Humanized Mouse based Tumor Modeling & Pharmacodynamics Services
Introduction
Creative Biolabs has developed a diverse portfolio of Humanized models, which overcome the limitations of traditional systems by accurately simulating human immune responses. They provide a highly predictive platform for preclinical evaluation, enabling researchers to analyze immune escape mechanisms and guide personalized treatment strategies.
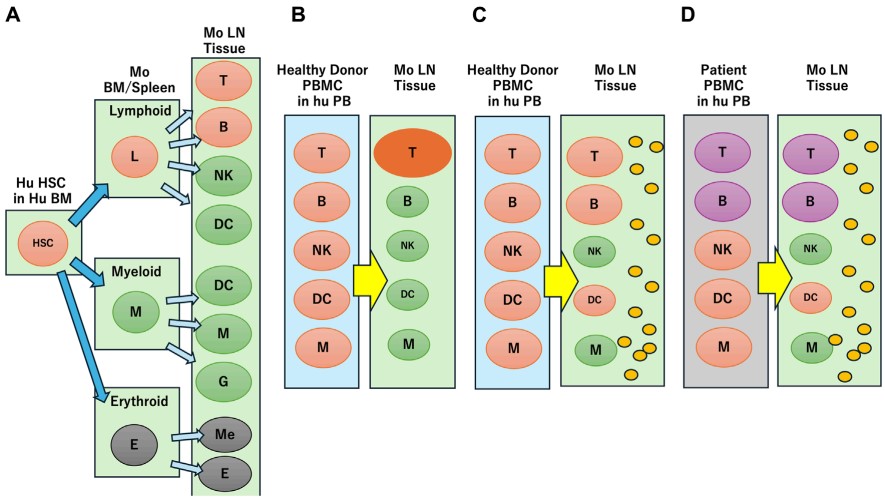 Fig.1 Reconstruction of human immune profiles in an immune humanized mouse.1,5
Fig.1 Reconstruction of human immune profiles in an immune humanized mouse.1,5
Available Humanized Models
Creative Biolabs' humanized tumor models are centered on their ability to accurately replicate human tumor reality, and their applications now extend across the entire spectrum of oncology research, from dissecting mechanisms to achieving clinical translation.
| Humanized Models | Modeling Method | Relevant Evaluation |
| Humanized PDX (hu-PDX) Mouse Models | Engrafting human tumor tissue into immunodeficient mice (e.g., NOG) reconstituted with human immune components (e.g., PBMCs or HSCs) | Short-term efficacy assessment for T-cell-dependent immunotherapies (e.g., CAR-T cell therapy, bispecific antibodies). |
| Humanized CDX (hu-CDX) Mouse Models | Engrafting human cancer cell lines (CDX) into immunodeficient mice reconstituted with human immune components (e.g., PBMCs or HSCs) | Evaluation of immuno-oncology drugs, including checkpoint inhibitors (PD-1/PD-L1), CAR-T cell therapy, and antibody based therapeutics. |
| Humanized Tumor Target Mouse Models | Utilizing gene editing technology to replace specific mouse genes (e.g., immune checkpoint genes PD-1, CTLA-4, CD47) to express the human homologous target protein. | Evaluation of the efficacy and toxicity of monoclonal antibodies, ADCs, and bispecific antibodies that target human-specific proteins. |
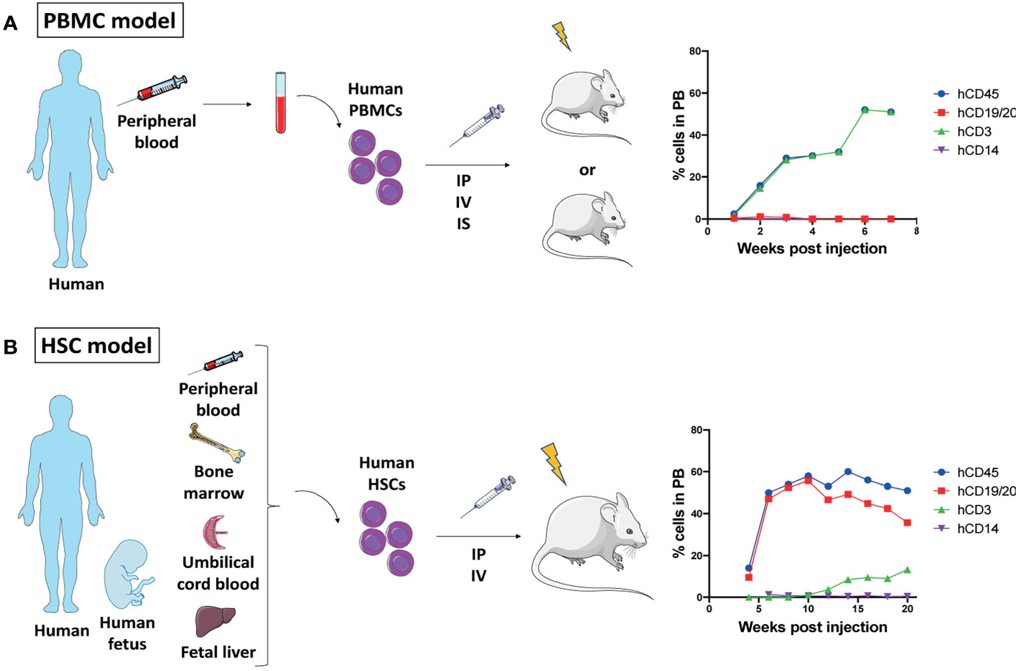 Fig.2 Mouse humanization models.2,5
Fig.2 Mouse humanization models.2,5
Evaluation Platform
