Xenograft Modeling & Pharmacodynamics Services
Introduction
Xenograft models are foundational tools in cancer research, created by implanting human tumor tissue into immunodeficient mice. They play an irreplaceable role in developing anticancer drugs and studying tumor biology. Specifically, these models are critical for the preclinical assessment of drug toxicity, pharmacokinetics, and efficacy, serving as the necessary bridge before human clinical trials. Creative Biolabs focuses on enhancing these models to better recapitulate the human tumor microenvironment. We achieve this through the development of advanced Xenograft Models, the integration of multi-omics technologies, and the creation of innovative cross-species platforms. Leveraging decades of expertise in oncology drug development, Creative Biolabs is uniquely positioned to guide your therapeutic candidates through rigorous preclinical efficacy testing. These comprehensive efforts significantly improve the translational value of our models for precision oncology, making them essential tools for bridging basic research and clinical application.
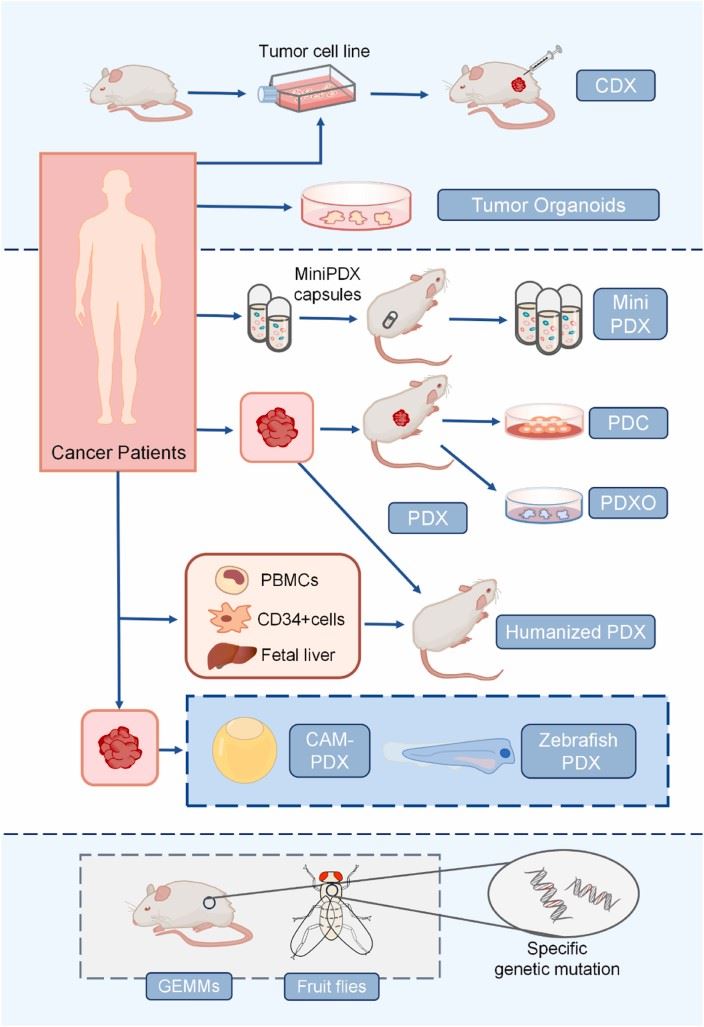 Fig1. Schematic representation of different preclinical models. PDO, patient derived organoids; Mini PDX, mini patient derived xenograft; GEMM, genetically engineered mouse model; PDC, patient derived cell cultures; CAM, chicken egg chorioallantoic membrane.1,3
Fig1. Schematic representation of different preclinical models. PDO, patient derived organoids; Mini PDX, mini patient derived xenograft; GEMM, genetically engineered mouse model; PDC, patient derived cell cultures; CAM, chicken egg chorioallantoic membrane.1,3
Available Xenograft Models
Creative Biolabs uses mice or rats with different degrees of immunodeficiency to establish Xenograft Models. For example:

| Xenograft Models | Modeling Method | Relevant Evaluation | Animal Species |
| Cell Line derived Xenograft (CDX) Models | In vitro cultured human tumor cell lines (e.g., A549, MCF-7, HCT116) are injected or implanted into immunodeficient mice. | Efficacy evaluation for traditional chemotherapies and small molecule targeted drugs (direct tumor cell killing). | Immunodeficient Mouse (e.g., Nude, SCID, NOG) |
| Patient derived Xenograft (PDX) Models | Fresh patient tumor tissue fragments are directly transplanted subcutaneously or orthotopically into highly immunodeficient mice. | Personalized drug screening, drug sensitivity testing, early-stage efficacy assessment for new drugs, and companion diagnostics development. | Severely Immunodeficient Mouse (e.g., NOG and derivatives) |
| Immunodeficient-Rat Xenograft Models | Human tumor cells or tissue are implanted into immunodeficient rats. | Larger tumor burden, suitable for imaging studies, radiotherapy evaluation, and PK/PD studies. | Immunodeficient Rat (e.g., Nude rat, RGD knock-out rat) |
