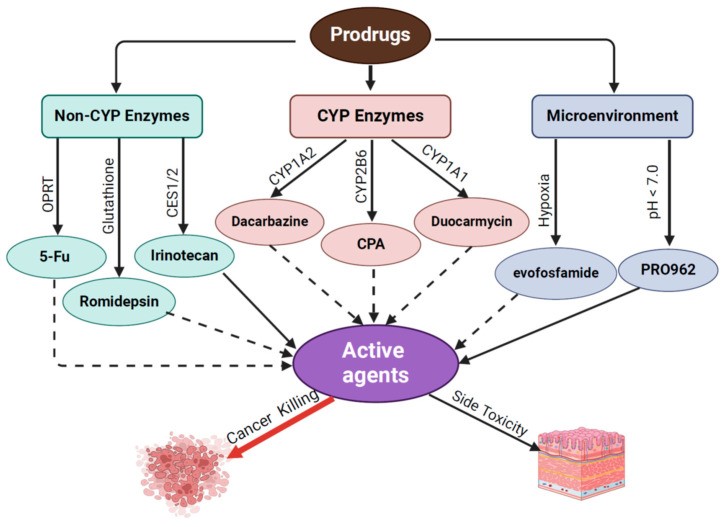 Fig.1 Schematic illustration of chemotherapeutic prodrugs.1,3
Fig.1 Schematic illustration of chemotherapeutic prodrugs.1,3
| Category | Animal Modeling | Characteristics | Key Evaluation Values |
| Carrier-Prodrugs | Subcutaneous/Orthotopic Xenograft Models | The active drug is temporarily masked by a simple, cleavable carrier (e.g., ester) to improve oral bioavailability or solubility. | Improvement in oral bioavailability (if applicable), consistency/superiority of PK/PD relationship (drug concentration vs. tumor inhibition) compared to the parent drug. |
| Bioprecursors | Humanized CYP450 Mouse Models (for metabolism study) and Xenograft Models. | Activation relies on metabolic conversion (e.g., by CYP450 enzymes) within the host to release the active drug. | Efficiency of in vivo metabolic activation (prodrug-to-active drug conversion rate), impact of metabolic variability on efficacy, and potential for drug-drug interactions. |
| Self-eliminating Prodrugs | Subcutaneous/Orthotopic Xenograft Models. | The carrier is cleaved enzymatically, followed by the spontaneous decomposition of the remaining structure to release the active drug. Aims for efficient and controlled release. | Stability of the intermediate compound, kinetics of the self-immolation step (to ensure sustained, efficient release), and comparison of tumor inhibition efficacy. |
| Target-Specific Prodrugs | Tumor-Specific Activation Models (e.g., xenografts expressing a high level of a tumor-associated enzyme). | Activation relies on unique tumor microenvironment conditions (e.g., low pH, hypoxia, or overexpressed tumor-specific enzymes). | Specificity of activation at the tumor site (e.g., confirming activation through imaging or tissue analysis), demonstration of reduced systemic toxicity, and enhanced efficacy. |
| Macromolecular Prodrugs | Subcutaneous/Orthotopic Xenograft Models exploiting the EPR effect (Enhanced Permeability and Retention). | Active drug is conjugated to a large polymer (e.g., PEG), aiming to utilize the tumor's leaky vasculature and poor lymphatic drainage. | Prolonged half-life (T1/2), tumor accumulation (assessed via biodistribution studies), and maximal tolerated dose (MTD) and efficacy under long-term dosing regimens. |
Evaluation Platform
ThePD platform constitutes the core of the evaluation service, designed to assess the efficacy, safety, targeting specificity, and activation mechanism of a prodrug within a living system, typically mouse models. The primary goals include determining MTD and optimal dose, validating targeting specificity, and quantifying anti-tumor efficacy.
-
In vivo models
- Cell Line Derived Xenograft (CDX) Models: CDX Models offer rapid, high-throughput screening and cost-effectiveness, making them ideal for the early-stage assessment of prodrugs. Their core value lies in quickly determining dose-response relationships and the basic anti-tumor effect of simpler prodrug designs.