Our Humanized Tumor Models are evaluated through a state-of-the-art detection platform meticulously designed to deliver both scientific rigor and high translational value. We go beyond basic efficacy measurements, employing integrated, multi-dimensional analytical tools to ensure your data is both accurate and reproducible.
- Tumor Growth Inhibition and Survival Analysis: Directly measure changes in tumor volume and calculate the Tumor Growth Inhibition (TGI) rate. Evaluate the effect of treatment on extending survival using survival curves.
- Immune Response and Microenvironment Remodeling: Detect immune cell infiltration, cytokine levels, and the expression of immune checkpoint molecules to verify the activating or inhibitory effects of immunotherapy, revealing the drug's mechanism within the Tumor Microenvironment (TME).
- Drug Safety and Toxicity Assessment: Monitor blood biochemical indicators, histopathological changes, and immune-related adverse reactions (irAEs) to comprehensively evaluate drug safety, particularly the potential side effects associated with immunotherapies.
- Imaging Evaluation: Dynamically monitor tumor size (via CT/MRI), metabolic activity (via PET scans using metrics like SUVmax), and metastasis distribution to assess treatment response.
- Flow Cytometry (FACS): Quantitatively analyzes the proportion and activity markers of tumor-infiltrating immune cells, serving as the most direct and crucial method for verifying the mechanism of action of immunotherapy.
- Molecular Biology Assays: Utilize technologies like RNA Sequencing, Proteomics, and Single-Cell Analysis to deeply analyze the tumor's gene expression profiles, the activation status of signaling pathways, and the cellular characteristics of the immune microenvironment, providing high-dimensional mechanistic data.
Applications
- Tumor Biology and Mechanistic Research: Our tumor models provide unprecedented accuracy to deeply analyze tumor initiation and progression, intrinsic heterogeneity, metastatic pathways, and drug resistance mechanisms.
- Precision Medicine and Personalized Therapy Guidance: Our tumor models drive the individualized therapy revolution, enabling patient-specific drug sensitivity testing, precise resistance prediction, and providing the foundation for optimal treatment regimen optimization.
