Cancer Vaccine based Efficacy Modeling & Pharmacodynamics Service
Introduction
Cancer vaccines are a type of immunotherapy that helps the immune system recognize and combat cancer cells. Unlike traditional vaccines, which prevent infectious diseases, cancer vaccines are designed to either prevent cancer from developing, treat existing cancer, or prevent cancer from recurring. Cancer vaccines are broadly categorized into two main types: preventive cancer vaccines, which aim to stop cancer from developing (e.g., HPV vaccines), and therapeutic cancer vaccines, designed to treat existing malignancies. The therapeutic category encompasses several distinct platforms, including nucleic acid based Vaccines (e.g., DNA and mRNA vaccines), protein or peptide vaccines, cell based vaccines (e.g., Whole-Cell and Dendritic Cell vaccines), viral vector vaccines, and various Immune Stimulants (e.g., Bacillus Calmette-Guérin (BCG) and the oncolytic virus Talimogene laherparepvec (T-VEC)). All these diverse approaches share the common goal of activating the patient's immune system to recognize and eliminate cancer cells. Creative Biolabs offers industry-leading in vivo research services, tailoring pharmacodynamic and animal model studies to the unique mechanism of action of each cancer vaccine, accelerating your preclinical development.
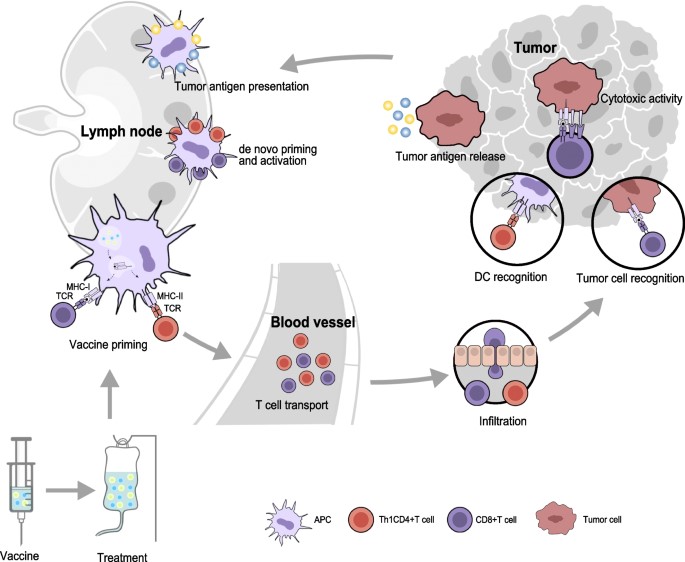 Fig. 1 Immune response induced by therapeutic cancer vaccines.1,5
Fig. 1 Immune response induced by therapeutic cancer vaccines.1,5
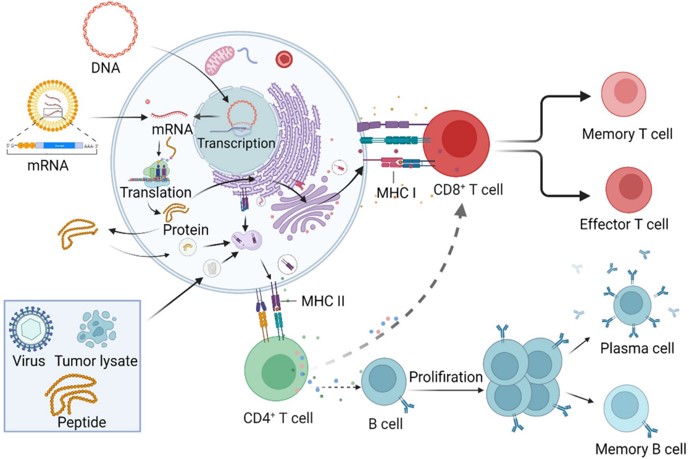 Fig.2 Tumor-immune cycle induced by cancer vaccines.2,5
Fig.2 Tumor-immune cycle induced by cancer vaccines.2,5
Available Cancer vaccines based Efficacy Services
| Vaccine Category | Core Classification | Key Efficacy Metrics | Preferred Animal Models | Typical Application Scenarios |
| Preventive Cancer Vaccines | Recombinant Protein/Virus-Like Particles (VLPs) | Antibody Titers, Reduction in Cancer Incidence | Wild-type Mice, Transgenic Mice | Healthy individuals, preventing virus-associated cancers (e.g., Cervical, Liver Cancer). |
| Cell based Vaccines | Autologous Dendritic Cells (DCs), Whole Tumor Cells | DC Maturation, T Cell Induction, OS | Syngeneic Mouse Models, Canine Spontaneous Cancer Models | Treating specific established cancers (e.g., metastatic prostate cancer with Sipuleucel-T). |
