CAR-T & CAR-NK Cell based Efficacy Modeling & Pharmacodynamics Service
Introduction
Chimeric Antigen Receptors (CARs) are synthetic immunoreceptors that redirect effector lymphocytes, such as T cells and NK cells, toward malignant cells. By genetically modifying these immune cells to express CARs, they are empowered to precisely recognize tumor-associated antigens and elicit a potent cytotoxic response, establishing CAR technology as a fundamental component of tumor immunotherapy. Leveraging a comprehensive background in oncology drug assessment, Creative Biolabs possesses the necessary expertise to support the development and evaluation of CAR-T, CAR-NK, and related cellular therapeutics. Specifically, we utilize highly predictive, integrated models that are built upon the specific characteristics of your CAR design, including the cellular vector, targeting strategy, manufacturing modality, functional enhancements, and signaling domain architecture. This comprehensive, tailored approach allows us to accurately assess therapeutic potency, optimize dosing regimens, and establish robust PK/PD correlations, accelerating your path to clinical success.
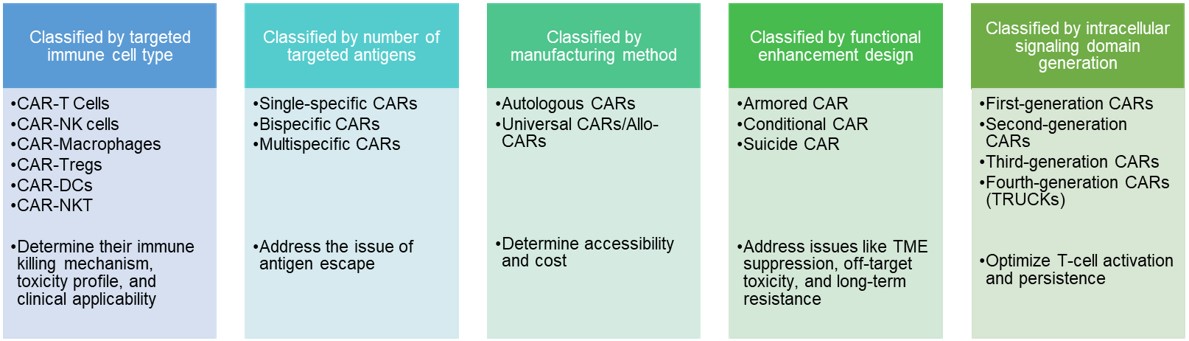 Fig.1 Differentiation of CAR-engineered immune cells.
Fig.1 Differentiation of CAR-engineered immune cells.
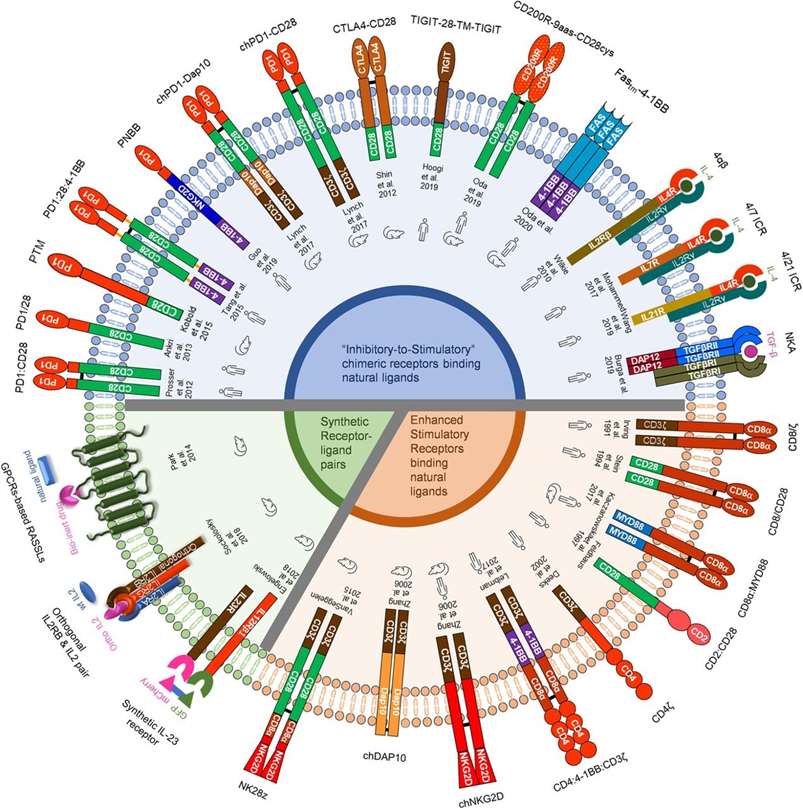 Fig.2 Schematic diagrams of chimeric non-antigen receptors for T cell-engineering in cancer therapy.1,4
Fig.2 Schematic diagrams of chimeric non-antigen receptors for T cell-engineering in cancer therapy.1,4
Available CAR-T & CAR-NK Cell based Efficacy Services
The efficacy evaluation of the major CAR-modified immune cell types exhibits significant differences, stemming from their intrinsic biological characteristics (proliferation, persistence, killing mechanism), mode of action (cytotoxic, regulatory, antigen-presenting), and clinical application (oncology, autoimmunity, transplant tolerance). We offer a systematic, mechanistic-driven approach to compare and design optimal efficacy studies across five critical dimensions, focusing on actionable experimental design logic for each cell type:
| Types | Core Assessment Goal | Key Efficacy Metrics | Preferred Animal Models | Typical Application Scenarios |
| CAR-T Cells | Evaluate in vivo anti-tumor activity, cell persistence, and toxicity (e.g., CRS, ICANS). | Tumor Volume or Burden change, Survival Rate, CAR Cell Expansion & Persistence, Cytokine Profile. | PDX/CDX models (immunodeficient mice, e.g., NOG), Syngeneic models (for immune microenvironment assessment). | Treating Hematological Malignancies (e.g., B-ALL, DLBCL), and solid tumor exploration. |