Evaluation Platform
Leveraging our state-of-the-art technologies, we provide a unified and robust preclinical drug evaluation platform. Every stage of our analysis is engineered for maximum translational value, delivering results that are demonstrably reproducible and scientifically rigorous.
-
Basic Phenotype and Efficacy Monitoring Platform
-
Tumor Growth and Survival Assessment
- Tumor Volume / Weight Measurement (using calipers, electronic scales): Used for basic efficacy evaluation, for example, to calculate the Tumor Growth Inhibition (TGI) rate.
- Survival Rate and Body Weight Monitoring: Assesses drug toxicity (a body weight decrease of >10% suggests a risk of toxicity) and long-term survival benefit, such as the extension of the median Overall Survival (OS).
- In vivo Bioluminescence / Fluorescence Imaging (IVIS, Lumina): Provides real-time, dynamic monitoring of tumor growth and metastasis by fluorescently labeling tumor cells.
-
Tumor Growth and Survival Assessment
-
Histopathology and Molecular Phenotyping Platform
-
Histomorphology and Biomarker Detection
- H&E Staining: Observes tumor tissue morphology and its relationship with surrounding tissues.
- Immunohistochemistry (IHC): Detects tumor proliferation, apoptosis, angiogenesis, and specific target expression, verifying the drug's mechanism of action.
- Immunofluorescence (IF): Used for co-localization analysis and assessing immune cell infiltration.
-
Molecular-Level Analysis
- Real-time Quantitative PCR (qPCR) / Digital PCR (dPCR): Detects changes in gene expression.
- Western Blot / Protein Arrays: Analyze protein levels and phosphorylation modifications to verify pathway activation.
- Next-Generation Sequencing (NGS): Analyzes the heterogeneity of cells within the tumor microenvironment.
-
Histomorphology and Biomarker Detection
-
Immune Microenvironment Assessment Platform
-
Immune Cell Subtyping and Functional Detection
- Flow Cytometry: Analyzes the proportion and activity markers of immune cell subpopulations.
- ELISA, CBA, Luminex: Quantifies cytokines and chemokines in serum or tumor tissue to assess the degree of immune activation.
- Enzyme-Linked Immunospot (ELISPOT) Assay: Detects antigen-specific T-cell responses.
-
Immune Cell Subtyping and Functional Detection
-
Imaging Diagnosis Platform
-
High-Precision Structural and Functional Imaging
- Magnetic Resonance Imaging (MRI): Evaluates the size, infiltration range, and tissue edema of orthotopic tumors; features high resolution.
- Computed Tomography (CT): Detects lesions such as bone metastases or lung nodules.
- Positron Emission Tomography (PET): Uses tracers like 18F-FDG to assess tumor metabolic activity, reflecting the drug's suppression of tumor viability.