| Viral based Vaccines | Oncolytic Viruses (OVs), Replication-Defective Vectors | Virus Replication/Oncolysis, CD8+ T Cell Response, OS | Immunocompetent Mouse Models, NHPs | Solid tumors, delivered intratumorally for direct cell killing and systemic immune priming. |
| Peptide based Vaccines | Short Peptides/Synthetic Long Peptides (SLPs) | HLA-Restricted CD8+ T Cell Induction, Progression-Free Survival (PFS) | HLA-Transgenic Mice | Personalized therapy targeting patient-specific Neoantigens, or common TAAs in HLA-matched patients. |
| Nucleic Acid based Vaccines | mRNA Vaccines, DNA Vaccines | Antigen Expression Level, T Cell and Antibody Response, PFS | Mice/Rats, NHPs | Flexible platform for personalized neoantigen vaccines, often used in combination with ICIs. |
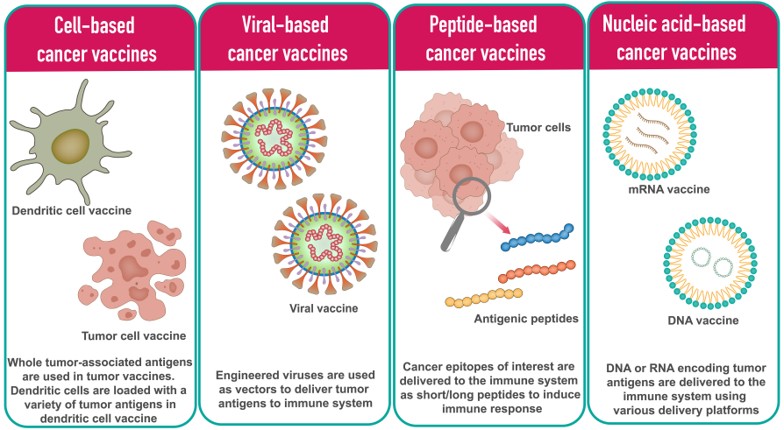 Fig.3 Different types of cancer vaccine platforms.3,5
Fig.3 Different types of cancer vaccine platforms.3,5
Evaluation Platform
Creative Biolabs ensures the selection of the most clinically relevant in vivo and advanced ex vivo models for your cancer vaccine, moving beyond standardized testing to offer mechanism-driven model customization.
Immunocompetent Models: Validating Efficacy in an Intact Immune System
- Syngeneic Mouse Tumor Models: Utilize mouse tumor cell lines (e.g., B16 melanoma, CT26 colon carcinoma) implanted into immunologically competent mice of the same genetic background. The gold standard for evaluating the anti-tumor efficacy of most therapeutic cancer vaccines (nucleic acid, peptide, viral, cell based). Essential for assessing whether vaccine induced T cell responses can overcome the tumor's immunosuppressive microenvironment, and for testing synergistic effects with Immune Checkpoint Inhibitors (ICIs).
- In Situ Vaccination Models: Tumor cells or viral vectors are injected directly into the skin, muscle, or existing tumor mass, mimicking clinical intratumoral delivery. Used to evaluate the local immune priming capacity of Oncolytic Viruses (OVs) and specific cell based vaccines, and their ability to induce a subsequent systemic immune response (the abscopal effect, targeting untreated distant tumors).