| CAR-NK cells | Assess non-MHC-restricted anti-tumor activity, safety, and immediate killing potential. | Tumor Volume change, Survival Rate, NK Cell Infiltration, Toxicity/Safety (often safer than CAR-T). | PDX/CDX models (immunodeficient mice), Humanized Immune models (if using human derived NK cells). | Treating Solid Tumors, Hematological Tumors (relapsed/refractory), and Allogeneic therapy platforms. |
| CAR-Macrophages (CAR-M) | Evaluate cell phagocytic activity, tumor microenvironment remodeling, and solid tumor penetration capability. | Tumor Volume change, Macrophage Polarization (M1/M2), Tumor Infiltration depth, Systemic Toxicity. | PDX/CDX solid tumor models (e.g., pancreatic cancer, breast cancer), Ectopic Implantation models. | Treating Solid Tumors, clearance of tumor stroma, and metastases. |
| CAR-Tregs | Assess immunosuppressive activity and therapeutic effect on autoimmune diseases or Graft-versus-Host Disease (GVHD). | Disease Scoring (Clinical Score), Inflammatory Biomarkers, Treg Cell Proportion & Function, Transplant Organ Survival. | GVHD models, Autoimmune Disease models (e.g., EAE, T1D). | Treating Autoimmune Diseases, Organ Transplant Rejection, and Inflammatory Diseases. |
| CAR-DCs (CAR-Dendritic Cells) | Evaluate the ability to present antigens, activate endogenous T cells, and induce long-term immune memory. | T Cell Activation markers, Cytokine secretion, Antigen-Specific CTL Response, Long-Term Immune Memory. | Syngeneic models, Humanized immune system models. | Cancer Vaccine development, Activation of Host Immunity against tumors. |
| CAR-NKT | Assess the ability to bridge innate and adaptive immunity, lower GVHD risk, and exhibit multi-target killing. | Tumor Volume, Survival Rate, NKT Cell Infiltration and Expansion, Cytokine release. | PDX/CDX models, Syngeneic models. | Treating Hematological and Solid Tumors, exploring Allogeneic applications. |
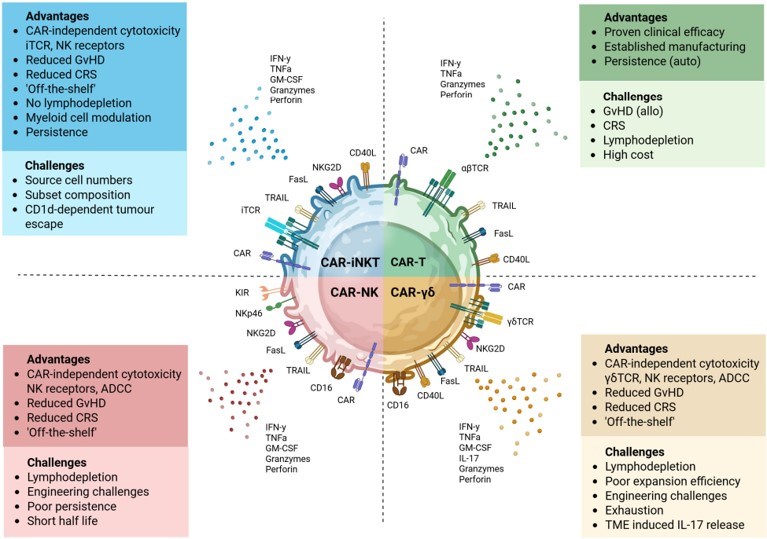 Fig.3 Key attributes of CAR-engineered lymphocyte platforms.2,4
Fig.3 Key attributes of CAR-engineered lymphocyte platforms.2,4
Evaluation Platform
We utilize an integrated hierarchy of models, selecting them specifically to predict biological outcomes across different phases of development:
