Immuno-Oncology Efficacy Modeling & Pharmacodynamics Services
Introduction
Immuno-Oncology (I-O) encompasses all therapeutic strategies aimed at activating, enhancing, or restoring the patient's own immune system to recognize, attack, and eliminate cancer cells, primarily including Targeted Molecule-Drug Conjugates (XDCs), Immune Checkpoint Inhibitors (ICIs), Cellular Therapies, Cancer Vaccines, Immunomodulators, and Oncolytic Viruses. I-O therapy activates, enhances, or modifies a patient's own immune system to identify and destroy cancer cells. The key strengths are its specific targeting of tumors and its long-term memory function, which helps prevent relapse. Creative Biolabs accelerates the research and development of cancer immunotherapies by providing a diverse portfolio of animal models for their assessment. The ultimate goal is to enable precision immunotherapy for all types of cancer, making it a central modality for achieving a cure.
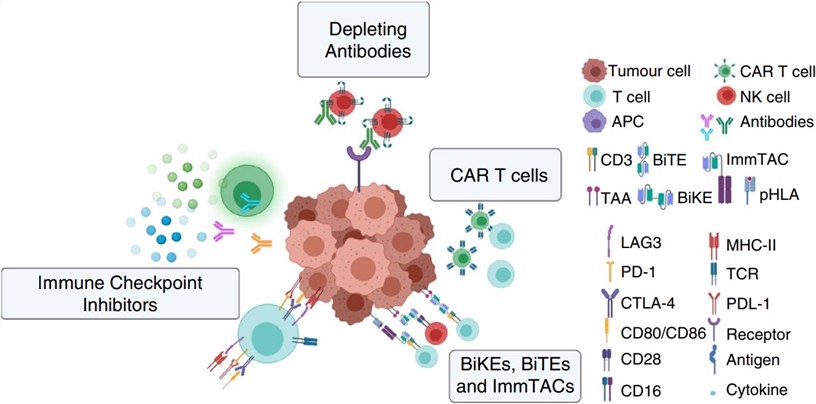 Fig.1 Current immunotherapies selectively enhance immune response.1,4
Fig.1 Current immunotherapies selectively enhance immune response.1,4
Available Immuno-Oncology Efficacy Services
Our Immuno-Oncology Efficacy Services are built upon years of deep, focused expertise and a strong track record in preclinical model execution and translational science. We offer a comprehensive and proven portfolio that spans the entire I-O pipeline, including established protocols for Immune Checkpoint Inhibitors, complex Cell Therapies (CAR-T, CAR-NK) in proprietary humanized models, Oncolytic Viruses, and Cancer Vaccines. This extensive experience ensures that we not only execute standard studies flawlessly but also possess the scientific acumen to design and manage complex, multi-modal combination therapies and troubleshoot challenging mechanistic questions. Partner with our seasoned team to leverage our proven success in generating predictive, high-quality data that reliably guides your therapeutic candidate from the bench to the clinic.
| Immuno-Oncology Efficacy | Modeling Method | Relevant Drug | Relevant Evaluation |
| X-DC based Efficacy Evaluation | CDX/PDX Models (expressing the target antigen) in immunodeficient mice. | ADCs (Antibody-Drug Conjugates), SMDCs (Small Molecule-Drug Conjugates), and Peptide-Drug Conjugates. | Drug Conjugates (ADCs/SMDCs) evaluation focuses on Tumor Growth Inhibition (TGI), Dose-Limiting Toxicity (DLT), off-target toxicity, and the bystander effect to comprehensively measure drug efficacy and safety. |
| CAR-T & CAR-NK Cell based Efficacy Evaluation | Humanized Mouse Models (NOG) engrafted with human PBMCs or CD34+ cells, bearing specific Target Tumor Models. | CAR-T Cell Therapy, CAR-NK Cell Therapy, and other Engineered Cell Therapies (e.g., TCR-T cells). | Cell Therapy evaluation covers pharmacodynamic metrics like in vivo anti-tumor efficacy, survival extension, and monitoring CAR cell expansion and persistence. It also strictly monitors key toxicities like Cytokine Release Syndrome (CRS). |
| Oncolytic Virus based Efficacy Evaluation | Syngeneic Mouse Models (for immune response) or Humanized Models (for viral replication and human-specific targets). | Oncolytic Viruses (OVs) (e.g., genetically modified Herpes Simplex Virus, Adenovirus), often in combination with ICIs (Immune Checkpoint Inhibitors). | Oncolytic Virus evaluation focuses on viral replication and dissemination efficiency within the tumor, assessing its synergistic anti-tumor effect when combined with ICIs (Immune Checkpoint Inhibitors), and analyzing immune activation. |
| Cancer Vaccine based Efficacy Evaluation | Syngeneic Mouse Models (e.g., C57BL/6, BALB/c) using Prophylactic or Therapeutic protocols. | Peptide Vaccines, DNA/RNA Vaccines, and Dendritic Cell (DC) based Vaccines. | Cancer Vaccine evaluation centers on measuring antigen-specific T-cell responses (e.g., ELISpot) and CTL activity as immunogenicity indicators, combined with tumor prevention rate, regression rate, and long-term survival to predict clinical endpoints. |