Advanced Translational Models: Simulating Clinical Complexity
- Genetically Engineered Mouse Models (GEMMs): Mice in which cancer arises de novo due to specific oncogene mutations, retaining the complex tumor architecture and a naturally evolved immune microenvironment. Ideal for assessing the vaccine's ability to break natural immune tolerance to self-antigens and to track the vaccine's influence throughout the entire tumor evolution and metastasis process, offering superior predictive value over implanted models.
- Tumor Organoids (Advanced Ex Vivo Platform): Patient derived, three-dimensional (3D) cultures that faithfully retain the tissue architecture and genetic heterogeneity of the original tumor. Serves as a high-throughput screening platform for personalized neoantigen selection and in vitro autologous T cell cytotoxicity validation by co-culturing the organoid with vaccine-primed T cells. This is a powerful tool in personalized medicine.
Humanized Immune Models: Enhancing Clinical Predictivity
- HLA-Transgenic Mouse Models: Mice are genetically engineered to express specific human MHC Class I (e.g., HLA-A2) or Class II molecules. Crucial for vaccines targeting human antigens or peptides. This validates the peptide's presentation efficiency in a human MHC context and its ability to induce CD8+ T cell responses, serving as a key preclinical hurdle for personalized peptide and nucleic acid vaccines.
- Humanized PBMC / CD34+ Mice: Immunodeficient mice engrafted with human Peripheral Blood Mononuclear Cells (PBMCs) or hematopoietic stem cells (CD34+). Used to assess the function and safety of vaccines within a human immune cell environment, particularly useful for evaluating the initial activation of cell based vaccines and the cross-reactivity of the human immune repertoire.
In Vitro High-Throughput Immunology Analysis
- Multi-Color Flow Cytometry: Multi-Color Flow Cytometry serves as the gold standard for immune cell analysis, precisely utilizing fluorescent labeling to identify APC maturation (CD80, CD86) and T cell subset status (CD4+, CD8+, Treg, and exhaustion markers like PD-1), allowing for early assessment of vaccine induced immune activation and potential suppression risk.
- ELISpot & Intracellular Cytokine Staining (ICS): The ELISpot and ICS are utilized for the quantitative measurement of functional T cell responses, employing high-sensitivity ELISpot to precisely quantify cytokine secretion (IFN-γ, IL-2) in Spot Forming Units (SFU) and ICS to assess T cell polyfunctionality, which is crucial for determining the quality of the immune reaction.
- Cytotoxic T Lymphocyte Assays: CTL Assays directly measure the ultimate functional endpoint of the immune response by quantifying the Specific Lysis Percentage of target tumor cells in vitro, providing direct evidence of the vaccine induced T cell killing efficacy.
- HLA Binding Assays: MHC Binding Assays are critical for peptide and nucleic acid vaccines, confirming that synthetic peptides or antigens possess the necessary affinity for human HLA by measuring Binding Affinity (IC50) and Peptide Stability, which is key to ensuring efficient immune presentation and target optimization.
Pharmacodynamics (PD) and Biomarker Analysis:
To deeply understand the ultimate mechanism of action of your vaccine, we conduct refined and comprehensive biomarker analyses:
- Tumor Microenvironment (TME) Analysis: We utilize Immunohistochemistry (IHC) and Multiplex Immunofluorescence (mIF) techniques to deeply profile the density, spatial location, and functional status of Tumor-Infiltrating Lymphocytes (TILs). This assesses the degree to which the vaccine has successfully "heated up" the TME.
- Cytokine/Chemokine Profiling: We employ high-throughput platforms like Luminex to quantify Th1, Th2, and Treg-associated inflammatory factors and immunoregulatory molecules in serum and tumor tissues. Evaluates the magnitude and specific nature (e.g., Th1 vs. Th2 skewing) of the systemic immune activation.
- Immune Memory Assessment: We track long-term immunological endpoints to evaluate the ability of vaccine induced memory T cells to sustain protective and cytotoxic function when faced with tumor rechallenge. Provides crucial data on the durability and persistence of the vaccine's therapeutic effect.