-
High-Precision Structural and Functional Imaging
-
Special Function Detection Platform
-
Pharmacokinetic (PK) and Pharmacodynamic (PD) Correlation Analysis
- LC-MS/MS: Detects drug concentration in tumors and blood. Combined with the tumor inhibition rate, it establishes a dose-exposure-effect relationship to optimize drug administration protocols.
-
Metabolomics and Lipidomics Analysis
- Core Technologies (GC-MS / LC-MS): Used to detect changes in tumor tissue metabolites, helping to understand the drug's effect on tumor metabolism.
-
Pharmacokinetic (PK) and Pharmacodynamic (PD) Correlation Analysis
Applications
- Disease Modeling & Translational Value: The CDX Model functions as a basic screening platform to quickly assess direct anti-tumor activity and preliminary toxicity, primarily simulating tumor growth associated with specific molecular targets. Conversely, the PDX Model is considered the gold standard for personalized oncology; it retains the original tumor's heterogeneity and molecular profile to the maximum extent, offering the highest translational value. PDX is key for companion diagnostics and drug sensitivity prediction to guide patient therapy.
- Advanced Model Development: The advancement of orthotopic implantation (e.g., patient derived orthotopic xenograft, PDOX) lies in its ability to more accurately simulate the complex tumor microenvironment, angiogenesis, and metastatic potential by placing the tumor in the organ of origin; whereas, the benefit of the immunodeficient-rat xenograft model is its larger size and tumor capacity, which significantly facilitates PK/PD studies, advanced imaging diagnostics (MRI/PET/CT), and radiotherapy evaluation for large tumors.
- Oncology Drug Development: The CDX Model serves as the classic PD assessment tool for traditional small molecule targeted drugs, chemotherapy, and radiotherapy, used to calculate the TGI rate and determine the optimal dose; while the more complex PDX and orthotopic models are utilized to validate the mechanism of action for new drugs in vivo and assess their impact on cancer stem cells and resistant clones to overcome acquired resistance.
- Tumor Mechanism Studies: Xenograft models are primarily utilized, often via fluorescence/bioluminescence labeling, for the real-time monitoring of tumor aggressiveness, metastatic pathways, and angiogenesis mechanisms; simultaneously, despite their immunodeficiency, these models can still be employed to study the roles of stromal cells, vasculature, and fibroblasts within the Tumor Microenvironment (TME), and when combined with humanized models, they can partially simulate the complexity of the human TME.
Our Advantages
- High-Quality Model Construction: Standardized xenograft model construction processes ensure a high model success rate (success rate ≥ 80%) and strong stability (reproducible tumor growth curves). Key model characteristics are precisely preserved, guaranteeing consistency between the model and clinical or research needs.
- Extensive Model Resource Library: A xenograft model library covering multiple cancer types and molecular subtypes allows for quick matching of a client's research needs, shortening the model preparation cycle.
- Advanced Technical Platform Support: Equipped with comprehensive in vivo evaluation technologies.
- Customized Model Construction: The ability to build specialized models, such as metastasis, drug-resistant, and orthotopic models, to meet the research needs for complex biological processes.
- Professional Experimental Design and Data Analysis: The ability to tailor experimental protocols to a client's research goals, such as efficacy evaluation, drug resistance mechanisms, toxicity testing, including sample size design, group allocation strategies, and selection of detection indicators. Provides in-depth data analysis services, interpreting efficacy results, such as tumor inhibition rate, prolonged survival, and immune microenvironment changes, in conjunction with model characteristics to support client decision-making.
- Efficient Service Response and Flexibility: CDX models are built within 1-2 weeks, and PDX models are successfully engrafted within 4-6 weeks, shortening the experimental cycle. Supports personalized adjustments, such as adding detection indicators or changing dosing regimens mid-experiment, to meet research needs from early screening to late-stage mechanism validation.