| CD19+ Humanized CDX/PDX Model based Efficacy Evaluation | Humanized Mice (with reconstituted human immune systems) bearing CD19-expressing B-cell tumor xenografts (CDX or PDX). | CD19-Targeting CAR-T/CAR-NK Therapies, and Bi-specific/Tri-specific Antibodies (e.g., CD19 × CD3). | CD19-Targeting Cell Therapy evaluation focuses on determining the optimal cell dosage and administration protocol. Efficacy is assessed in highly relevant PDX models, along with monitoring for antigen escape and tumor relapse mechanisms post-treatment. |
| Immune Checkpoint Inhibitor based Efficacy Evaluation | Syngeneic Mouse Models; Humanized Models (for evaluating human-specific ICIs). | Immune Checkpoint Blockers (PD-1/PD-L1, CTLA-4, LAG-3, TIGIT Inhibitors), and their Combination Therapies. | Immune Checkpoint Inhibitors (ICIs) evaluation assesses the anti-tumor effect of monotherapy and combination treatments. It involves biomarker discovery and validation by detailed analysis of TILs (e.g., CD8+Treg ratio, exhaustion markers) and their correlation with clinical response. |
Evaluation Platform
Our Immuno-Oncology platform stands at the technological forefront, dedicated to delivering the highest quality service and most predictive data in the field. We integrate strict Standard Operating Procedures (SOPs) with cutting-edge technologies, including multi-parameter flow cytometry, real-time IVIS imaging, and proprietary humanized mouse models to ensure all efficacy and mechanistic results are highly accurate, reproducible, and clinically predictive. By combining these advanced analytical tools with a comprehensive portfolio of biologically relevant models (Syngeneic, Humanized, and Tumor Organoids), we provide deep mechanistic insights into drug action, directly supporting critical development decisions, accelerating successful IND submissions, and ensuring your therapeutic candidates advance with maximum confidence.
- Xenograft Models: Patient derived Xenograft (PDX) Models preserve the original tumor's histology and genetic heterogeneity. They are often combined with humanized models to evaluate the efficacy of immunotherapies. When a cell line-derived xenograft (CDX) is implanted into humanized mice, the model contains both the tumor cells and a component of the human immune system. The resulting tumors are then used to study cancer progression, metastasis, and therapeutic responses to anti-cancer agents.
- Syngeneic Models: Because the immune system is competent, this model can effectively evaluate a drug's impact on the complete tumor-immune interaction. It is the preferred model for evaluating systemic immunotherapies like ICIs and cancer vaccines.
- Genetically Engineered Mouse Models: Mice are genetically modified to spontaneously develop tumors that are more reflective of human cancer pathogenesis. Useful for studying the role of specific genes in tumorigenesis and the testing of targeted therapies in an immunocompetent environment.
- Humanized Mouse Models: They can recapitulate human immune responses, making them invaluable for testing human-specific immunotherapies. Ideal for evaluating the efficacy and toxicity of biologics and human-targeted therapies, including monoclonal antibodies and adoptive cell transfer therapies like CAR-T cells.
- Orthotopic Tumor Models: Provide a more anatomically relevant tumor environment and metastatic patterns compared to subcutaneous models. Useful for studying tumor metastasis, invasion, and site-specific immune responses.
- Organoid Models: Utilizing patient derived organoids as personalized 'avatars,' the model accurately assesses a patient's specific response to immunotherapies, thus directly guiding individualized treatment and supporting high-throughput drug screening.