Applications
- Antigen Selection & Optimization: We utilize advanced bioinformatics and binding assays to identify the most potent and cancer-specific Tumor-Associated Antigens (TAAs) or Neoantigens. This application ensures your vaccine cargo targets epitopes with the highest potential to break immune tolerance and elicit a robust anti-tumor T cell response, moving beyond generalized targets to achieve clinical specificity.
- Adjuvant & Delivery System Synergistic Optimization: Our service systematically screens and optimizes the combination of adjuvants (e.g., CpG, STING agonists) and delivery vehicles (e.g., LNPs, viral vectors). This comprehensive approach maximizes APC uptake and maturation, ensuring the highest quality and magnitude of immune activation while minimizing the required dose for optimal therapeutic effect.
- Personalized Vaccine Design: We offer rapid, rigorous validation for personalized neoantigen panels derived from patient genomic data, crucial for mRNA and peptide platforms. This application provides essential in vivo preclinical proof that the unique set of antigens selected for a specific patient can functionally activate a tumor-clearing T cell response before expensive clinical manufacturing begins.
- Preclinical Efficacy Assessment: Efficacy is quantified through a robust, tiered approach, moving from in vitro functional assays (e.g., T cell killing) to rigorous in vivo modeling using Syngeneic or HLA-Transgenic tumor models. We deliver statistically significant data on Tumor Growth Inhibition (TGI) and Overall Survival (OS), providing the essential evidence required for IND submission.
- Pharmacokinetics (PK) Characterization: Our service analyzes the systemic and local PK characteristics of the vaccine construct, including stability and distribution within immune organs (like lymph nodes). This tracking is vital for optimizing the delivery system and establishing the ideal dose regimen, ensuring the vaccine reaches the correct immune compartment at the necessary concentration for successful immune priming.
- Combination Therapy Design: We model and test rational combination strategies, focusing primarily on pairing cancer vaccines with Immune Checkpoint Inhibitors (ICIs) or standard-of-care agents. This application is critical for identifying synergistic effects that convert immunologically "cold" tumors into "hot" tumors, a necessary step for overcoming primary and acquired resistance in advanced cancer treatment.
- Safety Assessment & Risk Prediction: Safety is addressed through targeted immunotoxicity studies in appropriate models, looking specifically for the activation of auto-reactive T cells and the prediction of immune-related adverse events (irAEs). This proactive assessment allows for early identification of potential risks, enabling formulation adjustments to ensure the safe and smooth transition of your innovative vaccine into human clinical trials.
Our Advantages
- Expertise and Experience: Extensive experience in oncology research and animal models. A professional scientific team knowledgeable in complex tumor biology and immunological mechanisms.
- Diverse Animal Model Portfolio: Provision of a wide range of animal models to meet different research needs. Capability to develop customized models according to specific research requirements.
- Advanced Technology and Facilities: State-of-the-art laboratory facilities and equipment, including bio-imaging technologies, flow cytometry, and genomic analysis. Ensure precise pharmacological evaluation and immune response detection.
- Flexibility and Scalability: Ability to adapt research protocols and quickly respond to changes in client needs. Offer a full spectrum of services from proof of concept to preclinical development.
- Rigorous Quality Control: Implementation of a stringent quality management system to ensure the reproducibility and reliability of each experiment.
- Data Analysis Capabilities: A specialized data analysis team capable of handling complex experimental data and providing meaningful interpretations.
- Use of advanced data management systems to ensure data integrity and security.
- One-stop service: Offering a full suite of preclinical oncology solutions, from model creation and efficacy testing to integrated pharmacokinetics and toxicology studies.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: How is the efficacy of a cancer vaccine assessed in animal models?
A: Efficacy is typically assessed by observing tumor size reduction, survival rate improvements, immune response indicators (e.g., T-cell activation), and sometimes through imaging techniques to observe tumor progression.
-
Q: What pharmacokinetic parameters are important in cancer vaccine studies?