- Anti-Cancer Drug Development: Our tumor models serve as the core bridge for preclinical validation, spanning drug target validation, precise efficacy evaluation, efficient combination therapy screening, and the entire process of accelerated clinical translation.
- Immunotherapy Research: Our tumor models are utilized for cutting-edge therapies such as immune checkpoint inhibitors, CAR-T cell therapy, and tumor vaccines, conducting in-depth mechanistic analysis and authoritative efficacy assessment.
- Translational Medicine and Clinical Translation: Our tumor models accelerate biomarker discovery, guide clinical trial patient enrichment, and establish reliable prognostic prediction models.
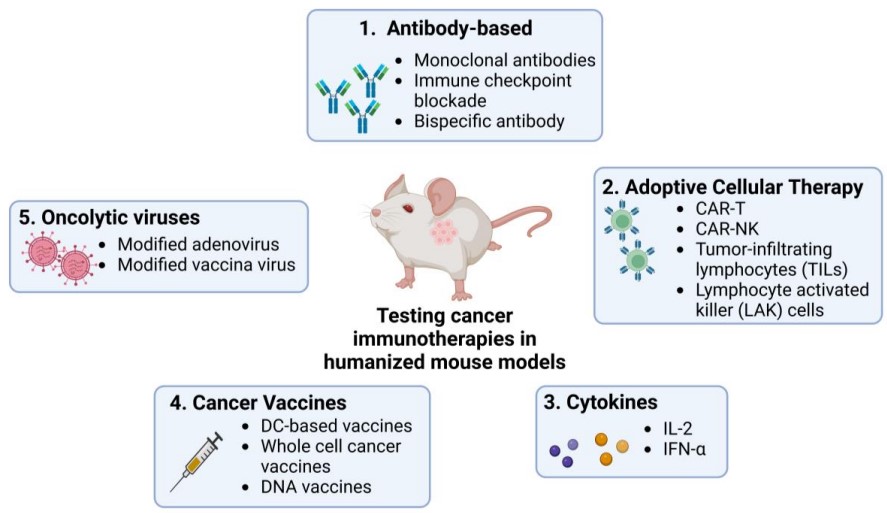 Fig.3 Use of humanized mouse models to test current immunotherapies in cancer research.3,5
Fig.3 Use of humanized mouse models to test current immunotherapies in cancer research.3,5
Our Advantages
- Highly Simulating the Human Immune Microenvironment: Providing a near-human experimental environment for immunotherapy evaluation, retaining tumor heterogeneity and immune infiltration characteristics, and significantly improving the accuracy of efficacy prediction.
- Broad Disease Model Coverage: We have developed a portfolio of models covering over ten major cancer types, including both solid tumors and hematological malignancies.
- Strict Quality Control and Standardized Procedures: Regular technical assessments and external expert evaluations reduce the impact of human operational variations on results.
- Customized Solutions: We provide differentiated model configurations tailored to your specific research needs: for short-term studies evaluating treatments like CAR-T or anti-PD-1, PBMC models allow for rapid study initiation (2–4 weeks); conversely, for long-term mechanistic studies requiring immune memory and chronic disease simulation, HSC models are recommended, utilizing a longer 12–20 week cycle.
- One-Stop Service: Our one-stop screening platform, featuring an extensive PDX model library and primary cell bank, accelerates the development of new anti-tumor drugs.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
Q: What is the core difference between humanized and traditional animal models?
A: Humanized models use gene editing or cell transplantation (like HSC/PBMC) to reconstruct a human immune system or tumor microenvironment in an immunodeficient animal.
-
Q: How do you choose between PBMC, HSC, and dual-humanized models?
A: PBMC models are suitable for short-term immunotherapy evaluations, such as CAR-T toxicity testing. They have a quick turnaround (1-2 weeks), but are prone to GvHD and have a short study window of just 4-8 weeks. HSC models are better for long-term studies. They have a more complete immune reconstruction (T/B/NK cells) and suffer from significantly less or delayed GvHD, but the timeline is much longer (12-20 weeks). Dual-humanized models (e.g., HU-PDX) involve the simultaneous implantation of tumor and immune cells. They are used to predict the efficacy of immune checkpoint inhibitors and have a clinical consistency of 87%.