Our Advantages
- Comprehensive Technology and Multi-Modality Support: Our services cover the entire preclinical pipeline, from in vitro assays to in vivo pharmacology. We possess expertise across a broad portfolio of therapeutic modalities, including immune checkpoint inhibitors, cell therapies (CAR-T/TCR-T), cancer vaccines, bispecific antibodies, and oncolytic viruses.
- Advanced and Clinically Relevant In Vivo Models: Our platform offers a comprehensive model matrix, including Syngeneic Models for immune-competent studies, Humanized Models (huPBMC, huCD34+) for human-specific therapy evaluation, and Patient-Relevant Models (huPDX, Tumor Organoids) to accurately mimic the patient-specific Tumor Microenvironment (TME).
- Expert Team and Facilities: Our dedicated team of Ph.D.-level immunologists and oncologists provides expert consultation. We utilize state-of-the-art laboratory facilities that support complex sample processing and the real-time, non-invasive tracking of anti-tumor responses.
- Mechanistic and Predictive Analysis: We possess strong translational science capabilities, utilizing multi-parameter flow cytometry for detailed immune cell profiling and a full suite of assays to generate mechanistically deep and highly predictive data, crucial for understanding the drug's Mechanism of Action (MoA).
- Flexibility, Customization, and Project Management: We possess the capacity to generate bespoke assays and models precisely tailored to the client's unique scientific query and therapeutic agent, ensuring efficient project management and precise results.
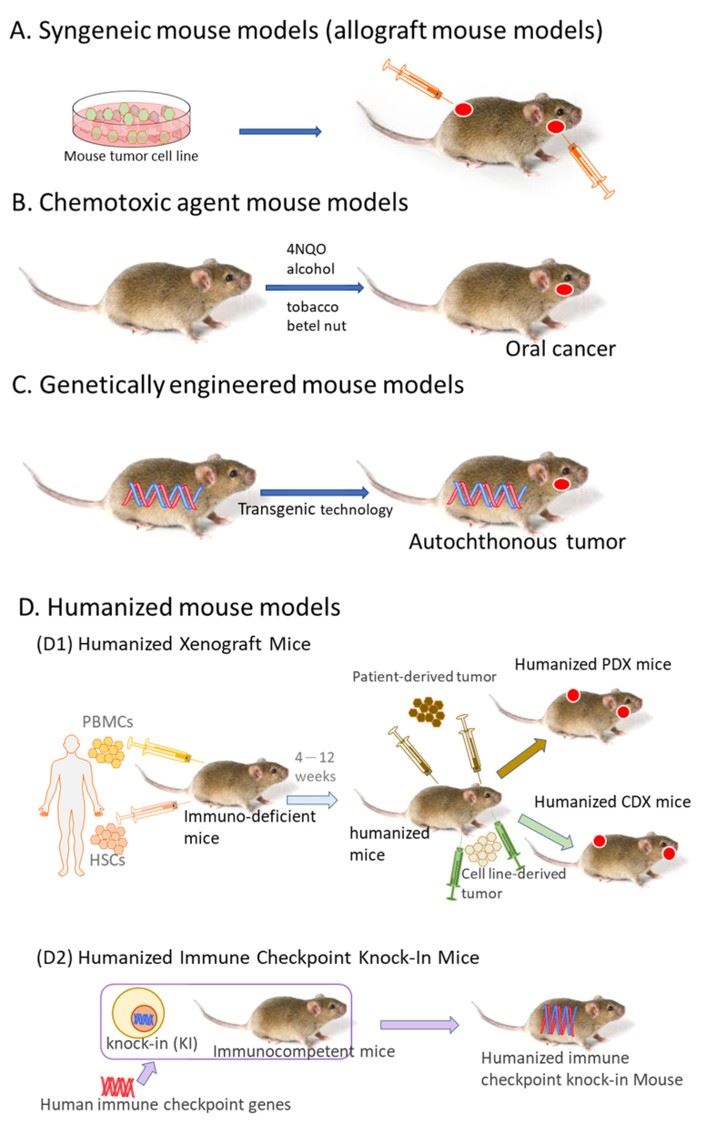 Fig.2 Murine models for tumor immunotherapy research.2,4
Fig.2 Murine models for tumor immunotherapy research.2,4
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q. What endpoints are typically measured to assess the efficacy of immunotherapies in animal models?