- Xenograft Models: These represent the most common and foundational platforms for CAR cell efficacy testing, employing human tumors grown in immunodeficient mice. Patient derived Xenograft (PDX) models utilize patient tumor tissue, preserving the tumor's genetic and histological characteristics for high clinical relevance. In contrast, Cell Line derived Xenograft (CDX) models inject established human tumor cell lines, offering high standardization and reproducibility.
- Syngeneic Models: Though less common for human CAR cells, the use of mouse tumors in immunocompetent mice uniquely enables the study of interactions between CAR-engineered cells and the host's endogenous immune system.
- Humanized Models: Humanized models are specifically designed to bridge the gap between animal studies and human clinical outcomes. These systems aim to reconstitute a partial or complete human immune system within mice, providing a more robust and predictive environment that better simulates the complex behavior and functional dynamics of CAR cells within the human body.
- Tumor Organoids: Tumor organoids overcome the inherent limitations of traditional 2D culture dishes by providing a high-throughput, high-fidelity platform. This 3D cellular structure closely mimics the in vivo tumor microenvironment (TME) and architecture, making organoids an increasingly valuable tool for early-stage evaluation of CAR efficacy and mechanism of action.
- Gene Editing Models: These specialized models are crucial for mechanistic studies and target validation. By using mice with specific genes knocked out or humanized, these models allow researchers to precisely evaluate the functional impact and safety of CAR cell genetic modifications (e.g., removing inhibitory checkpoints) on in vivo efficacy and toxicity.
-
Preclinical Efficacy Assessment: We employ multi-modal technologies to achieve precise monitoring of complex biological processes:
-
Key Efficacy Metrics: We move beyond simple tumor shrinkage by focusing on metrics that drive clinical success, anchored by robust quantitative analysis:
- Tumor Burden and Survival: These metrics, including Tumor Volume, Tumor Growth Inhibition (TGI), and Overall Survival (OS), directly quantify a drug's anti-tumor strength and long-term efficacy.
- Drug Metabolism and Effect: We establish a quantitative relationship between Pharmacokinetic/Pharmacodynamic (PK/PD) data and Concentration-Time-Effect (C-T-E) to build models that guide clinical dose selection based on exposure and efficacy.