-
Q: Is customized gene editing supported?
A: Yes, disease-causing mutations can be customized for testing gene therapy drugs and targeted drugs.
-
Q: What cancer research problems can humanized models solve?
A: Our models can accurately predict immunotherapy efficacy via Hu-PD-1 models, helping to avoid clinical failures; concurrently, by combining them with single-cell sequencing, we can deeply analyze drug resistance mechanisms and reveal immune escape pathways in the tumor microenvironment; furthermore, individualized patient derived xenograft models enable individualized patient drug screening, directly guiding the establishment of clinical treatment plans.
Published Data
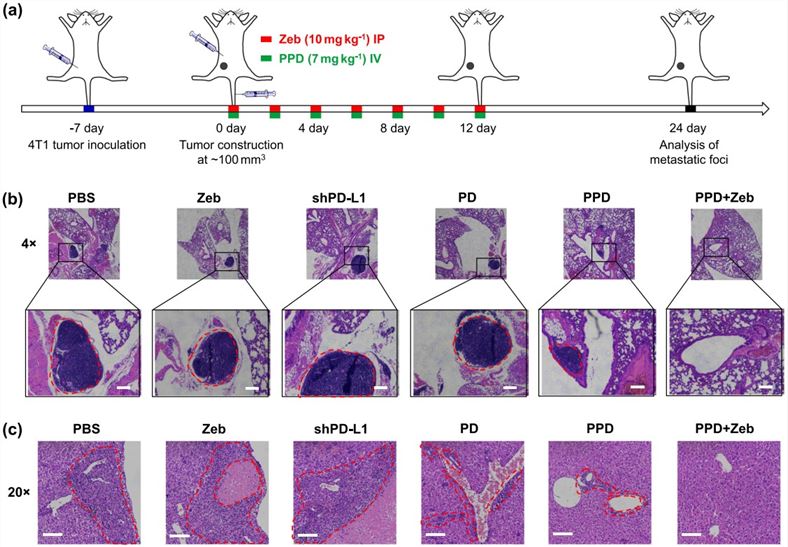 Fig.4 H&E staining of lung and liver metastatic foci.4,5
Fig.4 H&E staining of lung and liver metastatic foci.4,5
The 4T1 triple-negative breast cancer model demonstrated the profound efficacy of the combination therapy: mPEG-b-PLG/PEI-RT3/DNA + Zebularine completely inhibited lung and liver metastasis, a stark contrast to the significant metastatic foci observed in the control group. This outcome definitively validates the 4T1 model as a reliable and sensitive platform for evaluating anti-metastatic therapeutic strategies.
References
- Kametani, Yoshie et al. "PBMC-engrafted humanized mice models for evaluating immune-related and anticancer drug delivery systems." Frontiers in molecular biosciences vol. 11 1447315. https://doi.org/10.3389/fmolb.2024.1447315
- Cogels, Morgane M et al. "Humanized Mice as a Valuable Pre-Clinical Model for Cancer Immunotherapy Research." Frontiers in Oncology Vol 11 784947. https://doi.org/10.3389/fonc.2021.784947
- Karnik, Isha et al. "Emerging Preclinical Applications of Humanized Mouse Models in the Discovery and Validation of Novel Immunotherapeutics and Their Mechanisms of Action for Improved Cancer Treatment." Pharmaceutics vol. 15,6 1600. https://doi.org/10.3390/pharmaceutics15061600
- Fang, Huapan et al. "Combination of epigenetic regulation with gene therapy-mediated immune checkpoint blockade induces anti-tumour effects and immune response in vivo." Nature communications vol. 12,1 6742. https://doi.org/10.1038/s41467-021-27078-x
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