- Professional Technical Team with Accumulated Experience: A team of experts in fields like oncology, pathology, and pharmacology can provide end-to-end technical consultation, from model selection to result interpretation, to solve complex experimental problems. Extensive project experience and familiarity with the characteristics and common confounding factors of different models, such as immune rejection and spontaneous tumor regression, allow for proactive risk avoidance.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: How do you choose between CDX, PDX, Syngeneic, and Humanized Mouse Models?
In preclinical studies, model selection should be dictated by the research goal: for early-stage drug screening, the CDX model should be chosen; for precision medicine or mechanistic research, the PDX model is recommended; and critically, for the assessment of immunotherapy, a syngeneic or humanized mouse model must be selected to ensure a functionally intact immune background.
-
Q: What are the timelines and success rates for building Xenograft models?
The CDX model offers a higher success rate (over 95%) and a significantly shorter establishment period (2-3 weeks), whereas the PDX model requires a longer establishment period (8-12 weeks) with a success rate ranging between 60% and 90%.
-
Q: How is drug efficacy evaluated in Xenograft models?
Drug efficacy evaluation encompasses macroscopic assessments such as Tumor Growth Inhibition (TGI) rate, tumor regression, and prolonged survival; molecular analyses, including Ki67 proliferation, TUNEL apoptosis, and drug-specific target engagement analysis; and imaging techniques like IVIS in vivo imaging and MRI/CT for measuring the size of orthotopic tumors.
-
Q: How are failed cases handled?
We guarantee service quality by offering free re-inoculation in cases of tumor formation failure, and by reviewing the experimental process and providing optimization suggestions (such as dose adjustment or model change) if abnormal efficacy results are observed.
Published Data
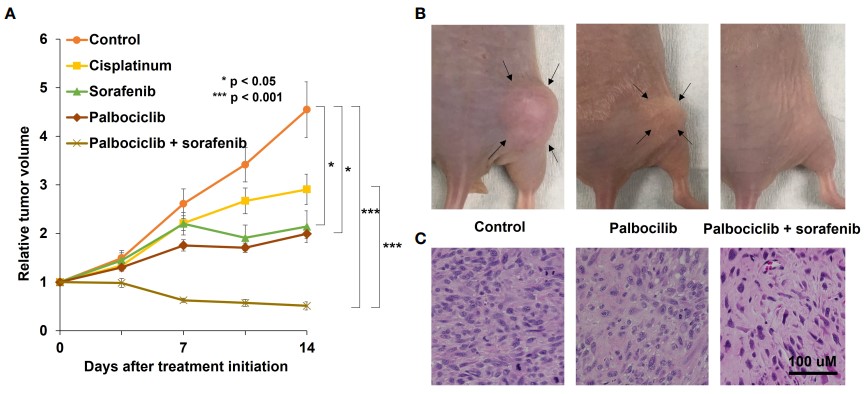 Fig. 2 A osteosarcoma PDOX study identifying an effective combination treatment with palbociclib.2,3
Fig. 2 A osteosarcoma PDOX study identifying an effective combination treatment with palbociclib.2,3
The AAV-sh-PPY and anti-PD-1 combination achieved a high survival rate of 83.3% and significantly prolonged survival. Crucially, tumors showed reduced metabolic activity (lower UVmax via PET-C). This synergistic efficacy validates the model's power in identifying potent combinations and accelerating oncology R&D.
References
- Liu, Minqi, and Xiaoping Yang. "Patient derived xenograft models: Current status, challenges, and innovations in cancer research." Genes & diseases vol. 12,5 101520. https://doi.org/10.1016/j.gendis.2025.101520
- Higuchi, Takashi et al. "Review: Precise sarcoma patient derived orthotopic xenograft (PDOX) mouse models enable identification of novel effective combination therapies with the cyclin-dependent kinase inhibitor palbociclib: A strategy for clinical application." Frontiers in Oncology Vol 12 957844. https://doi.org/10.3389/fonc.2022.957844
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