- Patient Derived Xenograft (PDX) Models: PDX Models utilize patient tumor tissue, ensuring high clinical relevance by preserving genetic and histological complexity. Their primary value is in validating advanced prodrug candidates and providing the strongest available preclinical prediction of efficacy in human clinical trials.
- Specialized Models (Orthotopic/Enzyme Overexpression): Specialized Models are customized to simulate specific activation conditions (e.g., low pH, high enzyme concentration) or anatomical sites (e.g., orthotopic tumors). Their essential value is rigorously proving the site-specific activation and targeting efficiency of the prodrug design.
- Genetically Engineered Mouse Models (GEMMs): GEMMs feature endogenously developed tumors within an immune-competent host and native microenvironment. Their core value is critical for assessing immunomodulatory prodrugs and for in-depth, high-translatability studies of complex mechanisms and long-term safety.
- Tumor Organoids: Tumor Organoids are patient derived 3D ex vivo structures suitable for rapid, individualized screening. They provide an efficient platform for optimizing dosing regimens and supporting personalized medicine decisions before committing to large-scale in vivo studies.
-
Key Efficacy Endpoints
- Tumor Growth Inhibition (TGI): Regular measurement of tumor volume using high-precision calipers combined with live imaging techniques (e.g., Micro-CT/PET) to calculate the rate of volume change. This is the primary efficacy endpoint, aiming to assess the degree (TGI%) and duration of tumor suppression, thereby determining the minimum effective dose (MED).
- Survival Analysis: Continuous monitoring of animal survival status, utilizing Kaplan-Meier curves for rigorous statistical analysis. As a secondary efficacy endpoint, it evaluates whether the prodrug significantly prolongs the animal's lifespan, which is a direct reflection of the ultimate clinical benefit.
- Histopathology: Post-study collection of tumor and major organ tissues (e.g., liver, kidney, heart) for H&E staining and digital pathology scanning analysis. Used to assess the ratio of tumor cell apoptosis/necrosis (efficacy) while also serving as a critical tool for evaluating organ toxicity (safety).
- Biomarkers: Employing high-sensitivity techniques (e.g., Immunohistochemistry (IHC), ELISA, Western Blot), sometimes integrated with cell sequencing analysis. Verifies the prodrug's mechanism of action by detecting signal pathway proteins, proliferation (e.g., Ki-67), or apoptosis (e.g., Caspase-3) markers, confirming successful target modulation by the active drug.
Applications
- Early Screening & Candidate Optimization: This application utilizes CDX models and in vitro conversion assays to rapidly filter out unstable or inefficient prodrug molecules in the early stages of development, thereby enabling the quick selection of optimal candidates.
- PD Mechanism Elucidation: By employing specialized models and biomarker analysis, this application quantifies the prodrug's targeting enrichment, strictly verifying that the active drug activates according to its intended mechanism and site.
- Efficacy Assessment: This service performs TGI and survival analysis in PDX/GEMMs and other high-translatability models to quantify the prodrug's therapeutic advantage and confirm its substantive benefit over the parent drug or standard therapies.
- Therapeutic Strategy Optimization & Combination Prediction: By establishing robust PK-PD correlation models and conducting studies in complex animal models, this application simultaneously determines the optimal single-agent dosing regimen (dose, frequency, route) and predicts the synergistic effects of combination therapies, thereby providing a data-driven approach to optimize scheduling and maximize clinical benefit.
Our Advantages
- Expertise and Experience: We hold substantial, well-established proficiency in pharmaceutical development, with core competence areas encompassing oncology and anti-tumor therapeutics. This expertise spans key preclinical disciplines, including pharmacology, toxicology, and PK.
- State-of-the-Art Technology and Equipment: Research efficiency and data fidelity are maximized through access to advanced technological platforms and instrumentation, including high-throughput screening systems, along with comprehensive genomic and proteomic tools.
- Customization and Flexibility: We possess the capacity to deliver highly adaptable and bespoke research solutions, allowing us to align services precisely with the distinct specifications of varied project mandates.