A: For most cancer vaccines, traditional plasma pharmacokinetic (PK) parameters are less relevant; the focus shifts entirely to Biodistribution and Pharmacodynamics (PD). Key considerations involve tracking the vaccine components (e.g., mRNA-LNPs, viral vectors) to confirm proper delivery to target sites like lymph nodes and immune cells. This is paired with measuring the duration and level of target antigen expression and, critically, assessing the persistence of the anti-tumor immune response, such as memory T-cells, as the true measure of the vaccine's long-term effect.
-
Q: How are toxicological assessments conducted for cancer vaccines in animal models?
A: Toxicology evaluations focus on identifying potential adverse effects, determining the safe dose range, and assessing any physiological or biochemical changes in major organ systems following vaccine administration.
-
Q: How is immune response measured in these studies?
A: Immune responses can be evaluated by measuring levels of specific antibodies, cytokine production, and the activity of immune cells such as T-cells and dendritic cells, often using assays like ELISA or flow cytometry.
-
Q: What endpoints are considered in the efficacy studies of cancer vaccines?
A: Common endpoints include tumor regression, survival analysis, reduction in metastasis, and biomarkers indicative of a successful immune response.
-
Q: How long do these studies typically last?
A: The duration varies depending on the specific goals and design of the study. Short-term studies might last days to weeks, while long-term survival or tumor regression studies could last several months.
Published Data
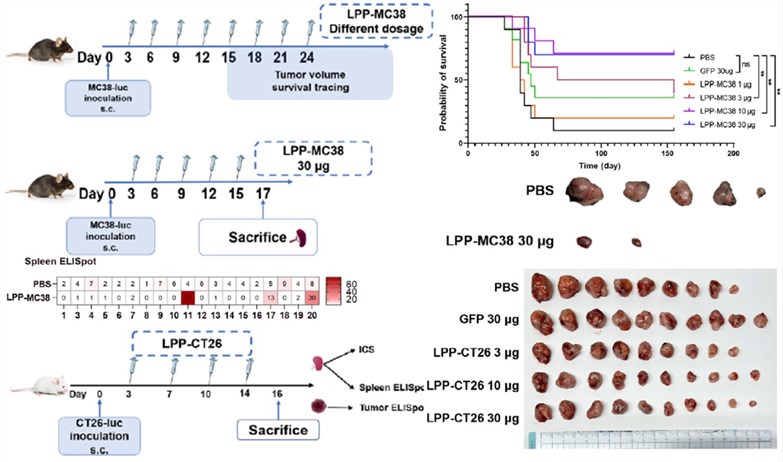 Fig.5 Tumor inhibition rates of the LPP-mRNA vaccine in MC38 and CT26 models.4,5
Fig.5 Tumor inhibition rates of the LPP-mRNA vaccine in MC38 and CT26 models.4,5
In the LPP-MC38 group, tumor weight was reduced by 75%, and 7 out of 10 mice were tumor-free at the 6-month follow-up. IFN-γ ELISpot analysis of splenocytes in response to MC38 neoantigens showed that 3 out of 20 neoantigens induced a strong response, while the control group showed no response. The LPP-CT26 group showed a 50% reduction in tumor weight.
References
- Lei, Wanting et al. "Cancer vaccines: platforms and current progress." Molecular biomedicine vol. 6,1 3. https://doi.org/10.1186/s43556-024-00241-8
- Liu, Jian et al. "Cancer vaccines as promising immuno-therapeutics: platforms and current progress." Journal of hematology & oncology vol. 15,1 28. https://doi.org/10.1186/s13045-022-01247-x
- Vishweshwaraiah, Yashavantha L, and Nikolay V Dokholyan. "mRNA vaccines for cancer immunotherapy." Frontiers in immunology vol. 13 1029069. https://doi.org/10.3389/fimmu.2022.1029069
- Fan, Ting et al. "Lipopolyplex-formulated mRNA cancer vaccine elicits strong neoantigen-specific T cell responses and antitumor activity." Science Advances vol. 10,41 (2024): eadn9961. https://www.science.org/doi/10.1126/sciadv.adn9961
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