- Immune Microenvironment: Assessment of immune cell infiltration (e.g., CAR cells, CD8+ T cells), cytokine profiles (e.g., IFN-γ, IL-6), and checkpoint expression is crucial for deciphering the drug's Mechanism of Action (MOA) and evaluating its immune modulation effect.
- Molecular Biomarkers: By analyzing target expression levels, pathway activation status (e.g., Western Blot, ELISA), and resistance genes, we predict high-responding patient populations and guide the necessary biomarker development.
- Safety and Toxicity: This dimension focuses on evaluating the safety window by determining the Maximum Tolerated Dose (MTD), tracking body weight changes, assessing organ toxicity (e.g., liver/kidney function), and predicting Off-Target Toxicity.
-
Advanced Assessment Methods: We employ an integrated suite of advanced technologies for precise monitoring and deep mechanistic study:
- Multi-Dimensional In Vivo Imaging: PET, SPECT, and MRI enable precise, non-invasive, and longitudinal monitoring of tumor volume, activity, cell tracking, and pharmacokinetic distribution.
- High-Throughput Molecular Analysis: High-Throughput Molecular Analysis, including NGS/qPCR and Mass Spectrometry, is utilized to evaluate genetic changes (gene expression, mutation burden, pathway regulation) and perform full-component bioanalysis of XDC drugs and biomarker discovery.
- Histological & Cellular Analysis: Histological and Cellular Analysis, including IHC/IF and Flow Cytometry, provides precise localization and quantification of targets, cell infiltration, mechanism-of-action proteins, and detailed phenotyping of immune cell subsets in both tumors and peripheral blood.
-
Key Efficacy Metrics: We move beyond simple tumor shrinkage by focusing on metrics that drive clinical success, anchored by robust quantitative analysis:
Applications
- CAR Design and Optimization: We use advanced modeling to optimize the CAR's structural components, including signaling domains, spacer length, and novel architectures (e.g., tandem CARs). This predictive approach enhances cell persistence and function while minimizing exhaustion.
- Preclinical Efficacy Assessment & Screening: We conduct rapid, high-fidelity screening using diverse models (PDX, Organoids) to quantify cell killing kinetics, potency, and specificity. We efficiently prioritize lead candidates and identify indications resistant to antigen loss.
- PK/PD Characterization & Modeling: We go beyond traditional PK by modeling the quantitative "dose-expansion-effect" relationship. This tracks CAR cell expansion, functional persistence, and trafficking in vivo, providing the critical data needed to determine the optimal clinical infusion dose and schedule.
- Safety Assessment & Risk Prediction: We conduct rigorous safety modeling across all phases: Off-Target & Controllability is assessed using GEMM models to predict off-target toxicity risk and validate inducible safety switches for strict in vivo control. Simultaneously, we predict Immune-Related Toxicity by utilizing Cytokine Storm Modeling to simulate the risk of CRS/ICANS (Cytokine Release Syndrome/Immune Effector Cell-Associated Neurotoxicity Syndrome) and guide the design of preventative pre-treatment regimens.
- Combination Therapy Design & Validation: We rigorously model and quantify the synergistic potential of CAR therapies with immune checkpoint inhibitors, targeted drugs, and chemotherapy. This helps optimize dosing sequences and combination ratios to enhance infiltration and improve overall remission rates.
- Novel CAR Applications: We offer specialized modeling expertise for advanced platforms, including assessing the safety and scalability of CAR-NK cells, evaluating the immunosuppressive function of CAR-Tregs (for autoimmune diseases), and analyzing the TME-remodeling capacity of CAR-Macrophages.
Our Advantages
- Comprehensive Animal Model Platforms: We leverage expertise across a diverse model library and utilize specialized toxicology models to provide the most suitable efficacy and safety platforms based on your specific research stage and goals.
- Extensive Project Experience and Rigorous Management: Our investigative group consists of seasoned experts who provide strategic consultation and technical resolution, while the implementation of standardized SOPs ensures the accuracy, traceability, and high reproducibility of all resulting datasets and high-quality reporting.