- Project Management Experience: Project execution is streamlined by a team of seasoned managers who ensure efficient coordination across diverse mandates, thus guaranteeing completion that is both timely and compliant with budgetary specifications.
- Comprehensive Service Range: We deliver comprehensive services spanning from early discovery and preclinical investigation through regulatory support, consolidating all client requirements onto a unified operational platform.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q. Why are both CDX and PDX animal models used to evaluate the anti-cancer efficacy of a prodrug?
A: CDX models are suitable for rapid, high-throughput preliminary screening and mechanism studies, while PDX models, due to their higher clinical relevance, are often used to validate CDX results and more accurately predict the efficacy of a prodrug in human clinical trials.
-
Q. Beyond tumor size, what other metrics are evaluated in prodrug efficacy studies?
A: Efficacy evaluation for prodrugs primarily involves survival analysis to determine if the drug significantly prolongs the animals' lifespan, and histopathology to analyze tumor and organ tissues for assessing toxicity and therapeutic impact; most crucially, it requires establishing the PK-PD Correlation, linking the concentration of the prodrug and its active metabolite in the body (PK) with the resulting anti-tumor effect (PD) to optimize dosing.
-
Q. What unique safety considerations apply to prodrug toxicity evaluation?
A: Toxicity assessment of prodrugs is a critical component of pharmacodynamic studies, involving the quantification of the intrinsic toxicity of the active drug on major organ systems like the hepatic, renal, and cardiac compartments in relevant animal models; this is followed by confirming the on-target toxicity by measuring the active drug's concentration across diverse organs to verify its intended tumor confinement and site-specific activation; finally, comprehensive safety profiling requires assessing off-target toxicity, which evaluates potential cytotoxic effects independent of the intended mechanism for the entire therapeutic entity, encompassing the parent compound and all non-active metabolites.
Published Data
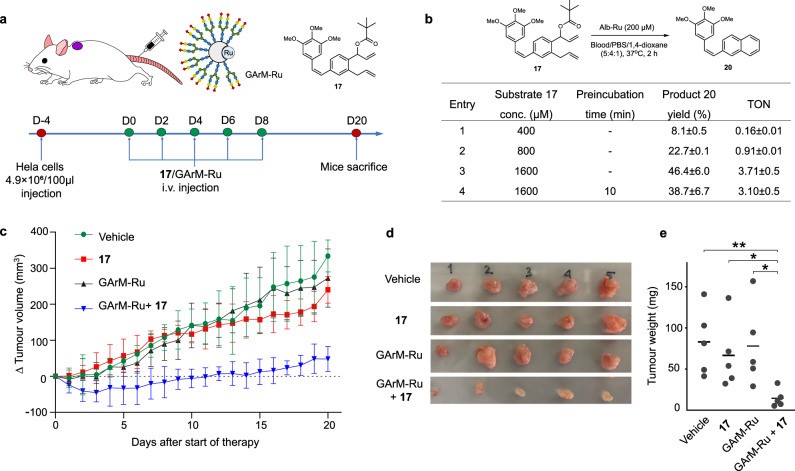 Fig. 2 Catalytic prodrug activation achieves significant tumor suppression with no systemic toxicity in the HeLa model.2,3
Fig. 2 Catalytic prodrug activation achieves significant tumor suppression with no systemic toxicity in the HeLa model.2,3
The in vivo synthetic prodrug therapy utilizes a Glycosylated Artificial Metalloenzyme (GArM-Ru) to activate a naphthyl-combretastatin prodrug. This targeted activation demonstrates significant suppression of HeLa tumor growth in mice while exhibiting no discernible toxic side effects, validating the therapeutic advantage of enzyme-driven, site-specific prodrug activation.
References
- Kurian, Ritika, and Hongbing Wang. "Prodrugs in Oncology: Bioactivation and Impact on Therapeutic Efficacy and Toxicity." International Journal of Molecular Sciences vol. 26,3 988. https://doi.org/10.3390/ijms26030988
- Nasibullin, Igor et al. "Synthetic prodrug design enables biocatalytic activation in mice to elicit tumor growth suppression." Nature communications vol. 13,1 39. https://doi.org/10.1038/s41467-021-27804-5
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