A: Endpoints include tumor growth inhibition, survival rates, immune cell infiltration, cytokine and chemokine profiles, and the expression of immune checkpoint markers.
-
Q. How is the immune response monitored in preclinical studies?
A: Immune responses are monitored through flow cytometry, immunohistochemistry, cytokine assays, and genomics and proteomics analyses to understand the activation and interactions of immune cell populations.
-
Q. Why is the tumor microenvironment significant in evaluating immunotherapy efficacy?
A: The tumor microenvironment influences immune cell function, drug delivery, and resistance mechanisms. Understanding these interactions is vital for optimizing therapeutic strategies.
-
Q. How can the pharmacokinetics of monoclonal antibodies differ from those of small molecules?
A: Monoclonal antibodies typically have longer half-lives, distinct distribution profiles, and unique elimination pathways compared to small molecules, often involving receptor-mediated processes.
-
Q. What challenges exist in evaluating pharmacokinetics in animal models?
A: Challenges include species-specific differences in drug metabolism and distribution, which may affect the extrapolation of results to humans.
-
Q. What are the primary objectives of toxicology studies in tumor immunotherapy?
A: The primary goals are to assess the safety profile, dose-response relationships, potential off-target effects, and the identification of toxicological endpoints in preparation for clinical trials.
-
Q. What types of toxicity are of particular concern in immunotherapy?
A: Special concerns include immune-related adverse effects such as cytokine release syndrome and off-target immune activation, which require careful monitoring and management strategies.
Published Data
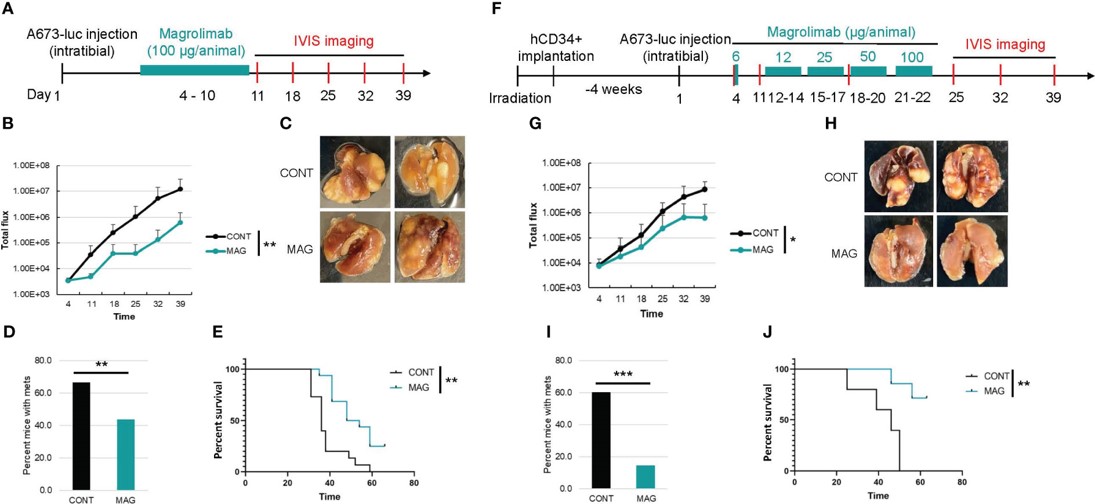 Fig.3 Comparison of the efficacy of CD47 blockade in non-humanized and humanized orthotopic mouse model of ES.3,4
Fig.3 Comparison of the efficacy of CD47 blockade in non-humanized and humanized orthotopic mouse model of ES.3,4
In the humanized mice, the interaction is between a human tumor and human macrophages. This fully human system is more potent, leading to a stronger anti-tumor effect.
References
- Teijeira Crespo, Alicia et al. "Pouring petrol on the flames: Using oncolytic virotherapies to enhance tumour immunogenicity." Immunology vol. 163,4 (2021): 389-398. https://doi.org/10.1111/imm.13323
- Chiu, Wei-Chiao et al. "Mouse Models for Immune Checkpoint Blockade Therapeutic Research in Oral Cancer." International Journal of molecular sciences vol. 23,16 9195. https://doi.org/10.3390/ijms23169195
- Luo, Wen et al. "A humanized orthotopic mouse model for preclinical evaluation of immunotherapy in Ewing sarcoma." Frontiers in immunology vol. 14 1277987. https://doi.org/10.3389/fimmu.2023.1277987
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