- Customized Experimental Design and Flexibility: We precisely tailor experimental frameworks to accommodate distinct client specifications, offering robust capability for swift modification and providing extensive preclinical consultation to ensure that the selected research trajectory meets prespecified project objectives.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: What endpoints are used to measure efficacy?
A: Primary outcome measures encompass tumor growth inhibition (TGI), the rate of neoplasm regression, and overall survival (OS). Supplementary readouts commonly integrate Bioluminescence Imaging (BLI) for longitudinal in vivo tumor burden tracking and flow cytometry for the quantification of immune effector subpopulations.
-
Q. How can I ensure the reliability and reproducibility of my results?
A: Model Quality: The integrity of preclinical findings is predicated on the mandatory and systematic quality assurance of both PDX and humanized murine platforms. Cell Quality: Preceding in vivo application, the therapeutic cellular product necessitates stringent validation: viability, purity, and the precise cellular phenotype must be empirically verified utilizing standardized in vitro assays. Standardized Procedures: To minimize inter−operator variability, all operational phases, encompassing therapeutic administration, neoplasm quantification, and biomaterial procurement, must strictly conform to predefined Standard Operating Procedures (SOPs).
-
Q. How much time and cost are involved in the evaluation process?
A: This depends on the complexity of the model you choose and the evaluation metrics. CDX models are typically the fastest and most economical option, with preliminary results available within a few weeks. PDX and humanized models have a longer setup and evaluation period, often taking several months or longer, which results in higher costs.
Published Data
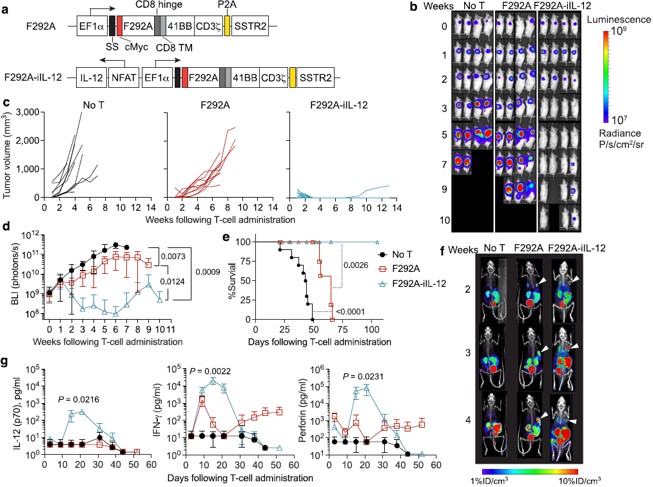 Fig.4 Schematic diagram of the vector, bioluminescence, individual tumor volume, whole-body bioluminescence kinetics, survival curve, PET-CT, and serum cytokine results.3,4
Fig.4 Schematic diagram of the vector, bioluminescence, individual tumor volume, whole-body bioluminescence kinetics, survival curve, PET-CT, and serum cytokine results.3,4
By analyzing the proportion and infiltration density of immune cells and the cytokine profile within tumor tissues, we demonstrated that CD47-CAR Macrophages not only directly phagocytose tumor cells but also significantly enhance CD8+ cytotoxic T cell infiltration and reduce the proportion of immunosuppressive cells. This achieves a synergistic anti-tumor effect through both direct killing and adaptive immune activation, validating its value in remodeling the TME.
References
- Guo, Jitao et al. "Chimeric non-antigen receptors in T cell based cancer therapy." Journal for immunotherapy of cancer vol. 9,8 (2021): e002628. https://doi.org/10.1136/jitc-2021-002628
- Niedzielska, Magdalena et al. "CAR-iNKT cells: redefining the frontiers of cellular immunotherapy." Frontiers in immunology vol. 16 1625426. https://doi.org/10.3389/fimmu.2025.1625426
- Yang, Yanping et al. "Inducible expression of interleukin-12 augments the efficacy of affinity-tuned chimeric antigen receptors in murine solid tumor models." Nature Communications vol. 14,1 2068. https://doi.org/10.1038/s41467-023-37646-y
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
